Journal of Veterinary Science & Medicine
Download PDF
Review Article
Alighting Dipterous Insects on Cattle are Associated to Contaminative Transmission of Foot-and-Mouth Disease During Epidemics in Ngaoundere- Cameroon
Sevidzem Silas Lendzele1,2*, Jacques François Mavoungou2,3, Zinga-Koumba Roland Christophe2,3, M’batchi Betrand4
1Ecole Doctorale des Grandes Ecoles (EDGE) de Libreville,
Gabon.
2Laboratoire d’Ecologie Vectorielle (LEV-IRET), BP: 13354,
Libreville, Gabon.
3Institut de Recherche en Ecologie Tropicale (IRET-CENAREST),
BP:13354, Libreville, Gabon.
4Laboratoire de Physiologie végétale et Phyto-alicaments; Unité de recherche Agrobiologie, Université des Sciences et Techniques de
Masuku (USTM); Franceville, Gabon.
*Address for Correspondence: Sevidzem Silas Lendzele, Laboratoire d’Ecologie Vectorielle (LEV-IRET), BP: 13354, Libreville, Gabon; E-mail: sevidzem.lendze@gmail.com
Submission: 16-August-2019;
Accepted: 21-September-2019;
Published: 23-September-2019
Copyright: © 2019 Lendzele SS, et al. This is an open access article distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Abstract
This study was designed to identify the landing preference sites of
common hematophagous symbovine Dipterans and relate it to the FMD
shedding sites. Three sets of zebu Goudali (from the DFG-COBE project
herd) of different colors (black, brown and white) of same ages were
restrained to sticks. Observations were made in October/November 2016
(seven days consecutively) and January 2017 (7days consecutively).
Data from literature on FMDV infectious doses (TCID50/ml) shedding
areas in clinical cases was used to map such sites on cattle and associate
it to the landing predilection sites of hematophagous dipterous insects.
The total number of observed biting insects on cattle was 26779 and
the following fly-groups were identified in order of magnitude: Stomoxys
(17453), culicids (8925), Simulium (293), Chrysops (74) and Tabanus (34).
Chrysops preferred biting front legs. Culicids preferred biting around legs
and neck. Tabanus preferred biting around head and legs. Stomoxys
preferred biting around neck and legs. The neck and legs were body
parts with the highest insect-vector frequency. The alighting predilection
of each insect-group differed statistically (P<0.05). From the association
test, an important number of each insect group was associated to at
least one of the FMD contamination spots on cattle, but Stomoxys and
culicids had a higher propensity of being contaminated as compared
to others based on their high landing numbers on the exposed animals.
Keywords
Haematophagous flies; Foot-and-Mouth Disease Virus; Cattle
Introduction
Foot-and-Mouth Disease is an Apthovirus of the family
Picornaviridae and is a highly contagious virus disease of even-toed
domestic and wild ungulates. It is caused by seven serotypes notably
O, A, C, Asia 1, SAT 1, SAT 2 and SAT 3, Bertram et al. identified
sequences of three serotypes (topotype O Africa/lineage East Africa,
A/Africa and SAT2 topotype and sub-lineage Lib-12) from the
Oropharyngeal Fluids (OPFs) and epithelia tissues of clinical and
subclinical cattle from Ngaoundere during the 2015 FMD epidemic.
Transmission pathways include contact with infected animals,
fomites, soil, air and animal secretions, but the role of invertebrates
in the spread of FMD is not clearly defined. USDA: APHIS: VS
categorized invertebrates especially biting insects as high hazards
in the spread of FMDV. This can be confirmed by the reports of
Carn, Ferris et al. and Hyslop who reported that viruses of the family
Picornaviridae as well as vesicle-forming viruses (like the vesicular
stomatitis virus-VSV and FMD) can be mechanically transmitted
by tabanids, Stomoxys and mosquitoes. FMD is an economically
important disease because it causes high morbidity and mortality rate
in calves but low in adults as well as precludes international trade
between endemic and non-endemic countries [1-7].
