Journal of Veterinary Science & Medicine
Download PDF
Research Article
Exploration Behavior of Pet Dogs During Off-Leash Walks
Foltin S1and Ganslosser U2
1Department of Applied Zoology, University Duisburg-Essen,
Germany
2Institute of Zoology and Evolutionary Research, Friedrich Schiller
University, Jena, Germany
*Address for correspondence: Foltin S, Dorstener Str. 525 46119 Oberhausen Germany; Tel: 49-2086988177, Email: sfoltin@web.de
Submission: 30 March, 2021;
Accepted: 1 May, 2021;
Published: 5 May, 2021
Copyright: © 2021 Foltin S et al. This is an open access article
distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Introduction
The majority of domestic dogs (approximately 80 % of the global
dog population)are considered feral or free roaming and live in a
human-dominated niche [[1,2]. Pet dogs therefore represent only a
fractional part of the entire dog population. In the main, these dogs
live either in enclosed properties or their exercise takes place in a daily
round of walks with their owner, often on a leash [3]
. This may raise health and welfare concerns [4-7]
.
In numerous studies the close bonding, reliance, attachment
and resulting specific behavior of domestic dogs with respect to
their human owner has been demonstrated[8-12]
therefore we hypothesized that dogs have high motivation based on this strong
bonding to return to their owner, regardless of breed, area or external
stimuli.
The majority of current literature proposes that domestic dogs,
primarily due to domestication, have lost or have a reduced ability
of spatial orientation compared to wolves [13,14]
. We hypothesized,however, that off-leash pet dogs will find back to a specified, varying
non-stationary locality (i.e. their owner) in different environments.
Even subsequent to having explored a certain distance i.e. out of
range of visibility or/and olfaction, based on their ability to establish
cognitive maps and the use of spatial reference systems [15,16]
.Herein all of the dogs found their respective owners on all runs >20m.
Exploration is important for animals to be able to gather information about features of their environment that may directly
or indirectly influence survival and reproduction [17]
as it enablesthem to collect information about food distribution, shelters or
escape routes [18]
. Exploratory behavior is considered as an aspect
of sensory processing involved in investigating novel stimuli, rather
than an instinctive behavior [19]and partially depends on motor and
spatial capabilities and on the motivation to explore [20]. Studies
have demonstrated a linkage between learning, memory, exploratory
behaviors and genetics [21,22], and breed differences have been
proposed [23,24]. Spatial orientation may either be maintained
allocentrically by memorizing specific landmarks (e.g. roads, trees,
etc.), positions and locations in known locations [15] in which the
dog updates its position in the environment using a reference system
external to the body and anchored in the environment [25]. Dogs may
also orient egocentrically by integrating signals or cues indicating
the extent of self-motion along their locomotion trajectory [26], in
which the dog updates an object’s location with respect to its own
body, using a reference system centered on the body, typically defined
by the reference directions of front, back, right and left [26,27]. We
therefore hypothesized, that dogs, depending on their individual
traits, character, learning experience, breed, socialization and age
would utilize different exploration patterns.
The aim of this study was to garner data about the factual
exploratory behavior of pet dogs. This is in particular important
because of the strict regulations and prejudices currently existing and
their implications for domestic pet dogs, specifically their physical
and psychological welfare and the impact on cognitive abilities and
functions by being frequently walked on a leash [28,29].
Taking into account legal restrictions in many countries as well as
the preconceptions often voiced i.e. by the hunting community that
pet dogs will chase prey, it is important to ascertain how dogs actually
explore whilst off the leash. Furthermore, recent studies showing the
result of Covid-19 restrictions on dogs underline the detrimental
effect of leash walking, even within a short period of weeks or month
[28,30].
Results herein might be useful in establishing areas where off leash
dog walking is allowed, help dog-trainers to get more information about exploratory behaviour of pet dogs and may benefit in designing
walking areas, as dog walking is a popular method for increasing
human physical activity [31,32]. The omnipresence of the domestic
dog inspired many scientific endeavors, but research on pet dog
walks mainly focused on applicable aspects of health effects for the
owner, epidemiology [33], or has been conducted in enclosed areas
or laboratory settings [34-36].
To our knowledge, no research has actually been done to
ascertain how domestic pet dogs in fact do behave whilst off the leash,
unrestricted and in an unenclosed area.
