Journal of Veterinary Science & Medicine
Download PDF
Research Article
Evaluation of Factors Affecting Pregnancy Rate after Cervical Insemination of Dairy Ewes in Greece
Priskas S, Termatzidou SA, Gargani S, Arsenos G
Laboratory of Animal Husbandry, Aristotle University of
Thessaloniki, Greece
*Address for Correspondence Priskas S, Faculty of Veterinary Medicine, Laboratory of Animal Husbandry, School of Health Sciences, Aristotle University of Thessaloniki, 54124, Greece; E-mail: stpriskas@vet.auth.gr
Submission: 04-November, 2019
Accepted: 09-December-2019
Published: 11-December-2019
Copyright: © 2019 Priskas S, et al. This is an open access article
distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Abstract
The objective of this study was to assess the factors affecting success
of cervical Artificial Insemination (AI) with chilled semen in intensively
reared dairy ewes in Greece. The study involved 1,785 adult ewes from
20 flocks of Lacaune and Chios sheep. A typical estrous synchronization
protocol, including intravaginal placement of progestogen sponge for 14
days and injection of equine chorionic gonadotropin at sponge removal,
was applied in ewes during mating period. All ewes were cervically
inseminated 53-57 hours after sponge removal with chilled semen
(15oC) collected from 10 adult purebred Lacaune rams. Pregnancy
Diagnosis (PD) was performed by trans-dermal ultrasonography at 35-
38 d after AI. The following data were available for each ewe: breed;
parity; Body Condition Score (BCS) at sponge placement, at AI and at
PD; presence of rams during synchronization and number of previous
synchronizations. Recordings during the AI procedure included: onset
of synchronization to AI interval; semen collection to AI interval; semen
deposition depth; cervical mucus presence; order of ewe; average
time required for each animal; ram semen used. The results showed
that farm, parity, semen deposition depth, ram, ewe BCS and changes
of BCS during the sponge placement-PD period, significantly affected
AI success (P<0.05). Overall fertility was 44%. Ewes of 2nd (50.3%) and
3rd lactation (48.1%) had the highest conception rates, that declined
as age increased. Ewes in moderate BCS (2.50-3.50) at onset of
synchronization (48.4%) and AI (49.4%) had significantly higher fertility
than ewes with low (<2.50) or high (>3.50) BCS. Positive energy balance
following the onset of synchronization seems to benefit animals with low
and moderate BCS, but decrease pregnancy rates of high BCS ewes.
In conclusion, selection of appropriate ewes and rams, BCS evaluation
prior to synchronization and adjustment of dietary management are key
factors dictating pregnancy rates following AI in greek dairy sheep.
Keywords
Sheep; Insemination; Fertility; Body condition score; Factors
Introduction
Assisted reproductive technologies in farm animals are used to
cater the needs for higher productivity and better quality of products.
In sheep production, the use of Artificial Insemination (AI) has
enabled the rapid introduction of valuable genes that improved
production traits and prevented disease transmission [1].
However, in comparison to other food producing animals, the
implementation of AI in sheep globally, is relatively limited [2,3]. The
only exception is France, where more than 410,000 inseminations
are performed annually in both nucleus and commercial dairy flocks
of the Lacaune breed [4]. An obstacle to the widespread use of AI
in sheep is the structural complexity of the ewe cervix that prevents
deep deposition of semen in uterus and leads to poor fertility rates
when frozen-thawed semen is used for cervical AI [5,6]. The notion is
that fertility rates can be enhanced by the application of laparoscopic
insemination, but the procedure has increased costs, requires
personnel with technical skills and raises welfare concerns. Using
chilled semen for cervical AI enhances fertility, but increases semen
production cost, has time limitations during transportation and often gives irregular results, since the success of the method is affected by
many factors. Environmental conditions, management factors, health
of males and females and physiological status of ewes are among the
factors that need to be controlled before AI implementation [7-11].