Based on the abundant nature of tabanids and muscids especially
Stomoxys in the absence of tsetse flies, it has been reported that
such biting flies are responsible for the spread of dangerous diseases
such as FMD in cattle herds in Ngaoundere. The recovery of the
FMDV RNA from S. n. niger body parts around the cattle market
in Ngaoundere raised an alarm on the implication of biting flies in
the spread of dangerous pathogens. The experiment of Arzt et al.
revealed the transmission of FMDV from persistently infected cattle
to naïve cattle recipients via mechanical transfer of unprocessed
Oropharyngeal fluids (OPFs) and Vesicular Epithelial Tissues
(VETs). The occurrence of high anti-FMD antibodies in brown
cattle as compared to other color coat of cattle has been reported
by Dickmu et al, but there is no link between FMD infection and
color of host. However, high cases in brown animals can be related
to the contaminative transmission caused by insect vectors which
are mostly attracted to color of host and preferably brown and black
colors. Moreover, if cattle were to be used as live attractive targets
or traps to reduce biting flies and other blood-feeding arthropods of
cattle, there is need to know the landing dynamics of these fly-groups
on live cattle and associate it to the risk of picking infective agents like
FMDV as baseline information for their control in pasture areas. The
present objective was to associate the landing dynamics data from live
cattle exposed to blood sucking insects and associate it with an FMD
risk cattle map to show the implication of the different fly-groups in
the contaminative transmission of the disease in Ngaoundere [8-13].
Materials and Methods
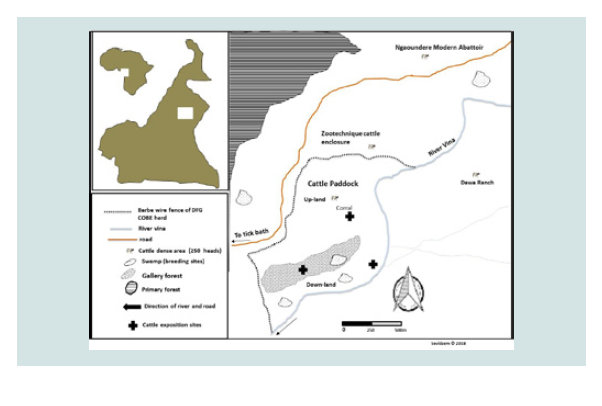
Description of the study area: The cattle paddock where the experiment was carried out falls
between Latitude 7° 11’N and Longitude 13° 34’E. It was elevated at
about 1000m a.s.l. The site was a cattle breeding zone of the Adamawa
plateau in Cameroon. The dominant cattle breed of this area was Goudali, but others like White Fulani, Red Fulani, Bokolodji,
Charlorais and their cross breeds (metis) prevailed in low numbers
in some herds. Cattle breeds of this region had different color coats
ranging from brown, white, black and a mixture of colors (red+brown,
brown+white, black+white etc.). Greater than 90% of herds in this
region are sedentary. The climate was the Soudan Guinean type with
landscape dominated by gallery forest and open grass savanna. The
main water body of this area consisted of river Vina du Sud (Figure 1).
Field exposure experiment to observe boophilic flies: Three animals from the DFG-COBE cattle herd were used for
the experiment, i.e. animal type 1 (red color), animal type 2 (white
color) and animal type 3 (black color) with minimum 80% color
coverage. The age of the animals was between 2 and 6 years. Animals
were restrained on fixed wood poles and kept at equi-distances of
5m. Cattle exposition was carried out in the potential breeding sites
(i.e. in the marshy low land beside river Vina du Sud, in the gallery
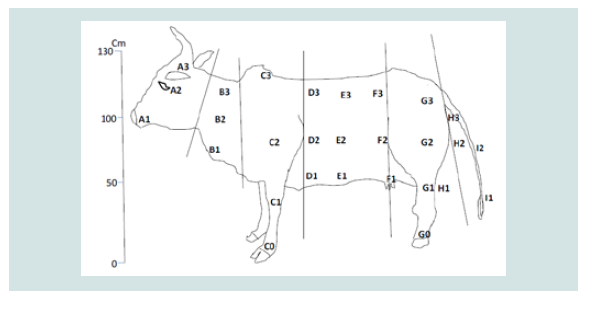
forest and around the cattle overnight park) of flies. The animal bodyparts
destined to indicate the fly predilections sites were mapped on a
bovine model. Three well trained observers (to distinguish the flies up
to the genus level) were close as 50 cm to the animal to identify and
count the flies per lateral side of each animal. Observation was carried
out following that of Hansen [13,14] (Figure 2).