Materials and Methods
GPS data were collected on trials (n = 3145) of free ranging,
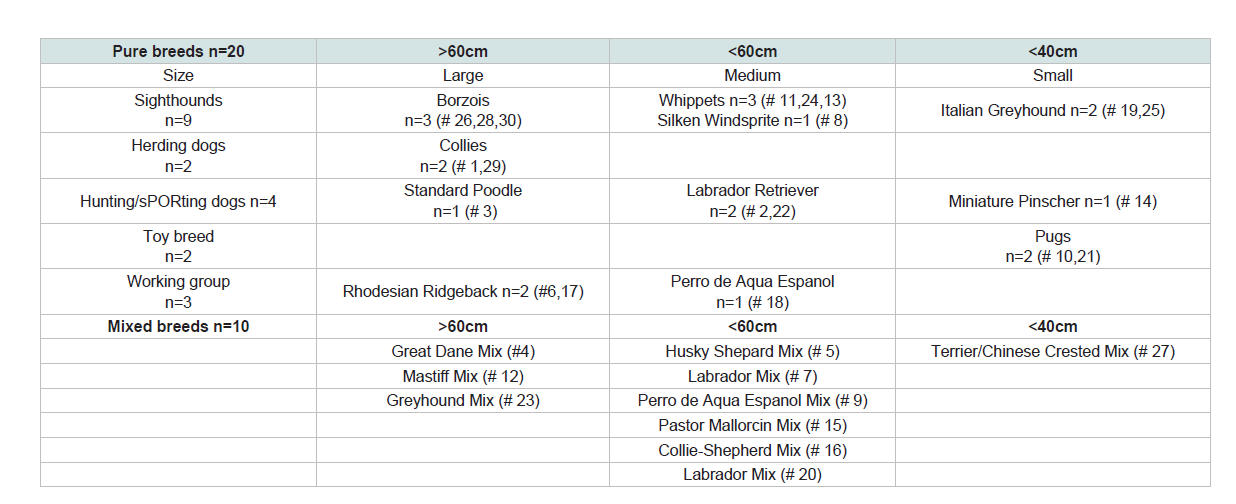
freely exploring domestic pet dogs (n=30) of different breeds, size,
reproductive status, sex and age (Table 1) while walking with their owners on four consecutive walks in two known and two unknown
areas in North Rhine Westphalia, Germany (n=120). A dog had to
travel a minimum distance of 20m away from the owner to be recorded
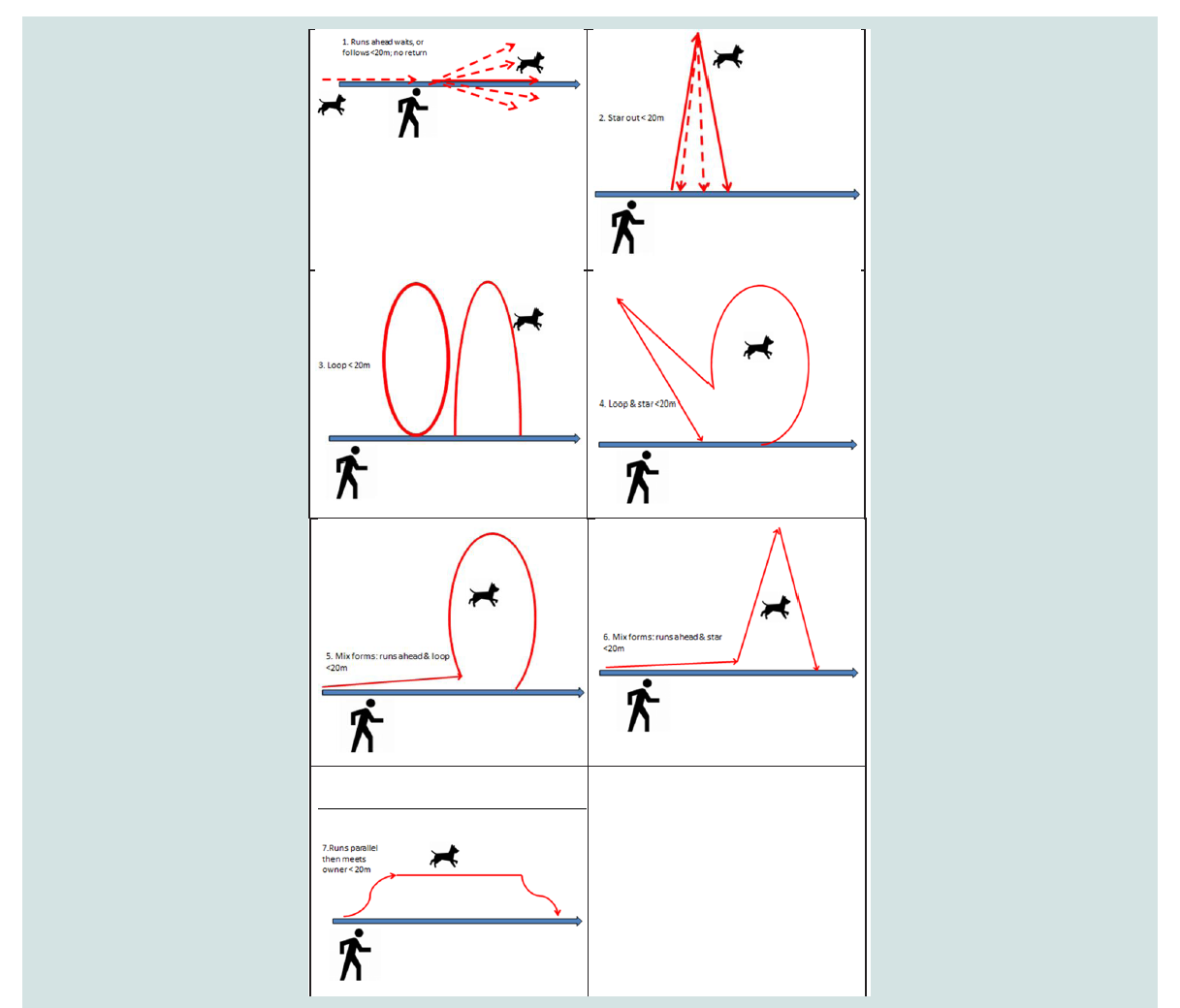
as a run, and seven different travelling patterns were distinguished,
see Figure 1, pictograms. Lengths of the walks depended on age and
physical ability of the dog, the average length was mean 89 min. +/- 24
min per walk.