Dairy sheep industry is of significant importance to national
economy of most Mediterranean countries and Greece is one of
them [12]. With around 6.6 million milking sheep, Greece is ranking
2nd on milking sheep population and 1st on ewe milk production
(670,000 tons)inside EU28, comprising the 45% of national milk
production [13]. Despite its importance, greek dairy sheep industry
is characterized by relatively low productivity and is in a transitional
phase of gradual intensification [14]. Usage of higher milk-production
breeds, like the native Chios and the imported Lacaune breed, and
application of better management techniques are becoming more
common among farms. Nevertheless, application of AI for the genetic
improvement of the animals is not used often and, despite the fact
that some breeding programs are in operation, they are supported
on the usage of home-bred or imported rams [15]. Although AI is
commercially available in most areas, the high irregularity on fertility
results makes difficult its widespread application
Our objective was to carry out an artificial insemination
programme to study the factors affecting the success of cervical
Artificial Insemination (AI) with chilled semen in intensively reared
dairy ewes in Greece. It is the first study that assesses the effect of
female, ram, AI procedure conditions and farm nutrition program
at the same time on fertility rates after AI on intensively reared dairy
sheep in the area.
Materials and Methods
Animals: The study was conducted during the usual breeding season in
Greece, from May to November, for two consecutive years (2017-
2018). A total of 1785 adult ewes (1247 Lacaune and 538 Chios) were
used from 20 commercial flocks located in North and Central region
of Greece. Selected ewes belong to the most common intensively
reared breeds in Greece and were born and raised in the above
regions. During the study, the animals were at the 5-7th month of
their lactation with an average daily milk yield of 1.32±0.22 liters.
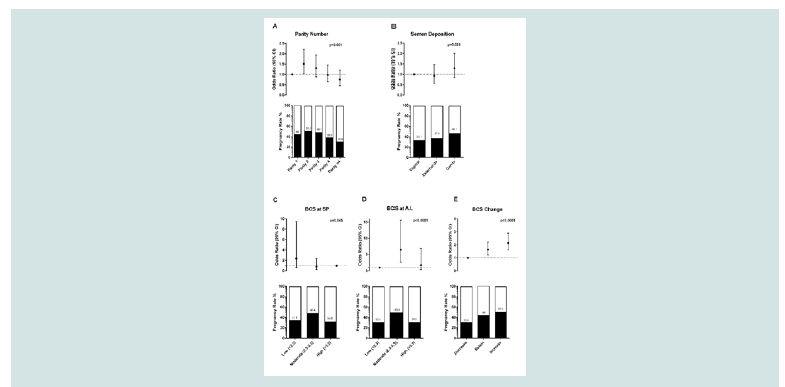
Figure 1: Pregnancy rates and adjusted odds ratio with 95% Confidence Interval of significant factors in final model: parity number (A), semen deposition site (B), BCS at SP (C), BCS at AI (D), BCS change (E).
Ewes received rations containing mainly alfalfa hay and alfalfa silage,
supplemented with 0.800-1.400 kg. Of a concentrate mix of maize,
soybean meal, barley and wheat middlings depending on their milk
production. Ewes had free access to water and wheat straw that was
offered ad-libitum in all cases.
Estrous synchronization: Each ewe was treated with a sponge containing 20 mg Fluogestone
Acetate (FGA) (CHRONOGEST CR®, MSD Animal Health), that was
placed intravaginally for 14 days. At the day of sponge withdrawal, 500
IU (Lacaune) or 400 IU (Chios) of equine Chorionic Gonadotropin
(eCG) (GONASER®, Hipra) were intramuscularly injected to the
ewes.
AI procedure: Semen was collected from 10 Lacaune rams that were located in the
same semen collection center (OVIS PC, Greece), using an artificial
vagina. Immediately after collection, motility and concentration of the
undiluted semen were assessed. Only ejaculates with concentration
greater than 3x109 spermatozoa/ml and mass motility greater than 4,
on the 0-5 scale described by Evans and Maxwell were used for the
study [16]. After this evaluation, semen was diluted to concentration
of 1,6x109 spermatozoa/ml using skimmed milk supplemented with
antibiotics gradually cooled at 15 °C and loaded into 0,25 ml mini
straws (400x106 spz/ dose) (IMV Technologies, France) [17]. The
straws were transported on farm for use inside thermos flasks with
acetic acid ampoules at 15 °C.