Fly identification: Idenfication was carried-out on the spot up to genus level by welltrained
observers by strictly applying the criteria found in already
published taxonomic keys. For tabanids, the key of Odroyd was
used. Stomoxys were identified using the identification key of Zumpt.
Anophelinae were identified using the key of Gilles and De Meillon
and that of Jupp was used for Culicinae identification. For Simulium,
the key of Freeman and De Meillon was used [16-20].
Data on FMDV excretion or secretion dose: Data from 32 published scientific articles conceringing FMDV
infection experiments was exploited by consulting the published
document of Bravo de Rueda. Data on FMDV in secretions and
excretions were collected from 32 scientific articles published
between 1965 and 2007 found in internal databases and through the electronic (external) databases Scopus and PubMed in 2010, all
reporting experimental trials involving FMDV infection. The quantify
of the FMDV excreted or secreted was maintained in Tissue Culture
Infectious Dose 50 per milliliter (TCID50/ml) unit. The infectious dose
ranged from 0.95 to 10.15 TCID50/ml was considered. The data was
from cattle, sheep and pigs [21].
Data analysis: Data analysis was carried out using the R-software (R version
3.4.0). The Principal Component Analysis (PCA) test was used to
associate the biting sites of various fly-groups to the FMD shedding
sites on cattle. The Kruskal Wallis non-parametric test was used
to compare the number of alighting biting insects with respect to
predilection sites. The significant level of all tests was kept at p<0.05.
Ethical Statement: Animal use protocols were reviewed and approved by the Ohio
State University Institutional Animal Care and Use Committee
(Protocol Number: 2012A00000154). Restrained cattle were supplied
with water and fresh grass ad libitum. Animals were changed and
a fresh set recruited to avoid stressing the animals. Experimental
herders were present to carefully restrain the animals. Written
authorization was received from the project Director of the DFGCOBE
project to use the animals.
Results
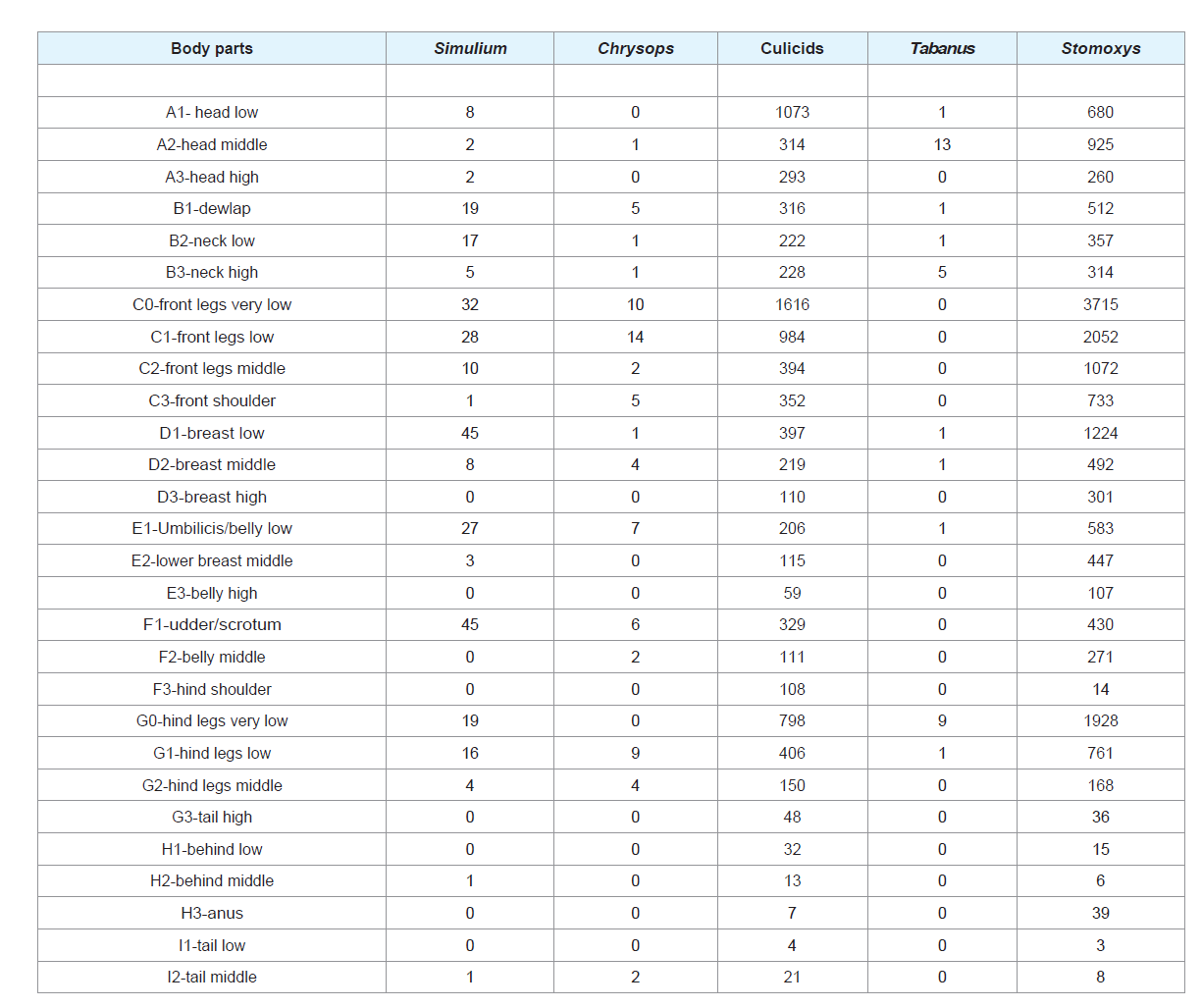
The total number of biting insect vectors observed on cattle