A total n=120 measurements with an n=3145 total runs >20 m
resulted. There were 18 different owners, 15 female (83 %) and three
male (17 %). 83 % of the owners had more than one dog. 17 % owned
one dog. Out of the 30 dogs eight belonged to a single dog household
owner, 22 to a two or more dog owner – in this group ten owners
accounted for 22 dogs. The median age of the dogs was 63.5 months.
40 % of the dogs were male (n=12), of which 75 % of were neutered
and 60 % were female (n=18), of which 72 % were spayed; of all dogs and the owner carried a hand held GPS device (Garmin Astro® 320) to
determine the distance between dog and owner. The margin of error
for the Astro® 320 is within +/- 3.65 m. Dog collar details: The DC™
50 weighs 289g (sender; antennae and collar); the size is (B x H x
T): 9 x 4.9 x 4.6 cm; distance of recording is up to 14.5km. The T5
weights 198g (sender; antennae and collar); size (B x H x T): 8.9 x 4.4
x 4.7 cm; distance of recording up to 12km. The Video camera used
was a Garmin VIRB® Elite. Size: (H x B x T): 32 mm x 53 mm x 111
mm; weight 170g. Datatype: MP4; 1080p-HD-Video: 1920 × 1080; 30
fps. Video data were also displayed on Garmin BaseCamp™4.5.2.1. The
camera, however, was only used on the larger dogs as it was too heavy
and difficult to attach to the small dogs.Wind speed and direction was
measured with an anemometer (Technoline EA-3000) and handheld
compass. Ambient temperature was also recorded therewith.
Trials were performed in wooded areas, preferably with dense
underbrush to prevent visual contact. If two dogs from one owner
participated in any given trial data from both dogs, as visualized
on Garmin BaseCamp™, were used and compared, individually and
grouped. Solely uninhabited areas, without roads or major pathways,
were visited. The owner was not to whistle or call or offer any other
kind of acoustic or visual signal. Seven travelling patterns were
differentiated for each run > 20m: 1. Dog runs ahead and waits/
follows; 2. star; 3. loop; 4. Loop + star; 5. Mix forms: runs ahead &
loop; 6. Mix forms: runs ahead & star; 7. Runs parallel then meets owner. Regarding the travelling patterns, see Figure 1 for pictograms.
Descriptive analysis was performed calculating number of
valid measurements (n), mean (m), median, quartiles and standard
deviation (SD). Relationships were plotted using scatterplots, bar
charts, boxplots or mean +/- 95 % confidence interval. Nonparametric
tests were used for inductive statistics. Mann-Whitney U Test was used
to compare two independent samples, Wilcoxon test for dependent
samples of ordinal data. Also randomization (or permutation) tests
were applied for comparison of dependent or independent groups of
interval scaled data.