Cervical fixed-time AI was performed on each farm 53-57 hours
after the sponge removal. Ewes were immobilized by two assistants,
with hind legs lifted. In case of mucus presence inside vagina, the
animal was put again in horizontal position and the mucus was
removed using a speculum. AI was performed afterwards with the help of a speculum equipped with light source and an ovine AI gun
(IMV Technologies, France). All artificial inseminations were carried
out by the same technician within 8 hours after semen collection.
During the procedure, ewes were kept on a restrained area and
released to their boxes after insemination, or they were head-locked
in feed alley whenever this was applicable Ultrasonography was
performed 35-38 days after AI for Pregnancy Diagnosis (PD) using
5MHztransducerwith sector probe (ANIMAL PROFI, DRAMINSKI,
Poland).
Data collection: For all inseminated ewes, data concerning breed, parity and
number of previous synchronizations were recorded. Body Condition
Score (BCS) was assessed for each ewe at the time of sponge placement,
AI and PD. BCS was assessed by palpation in the lumbar region by the
same experienced evaluator. Scores assigned to the ewes were based
on the existing scale of Russel et al. ranging from 0 to 5, according
to which score (0) represents extremely emaciated animals, while
the highest score (5) represents obese ones [18]; 0.25 and 0.5 unit
increments were used. Changes of BCS between sponge placement
and pregnancy diagnosis (51-54 days) were evaluated to determine
whether the animals were in negative, zero or positive energy balance
during that period.
At the time of AI, the following data were collected for each
ewe: ram semen identification, semen collection-AI interval, sponge
removal-AI interval, time required per AI, order number of the
AI, presence of mucus in vagina, availability of headlocks for the
procedure, presence of rams near the females during synchronization
period and semen deposition site. The latter was distinguished in
3 classes depending on the deposition depth of the catheter and
retrograde flow of semen: vaginal deposition, external cervical os