was 26779. The following taxonomic taxa were identified: Stomoxys
(17453), culicids (8925), Simulium (293), Chrysops (74) and Tabanus (34). The observed insect vectors on the exposed experimental animals
differed with biting insect-groups. The predilection sites of Simulium
was around the udder/scrotum (F1) and there was a statistically
significant difference (P<0.05) in their alighting predilection sites
on cattle. The predilection sites of Chrysops was the front legs (C0
and C1) and there was a statistically significant difference (P<0.05)
in their alighting preference on exposed cattle. Culicids were
most frequent around legs and neck (C0 and G0), but there was a
statistically significant difference (P<0.05) in their alighting sites on
cattle. Tabanus were most frequent on the head and legs (A2 and G0)
with a statistically significant difference (P<0.05) in their alighting
sites. Stomoxys were most frequent around the neck and legs (C0 and G0) with a statistically significant difference (P<0.05) in their landing
sites. It occurred that most biting insect-groups had preference for
the legs and neck region and there was an overlap of some biting
insect groups for some body parts like legs (Table 1).
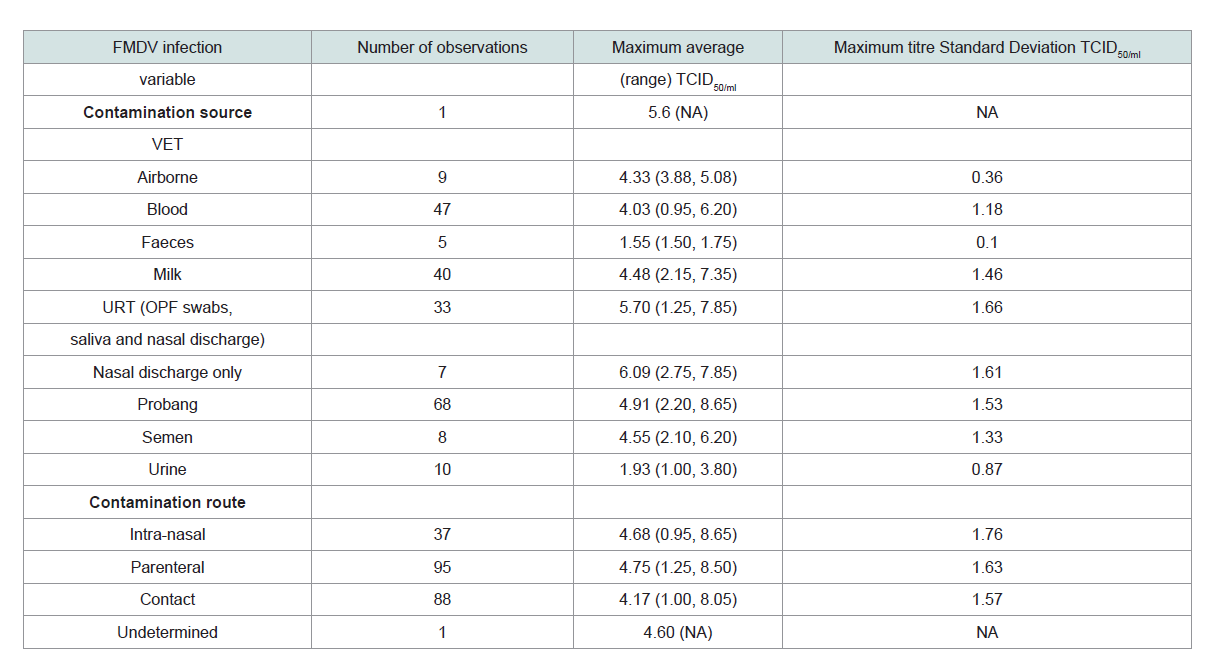
The FMD discharge doses (TCID50/ml) was gotten from already
published experimental works. According to Bravo de Rueda,
excretion of FMD occurred in unequal concentration in excretions
from the different body parts of domestic animals (cattle, sheep
and pigs). For the Upper Respiratory Tract (URT) consisting of
Oropharyngeal Fluid (OPF) swabs, salivary and nasal discharges
had mean infectious dose of 5.70±1.66, Vesicular Epithelia Tissues
(VETs) around the mouth, tongue, udder, scrotum and interdigital
spaces had mean infectious dose of 5.6, milk with infectious
dose of 4.48±1.46, semen with infectious dose of 4.55±1.33, urine
with infectious dose of 1.93±0.87 and faeces with infectious dose of
1.55±0.10 [21,22] (Table 2).
Total refers to all the maximum titres observations that were encountered. VET, vesicular epithelia tissue NA (not available), *TCID50 per animal per day for airborne