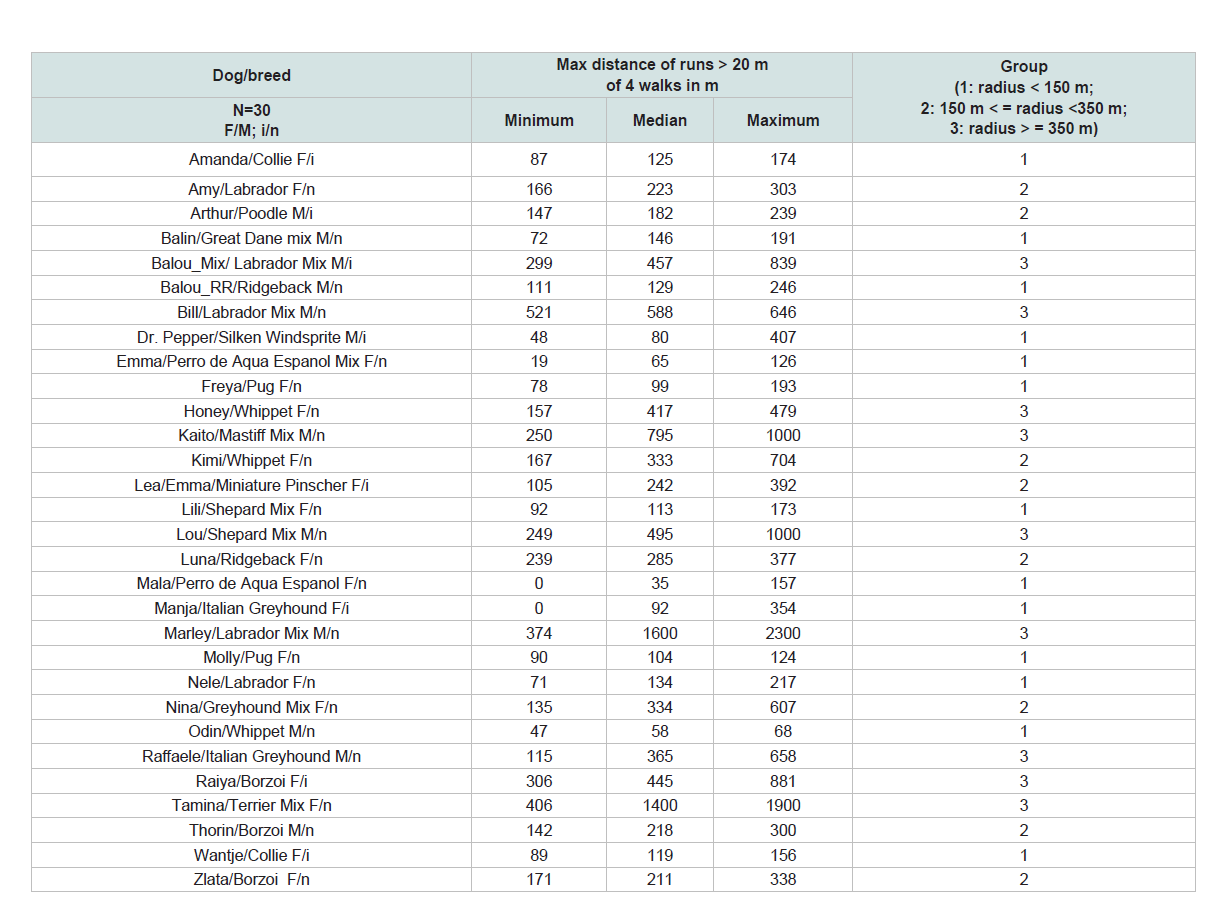
Table 2: Each dog minimum, maximum and median distance of runs >20 m, Grouped: Group 1: radius < 150 m; Group 2: 150 m < = radius <350 m; Group 3: radius > = 350 m
Hierarchical cluster analysis was applied to find walks with similar
running patterns and to find the appropriate number of clusters.
Squared Euclidian distance was used as measure of distance and
Ward method was used as linkage method. K-Means clustering with
three Clusters was applied to assign all walks to the cluster. A Cluster
analysis is neither parametric nor non-parametric as the algorithms
are based on exploratory data mining.
All tests were performed two-tailed on a 5 % level of significance.
Standard Bonferroni correction of p-values < 0.05 was applied in case
of multiple testing. Two-tailed tests were performed unless otherwise
denoted. SPSS version 25, IBM Inc. was used for analyzing the
data. StatKey (http://www.lock5stat.com/) was used for performing
randomization tests using a simulated sample of size n= 5000.
Ethical Approval
All applicable international, national, and/or institutional
guidelines for the care and use of animals were followed.
Results
All dogs, independent of sex, age, reproductive status or breed,
found and returned to their owner in different environments
subsequent to having travelled at least 20 m away from their owner.
Length of trials (runs > 20 m) varied greatly, therefore three groups
were established to determine travelling patterns in more detail and
describe difference between the dogs more specifically: Group 1:
radius < 150 m maximum distance from owner travelled by the dogs;
Group 2: 150 m = 350 m distance from owner.
Data demonstrated great intraspecific differences: 50 % of the
dogs showed a median difference in walking distance to their owner
of 1000 m, with a lower quartile of 400 mand an upper quartile of
2300 m difference between owners and dog distance walked, which
corresponds to a 43 % increase. The distance difference travelled was
significantly larger for dogs compared to owners (p<0.001). Dogs of
all three groups travelled longer distances than their owners.