deposition with partial semen backflow or deeper cervical placement without semen backflow.
Statistical analysis: Binary logistic regression analysis (SPSS ver. 25.0, IBM)
was used to determine the effect of risk factors to fertility of AI. Pregnancy diagnosis outcome at ultrasonography was used as the
dependant variable. Logistic regression was conducted according
to the method of Hosmer and Lemeshow with the following
five steps: 1) preliminary screening of all variables for univariate
associations, 2) construction of a full model using all the variables
found to be significant in the univariate analysis at P <0.25 level, 3)
stepwise removal of nonsignificant variables from the full model and
comparison of the reduced model with the previous model for model
fit and confounding, 4) evaluation of interactions among variables
and 5) assessment of model fit using Hosmer-Lemeshow statistics
until all the main effects or interactions were significant at P <0.05.
Chi square test was used to compare classes of the variables for better
interpretation of the results if required [19].
Results
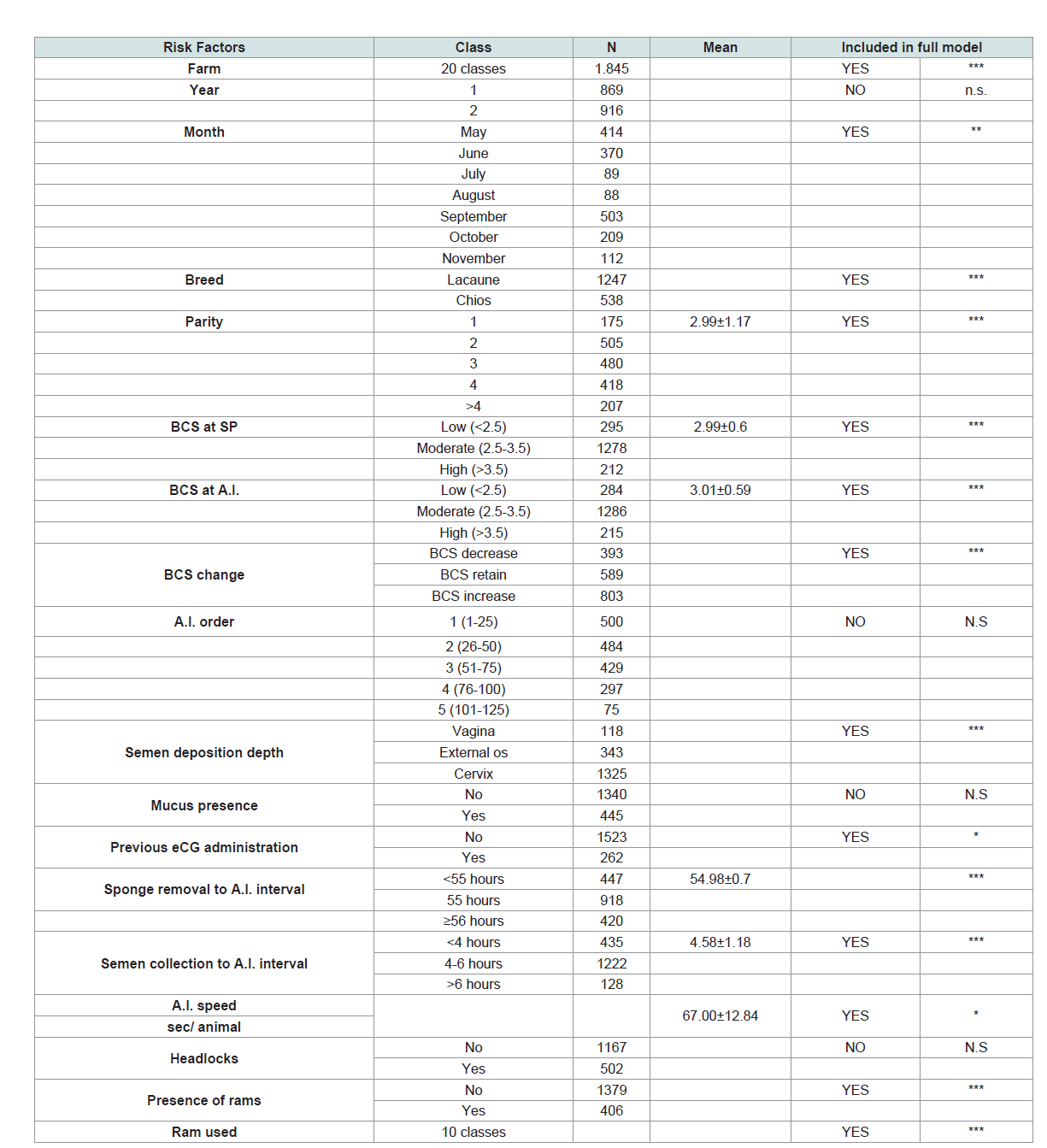
The description of examined factors included in univariate
analysis as well as in the multivariate model is presented in (Table 1).
Among these factors, ewe parity number, semen deposition site, BCS
at sponge placement and AI, BCS change from sponge placement to
pregnancy diagnosis, farm and ram used had a significant effect on
conception rates.
The overall pregnancy rate was 44%. Ewes that were on their
second lactation period showed to have higher fertility rates (50.3%)
compared to the rest, while odds were decreasing as the age increased
(Figure 1A). Pregnancy rates increased as the deposition depth
increased from vagina (33.1%) to external cervical os (37.4%) and to
inside cervix (46.7%) (Figure 1B). The depth of semen deposition was
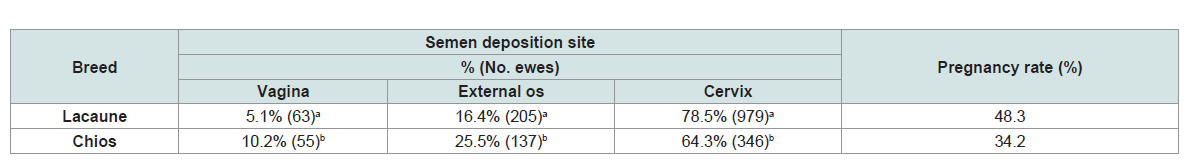
found to differ significant between the 2 breeds (Table 2). Although
breed was found to have a significant effect on fertility in the univariate
analysis, it was not included in the final regression model (P> 0.05).