excretion; dose of infection and days post infection were divided as above and below the median of the maximum titer calculated using the maximum titres when
either the dose of infection or the days post infection was available [21, 22].
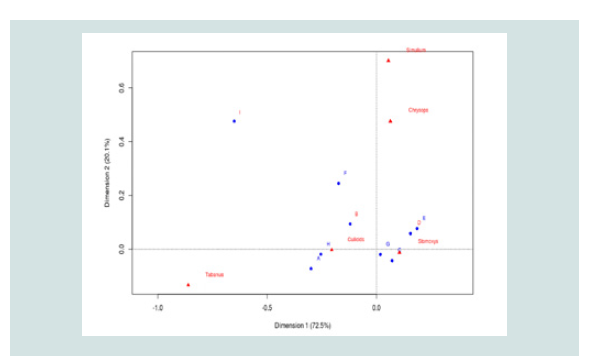
Association of predilection sites of haematophagous flies and FMD discharge spots on cattle: The multiple analyses of variance [Principal Component
Analysis (PCA)], where body parts constituted the rows and species
constituted the columns was used to associate alighting predilection
sites of biting insect vectors with sites of contamination with FMD.
The letters in red stood for low FMDV shedding sites while those
in blue stood for high FMDV shedding sites. From the association
test, all the biting insect vectors had a high probability of landing on
FMDV high risk spots [A1, C0, E1, F1, G0 and H3] due to their high
alighting propensity for such zones (Figure 3).
Figure 3: Analysis of correspondence showing the association of FMD risk
grading and fly-groups preferred body-parts of cattle. *Letters in blue signify
body part regions with high FMDV excretion potential while those in red
signifies body regions with low FMDV excretion. Letters A to I represent biting
preference sites of the various fly-groups as annotated on the cattle model
where: A-head regions (high FMD risk region), B-neck region (low FMD risk),
Co/Go-lower-leg regions (high risk of FMD), D-the trunk region is a low FMD
excretion/secretion zone, EF-belly region (high risk only around E1 and F1,
around the udder (female) and testes (bull)), GH-back regions (highly close
to urine or faecal discharge hence high risk of contamination with FMDV) and
I-tail region is a low FMDV risk region.
The sketch of FMDV excretion/secretion spots on cattle using
the published data of Bravo de Rueda can be seen in figure 4. The
mouth and nasal pathways are high sources of FMDV contamination,
followed by VET around the mouth, legs and testicular/mammary
region, milk, semen, urine, faeces and urine. Such spots are FMDV
contamination risk areas for alighting boophilic hematophagous
insects because of the infectious dose (1.55 to 5.60 TCID50/ml)
discharged from those sites [21] (Figure 4).