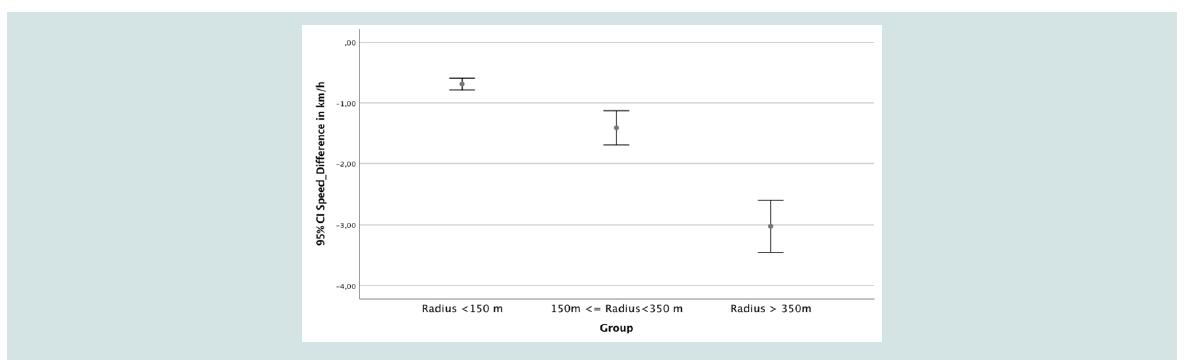
Results also showed significant speed difference between owners and dogs (p<0.001; Wilcoxon Test).The mean speed of owners
was 4.4 +/- 0.4 km/h compared to 6.0 +/- 1.2 km/h of the dogs,
independent of the group the dogs belonged to or factors like age, size
or exploration patterns see (Figure 2). Altogether 50 % of the owners
displayed a walking speed of less than 4.4 km/h; 50 % of the dogs had
a walking speed of less than 5.7 km/h. A quarter of the owner had a
travelling speed of less than 4.2 km/h while the lower quartile of the
dogs ‘speed was 5.2 km/h. The higher quartile of owner walking speed
was 4.7 km/h; compared to the dogs travelling speed of 6.6 km/h. The
total median speed difference amounted to -1.1 km/h; mean 1.6 km/h.
Speed differences could also be established between the three groups,
with Group 3, the dogs with the largest radius, displaying the highest
speed differences compared to their owners, see (Figure 2) (in km/h).
Great intraspecific differences could be perceived between the
maximum median distances of runs > 20 m explored by each dog
(Table 2).The majority of dogs exhibited a median maximal difference
of less than 150 m away from the owner (13 of 30 dogs =43 % Group
1); eight (27 %) of the dogs displayed a median maximal difference
away from the owner of less than 350 m (Group 2); nine of the 30
dogs (30 %) had at least one run over 350 m away from the owner
(Group 3).
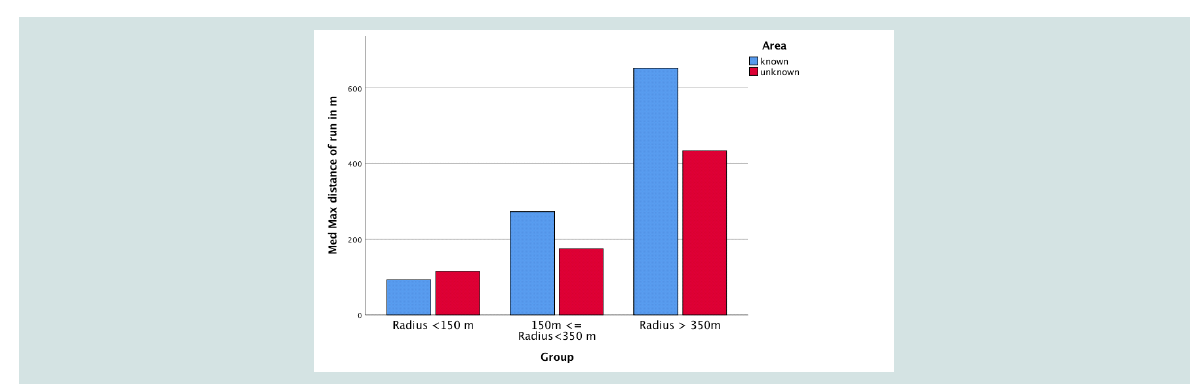
For each group the median of the maximum distance away
from the owner of runs >20 m in known as well as unknown areas was assessed. Looking at the median of the total runs >20
m in known and unknown areas, values of Group 1 were almost
identical (known 572 m/unknown 435 m) (p = 0.796, Mann-
Whitney-U Test). In Group 2 dogs travelled shorter distances in
unknown areas (known 3101 m/ unknown 2524 m – reduction
19 %, p = 0.491, Mann-Whitney-U Test). Group 3 however shows
the clearest reduction in travelling distance of runs >20 m from
known 5709 m to unknown 4378 m (decrease 23 %, p = 0.126, Mann-
Whitney-U Test), Figure 3. For no group a significant reduction could
be demonstrated however. Between the three groups differences in
exploration time could clearly be seen, with an increase from Group 1
to Group 3. All groups displayed longer exploration times of runs >20
m in known than unknown areas.
Figure 3: Median of maximum distance in meter all runs >20 m; grouped; known (blue) versus unknown (red) area.