Higher fertility rates were observed at animals with moderate
BCS (2.5-3.5) at sponge placement and at time of A.I (Figure 1Cand1D). Conception rate odds ratio of ewes increasing or retaining their
BCS were 2.14 and 1.63 times to ewes in negative energy balance
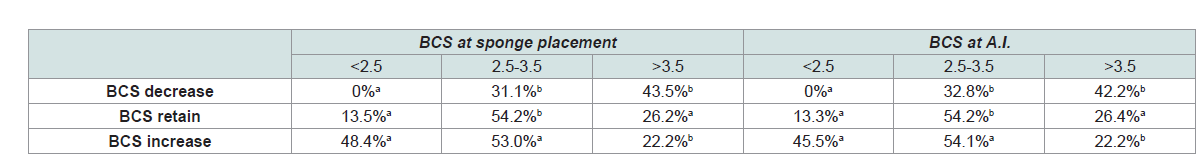
respectively (Figure 1E). However, at first step, interactions were
found between BCS at sponge placement and BCS change as well as
between BCS at A.I. and BCS change on their effect on conception
rates. Fertility was increased only in animals with low (45.5%) or
moderate (54.1%) BCS when they restored their body reserves. On
contrast, fertility rates were low when high BCS animals gained
more weight (22.2%) (Table 3). The effect of these interactions was
significant on the fertility of the animals in the univariate model
(p<0.25), but the interactions were (Hosmer and Lemeshow
test= 10.62; df= 8; P= 0.224; Classification accuracy= 64.9%) finally
excluded from the final model as non-significant. No other significant
interactions between variables were found. Regarding the rest factors in the final model, pregnancy rates
were found to differ significant between farms (p<0.001) and used
rams (p=0.007).
Discussion
The present study addressed the main factors affecting fertility
after AI using chilled semen in the most common intensively reared
dairy breeds in Greece, Chios and Lacaune. It is the first study to
examine the application of AI in Greece, including females of a
native and foreign breed, born in the area and well adapted in local
conditions and management systems.
Ewe parity and age have been widely assessed as factors
influencing fertility. Shackell et al. reported a 3% decrease of fertility
per year as average ewe age increased. Similar were the findings of
Arranz et al. [20,21], Fukui et al [22], and Palacin et al. [23], who
regarded the reduced fertility of older ewes as consequence of the
decreased quality of ovulated oocytes, despite the improved cervical
penetration that comes with increasing parity [24,25]. In our study,
pregnancy rates of primiparous ewes were lower compared to ewes
at second lactation period and declined rapidly beyond 4th parity.
According to Anel et al. [7], young ewes show reduced fertility due to
their inclusion with older ewes that usually lamb earlier. Additionally,
nutritional deficiencies are often observed in young ewes during the
mating period as a result of their larger requirements for growth. Our
findings are in contrast with other studies that found no effect of ewe
age on their fertility [26,27].
The deposition site of semen into female’s reproductive canal
is considered very important factor by many authors. In our study,
conception rates were improved when semen was deposited inside
cervix without semen backflow. The deeper deposition allows
more semen to reach the fertilization site and increases pregnancy
chances [28,29]. However, Paulenz et al. and Masoudi et al. found
no difference in pregnancy rates between vaginal and cervical AI
when fresh semen was used, stating that fresh semen contains enough
motile spermatozoa able to reach fertilization site. Conclusions of
Richardson et al. were the same using frozen-thawed semen, but with
lower pregnancy rates [30-32]. Attempts to deposit semen deeper invthe cervix with the use of specific catheters increase conception rates
of fresh semen especially when the catheter was introduced beyond
4 cm [33]. However, Kaabi indicated that pregnancy rates increased
with deposition of semen in cervix up to 3 cm, but deeper deposition
decreases fertility, maybe due to catheter and cervical manipulations that could activate pathways that interrupt pregnancy [34,35].
Reproductive performance is affected by the nutritional status of
animals. Our findings are in agreement with other studies according
to which animals in good condition respond well in onset of breeding
season and have higher ovulation and lambing rates [22,36]. At the
same time, ewes with low BCS activate their ovulation potential less
effectively [26,37]. However, there seems to be a plateau on the effect
of BCS on fertility as there is no benefit of increased BCS beyond a
point, and conception rates decline in animals with BCS> 3.5. Very
fat animals fail to show estrous at predicted time and exhibit lower
lambing rate potential [38-40]. Our study agrees with many authors
ending up that ewes should have a BCS of 2.5-3.5 at mating period
[22,41,42].