Discussion
The total number of alighting biting insects on cattle in the present
study was alarming and was dominated by culicids and Stomoxys. Flygroups that were likely to be observed or caught on cattle included
surface feeders, blood-sucking and myiasis causing flies. The most
dominant group observed on cattle constituted of muscids. Such an
observation was like that of Lloyd and Dipeolu. The abundant nature
of Stomoxys in the present study was in line with the report of Mihok
and Clausen [23,24]. Stomoxys have been reported to adapt in several environments [17,25]. The second most dominant group constituted of culicids and was like the observation made by Muenworn et al. [26] who showed that culicids preferred biting cattle than humans.
However, the biting predilection for cattle body parts by the different
fly-groups was different and most of the times overlapped. This biting
site-overlap resulted in scramble-feeding which was a possible risk
factor for their contamination when feeding on open sores or leisions
in the case of vesicle forming diseases like Vesicular Stomatitis Virus
(VSV) and FMDV. Biting preferences recorded in the present study
revealed alighting preference discrepancies by the biting insect
groups identified where Stomoxys prefered the legs and belly region,
Anopheles/Culex preferred legs and head region, Simulium preferred belly region, Chrysops preferred legs and Tabanus preferred legs and head regions. An overlap in the different fly-groups predilection sites in our study has been observed for some groups like muscids and tabanids, scrambling for biting sites around the lower limb [24,27]. The probable reason for the choice of legs by most biting insects was that the skin there was thinner and blood capillaries were closer to the surface of the skin. Those that preferred the head region like culicids and Tabanus might be because they were orientated to CO2 emissions from this part of the body [28,29].
The implication of color coat in the prevalence of FMD in
Cameroon by Dickmu et al. showed that brown cattle were the most
infected and this was in line with the present finding that brown cattle
were preferably attacked by three out of four fly-groups of biting
flies identified. Among the three fly-groups associated with brown
color coat was Stomoxys where S. n. niger was recently shown to be contaminated with infective doses of the foot and mouth disease virus RNA during the 2016 epidemic in the environs of the Ngaoundere cattle market. Arzt et al [7-9]. showed the mechanical transmission of FMD from persistently infected carrier cattle to naïve counterparts via the transfer of oropharyngeal fluid. Based on the association
of biting preference and FMD risk-graded spots, Stomoxys were
strongly-positively correlated with legs and belly areas (sub parts
C, D, E and G), Tabanus (head-sub part A), culicids (hind legs-sub
part H), Simulium (belly region-F) and Chrysops (head-sub parts DE). From the association of cattle fly-frequencies/bionomics with
respect to different annotated parts and the FMD risk graded maps,
it occurred that all the biting flies had a high probability of biting or
landing on FMDV high risk spots due to their high biting propensity
for FMD risk zones on cattle. This present result is in line with the
report of Carn [5]; Ferris et al. [6]; Hyslop [7] who reported that
tabanids, Stomoxys and mosquitoes can transmit viruses of the family Picornaviridae through horizontal transmission. However, since all
the groups showed equal chances of being contaminated, Stomoxys
and culicids will have a higher probability of being contaminated as
compared to others based on their high alighting densities on the
exposed animals.
Conclusion
The total number of observed biting insect vectors on cattle
was 26779 and the following insect-groups were identified in order
of magnitude: Stomoxys (17453), culicids (8925), Simulium (293), Chrysops (74) and Tabanus (34). Chrysops preferred front legs (C0 and C1). Culicids preferred biting around legs and neck (C0 and G0). Tabanus preferred head and legs (A2 and G0) and Stomoxys preferred neck and legs (C0 and G0). From the association test, all the biting insect vectors had a high probability of biting or landing on FMDV high risk spots [A1, C0, E1, F1, G0 and H3] due to their high biting propensity for such zones.
References
Citation
Lendzele SS, Mavoungou JF, Zinga-Koumba RC, M’batchi Betrand. Alighting Dipterous Insects on Cattle are Associated to Contaminative
Transmission of Foot-and-Mouth Disease During Epidemics in Ngaoundere-Cameroon. J Veter Sci Med. 2019;7(1): 4.