The dogs exhibited various travelling patterns. Out of all runs >
20 m (n= 3145) the dogs displayed n=1950 runs, 62 % the travelling
pattern of running ahead of the owner on the path and waiting/
following (see Figure 1 for pictogram). The star pattern was used
n=589 runs; 19 % of the time, thus the dogs basically returned on
their path. A loop was displayed n=291 runs; 9 % of the time and the
mixed forms together n=95 runs; 3 % of the time. Parallel runs to the
owner were used n= 192; 9 % of the time.
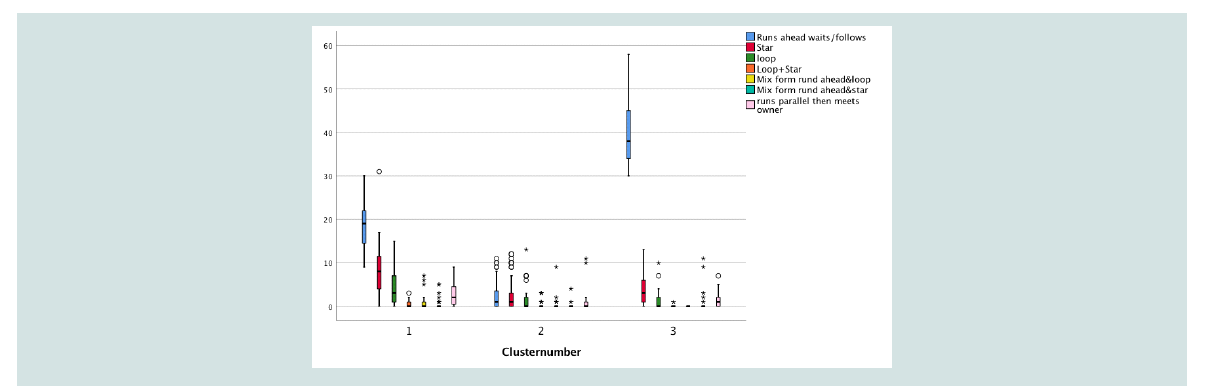
Based on the great individual variance displayed by the dogs
a hierarchical Cluster analysis using Ward linkage and squared
Euclidean Distance measure was applied to establish an appropriate
number of clusters. Based thereon a dendrogram was drawn. To
determine the optimal number of clusters a trade-off between
heterogeneity within the clusters and the number of clusters had to be
made. Here a number of three clusters for classifying all walks (total)
seemed an appropriate trade-off – For a distribution of patterns
between the cluster groups see Figure 4. Walks belonging to Cluster 3
were dominated by the pattern runs ahead waits/follows. Star and runs
parallel could also be observed. All other patterns were rare. Cluster 2
showed a low total number of runs, the most frequent patterns were
loop, star and runs ahead. Cluster 1 was mainly characterized by the
patterns runs a head waits/follows, followed by star, loop and also
parallel runs and mixed forms. The number of runs ahead was less
compared to Cluster 3.
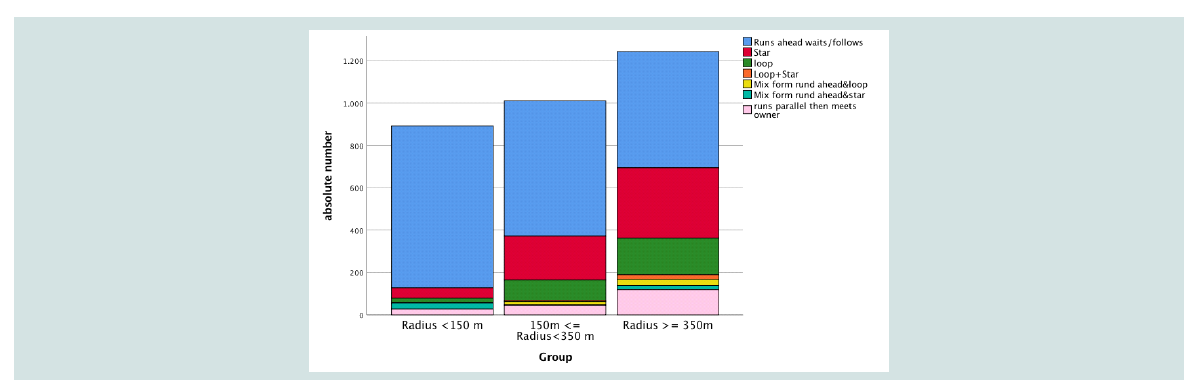
For the Distribution of patterns between Groups 1, 2 and 3
and travelling patterns of each group see Figure 5. Group 1 dogs
predominantly displayed the travelling pattern of running ahead and
waiting or following the owner with little variance of other travelling
patterns. With increasing distance of runs (Group 2 and 3) the dogs
displayed a greater variance of exploration patterns including more
cognitively challenging patterns like loops or mixed forms. Dogs
of different age and breed were represented in the different groups
as well as clusters. Group 1 dogs were underrepresented in Cluster
1 (observed: 8 vs. expected 19) and overrepresented in Cluster 2
(observed: 30 vs. expected 22) and 3 (observed: 14 vs. expected 11).