A positive effect of BCS increase on fertility of animals with low
and medium BCS was indicated in our study. Flushing during premating
period has positive effect on conception rates and can modify
the number and quality of embryos especially on thin ewes [43,44].
At the same time, low feed intake and BCS reduction results to lower
ovulation rate, decreased embryonic growth and increased fetal losses
[45-47]. Negative energy balance seems to have no significant effect
on animals with high BCS, indicating that the endocrine response
to undernutrition depends on body energy reserves [48]. However,
ewes with BCS> 3.5 demonstrated a decrease in pregnancy rate when
they were on positive energy balance. We suggest that weight gain
should be discouraged in fat sheep as can cause high ovulation rates
and increase embryonic losses [49].
Pregnancy rates of AI tended to be different among the farms
in our study. This could be attributed to variations on reproductive
planning and animal handling between farms [7], human-animal
interactions [23], or other stressful management conditions that
suppress fertility [50]. According to Santolaria et al. [10], fertility rates
can be affected positively by the improvement of handling conditions
in the farms.
Ram affected conception rates significantly, despite the strictly
selection of the ejaculates. Ram effect on AI success, evaluated
independently from the quality of the ejaculate, has been described
by many authors [23,51]. It remains unclear why same quality
semen produced from different rams and processed under the same
conditions, show significant variations in fertility rates. Differences
on seminal characteristics of the ejaculate that remain uncertain or
even genetic factors that influence ram’s fertility could have been
responsible for these variations.
Ewe breed has been described as a significant factor affecting AI
success by many authors [6,8,26]. Most studies are focused on the
differences among breeds on ovulation time, cervix morphology or
physicochemical properties of cervical mucus that can impair semen
transport [28,52,53]. In our study, breed did not affected pregnancy
rates significantly. However, we reported significant differences on
the deposition site of the semen between Chios and Lacaune breed.
This observation can be explained by the differences on cervix
morphology between the two breeds and could be responsible for
pregnancy rates differences observed.
The month of the insemination performance had no effect on
pregnancy rates, mainly due to the fact that this study was conducted from May to November, when both breeds exhibit strong cyclic
activity in this region. The lack of obvious heat stress effect during
July and August that could affect reproductive efficiency indicates
the need of evaluating more precisely the micro-environmental
conditions of each farm [54].
The presence of rams in the area near the synchronized ewes is a
factor that could affect pregnancy rates. Lucidi et al. found increased
pregnancy rate in Italian breed ewes that were exposed in ram after
sponge removal [55]. Exposure of females to rams before sponge
removal could reduce ecG administration-onset of estrous interval
and reduce the success of classic fixed time insemination protocols
[42,56,57]. That could be overcome by inseminating the ewes earlier.
In our study, rams presence near the ewes during synchronization
didn’t have any effect to fertility rates. This could be due to the fact
that rams didn’t join ewes inside their lot, but were kept in a nearby
area, limiting thus the male effect of a direct contact.
Conclusion
In conclusion, farm, ewe parity, semen deposition site, ram
and BCS of ewes, as well as changes of it during the period around
AI, were the main factors affecting fertility after AI on intensively
reared dairy sheep of Greece. Targeted selection of ewes, evaluation
of farm’s management practices and nutrition program, as well as
timely identification of the most fertile rams could lead to better and
more consistent results, contributing to the spread of AI application
in greek intensive flocks in larger scale.
References
Acknowledgment
This research is carried out / funded in the context of the
project “Recording and evaluation of factors affecting artificial
insemination success using fresh ram semen in Greek breeding
conditions” (MIS 5007366) under the call for proposals
“Supporting researchers with emphasis on new researchers”
(EDULLL 34). The project is co-financed by Greece and the
European Union (European Social Fund-ESF) by the Operational
Programme Human Resources Development, Education and
Lifelong Learning 2014-2020.
Citation
Priskas S, Termatzidou SA, Gargani S, Arsenos G. Evaluation of Factors Affecting Pregnancy Rate after Cervical Insemination of Dairy Ewes in
Greece. J Veter Sci Med. 2019;7(2): 7.