In other words dogs with a low maximal distance away from their
owner could be characterized by Cluster 2 (nearly no runs away
from owner) or Cluster 3 (high frequency of runs forward/backward
and waits). Dogs with a radius >350 m were over represented in
Cluster 1 (observed: 21 vs. expected 13) und underrepresented in
Cluster 3 (observed: 2 vs. expected: 8). Therefore these dogs may be
characterized by exploration patterns of star and loop and a medium number of runs ahead waits/follows.
It has been postulated that sex differences exist in regard to
exploration distance between male and female dogs, therefore the
distance of runs >20 m with regard to sex was analyzed. The variance
between female and male dogs with respect to the distance explored of
runs >20 m was significant (p = 0.003; Mann-Whitney-U Test). Male
dogs explored significantly longer distances than female dogs. The
mean distance of runs >20 m was larger for male dogs (3464 +/- 2732
m), than for female dogs (1862 +/- 1739 m) by an increase of 86 %.
Analyzing the duration explored by female and male dogs’ similar
results became apparent. The duration over all runs >20m was longer
in male dogs (22 +/- 16 min.) than in female dogs (13 +/- 16 min.).
The difference in the duration of runs > 20 m between female and
male dogs was also significant (p = 0.001; Mann-Whitney-U Test).
Discussion
Pet dogs growing up in our complex environment generally have
ample learning opportunities of how to interact and communicate
with humans, thus garnering experiences enhancing their cognitive
skills, an ontogenetic process called “enculturation” [26]. Studies
postulate an augmenting effect of domestication on the social skills
of dogs in cooperative–communicative tasks [37] like a social walk
with their owner, impacting on exploration behavior; on the other
hand it has been argued that as a result of domestication dogs’
spatial memory capacity has been reduced [38],. Our results reflect
intra specific movement variability, which has also been found in free
ranging dogs (see Hudson et al., 2017; 2019: three roaming patterns
“Stay-at-home dogs; “Roamer dogs” and “Explorer dogs”).
Sex differences have been postulated with respect to spatial
[39]. Results herein are in accordance with expected socioecological
differences, which encompass an extended home range for male
compared to female dog. Male dogs explored significantly further
and longer than female dogs. It has been suggested that female dogs
are more social in inter specific interactions with humans [40]
which may be an additional factor for female dogs staying closer to
the owner [41,42]than male dogs. Furthermore it has been argued
that male dogs are bolder, thus more proactive and explorative than
female dogs indicating a potential to explore further or longer.
Regarding their exploration patterns dogs herein were divided
into different groups and different clusters: Those dogs that explored
the longest and furthest (in known and unknown areas) displayed the
largest variety of exploration patterns, indicating cognitive variability,
experience and different strategy (allocentric and egocentric) use.
Another important factor impacting movement patterns were the
owners: Dogs form bonds with specific humans and make decisions by
attending preferentially to social signals from them [43]. Attachment
is the highest within the owner-dog dyad [44,45]. Therefore only
owner-dog dyads were used in this study as the motivation to return
was assumed the highest. Dogs displayed behaviors indicative of an
attachment relationship, for instance proximity seeking, where the
dog will seek the owner as a means of coping with stress [46,47], or
the safe haven effect [48] in which the presence of the owner may
reduce the effect of a stressful event.
It has been suggested that domestication has equipped dogs with two abilities prerequisite for cooperation—namely social tolerance
and social attentiveness, enabling them to adjust their behavior to
that of their social partners [49]. Social attentiveness, that is, paying
sufficient attention to one’s partners in order to adjust behavior and
thus to cooperate [50] would form the basis of attending to their
owner whilst walking, i.e. the dogs had to pay attention to movement
and location (and speed) of the owner in order to find him/her again
in addition to establishing a cognitive map of their own position in
space and time.
The Group 1 dogs stayed the closest to their owner and spent the
shortest time away, predominantly displaying the exploration pattern
of running ahead of the owner and waiting or following, arguably the
least cognitive challenging pattern and not requiring great orientation
skills.
Exploration distance and how a dog seeks support in challenging
or new situations during the walks may have been impacted by the
type of dog-owner attachment. Links between owner attachment
style and dog behavior imply that dogs develop different strategies
to handle particular situations, based on the type of support they
receive from their owner and influenced by the owner’s care giving
strategy as experienced in previous interactions [51]. Fearfulness or
anxiety for instance, thus an inhibitor of exploration, has been linked
to lack of experience and aversive learning strategies [52]. The extent
of exploration of an animal is balanced against its level of neophobia.
Neophilic animals are quick to approach and explore a novel object,
while neophobic animals are slow to do so [53]. Neophobic responses
are important because they reduce exposure to danger but, on the
other hand, they also constrain explorative behavior and thus
opportunities for learning and cognitive development. Using the
analog of the parent – child relationship it has been established that
two parenting dimensions have been consistently associated with
the development of anxiety in children: overprotection and anxious
rearing [54,55] . Overprotection has been described as parental
behaviors aimed at guiding children during their daily activities
thereby reducing the development of autonomy [56]. The exploration
(or lack thereof) behavior of Group 1 dogs might thus be influenced
by a deficit of cognitive development in that these dogs did not have
opportunities to explore, develop orientation strategies and garner
experiences, in particular as compared with Group 3 dogs [48]
postulated that an owner who is supporting the dog’s attempts to
independently deal with problems (or herein explore) leads to more
confident dogs. This may apply to Group 2 and particularly Group
3 dogs, who were secure enough to explore away from their owner
at significant distances and who had previously learned appropriate
strategies to find their owner again.Group 1 dogs displayed little
cognitive variance regarding exploration patterns and, independent
of known or unknown area, essentially ran ahead of the owner or
followed on the path. Group 3 dogs on the other hand exhibited a
wide range of strategies. Arguably having been able to learn sovereign
exploration behavior and thus becoming more autonomous, they
developed self-reliant movement resources and greater cognitive
orientation applications. This furthermore required memorization
of the spatial and temporal relations between the individual and
multiple goals e.g. the owner and different landmarks [56]. Memory
encompasses the acquisition, encoding, storage and retrieval of
information [57]. Retrieval of memorized information is context dependent i.e. using past experiences for decision making processes
and subsequent behaviors, again implicating owner provisions. Thus
within this context dogs would have been in similar contexts before
(free-ranging) to collect information (past experiences) to establish
subsequent exploration patterns.
In young animals spatial information is first used to encode
egocentric spatial memory and subsequently allocentric memory
[58]. Encoding memories requires learning periods [59], which
occur via social or cultural transmission, for instance through the
owner or conspecifics. Furthermore capacity, duration and precision
of memories are salient because memories will deteriorate unless
reinforced and maintained (use it or lose it). Memories should be
particularly valuable in landscapes of intermediate complexity like
the ones herein, where remembering several locations and their
attributes would be sufficient to accrue benefits like finding and
returning to the owner through efficient navigation or timely returns
as displayed by the dogs herein. Depending on the dogs’ biographies
and motivation, they investigated their environment with different
latencies and for variable periods displaying great variability. Other
biological or psychological variables, like breed or age, may of course
compound this effect. Data obtained herein reflects furthermore that
all dogs travelled significantly longer distances and at significantly
higher speed whilst off leash compared to their owner, see Table 1,
independent of the group the dogs belonged to or factors like age, size
or exploration pattern.
The importance of regular exercise for dogs is well known in
helping maintain the dog at a healthy weight and ensure it is less
susceptible to psychological conditions such as depression [60].
Each dog has an individual walking pattern, based on size, breed,
age and idiosyncrasies, its gait being defined by step frequencies,
velocity, length and width of step. To be on a leash restricts the
normal walking pattern of a dog, as the dog has to subsume its gait
pattern to that of its owner, which affects the function of the nervous
and/or the musculoskeletal system and disrupts their interactions,
possibly leading to gait disturbances [61]. A gait is formed through
complex interactions between the musculoskeletal and the central
and peripheral nervous system, where ambulation requires constant
adaptation to both intrinsic and extrinsic factors and dogs display a
wide range of step frequencies [62]. The biomechanical idiosyncrasies
of the dog gait may not be warranted while on the leash, no matter
how careful the owner.The leash and even the leash side influence
gait symmetry [64] entailing welfare concerns for those dogs often
on a leash. In their study [30] reported that German dog owners were
running with their dog (18.1%; mean 21 ± .61 h/week) and bicycle
riding (20.4%19 ± .53 h/week) which also shows the amount of
inappropriate dog exercise raising animal welfare concerns. Similarly
popular activities like agility and canicross have been associated with
injuries and health [63,64].
Conclusion
Taking into account our current perspective on what constitutes
a “good/obedient” dog (including the legal regulations) the Group
1 dogs would probably be considered thus, which may raise further
questions with regard to dog welfare, cognitive development
and learning issues in our society [65]. All dogs displayed a high
motivation arguably based on a strong bonding to return to their owner, regardless of breed, area or external stimuli whilst off the
leash, with a majority of dogs merely running ahead of their owner or
following. This strengthens the argument that dogs should be given
more opportunities to walk off leash and to establish safe areas where
they may explore. Overall dogs did display the cognitive ability of
spatial orientation to find their way back to the owner, in known and
unknown areas, regardless of distance explored. Dogs utilized various
exploration patterns, with those animals exploring furthest exhibiting
the greatest cognitive variability, thus signifying the importance of
early exploration opportunities being offered enabling the dogs to
gather practice. Popular dog-related on leash activities might raise
animal welfare concerns in that they disregard the dog’s individual
walking pace as needed for their physical and physiological health as
well as cognitive development.
References
Citation
Foltin S, Ganslosser U. Exploration Behavior of Pet Dogs During Off-Leash Walks. J Veter Sci Med. 2021;9(1): 9