Journal of Toxins
Download PDF
Review Article
*Address for Correspondence: Jane C Quinn, Graham Centre Plant and Animal Toxicology Group, School of Animal and Veterinary Science, Charles Sturt University, Boorooma Street, Locked Bag 588, Wagga NSW 2678, Tel: +61-2-6933-4208; Fax: +61-2-6933-1991; E-mail: jquinn@csu.edu.au
Citation: Alden R, Hackney B, Weston LA, Quinn JC. Phalaris Toxicoses in Australian Livestock Production Systems: Prevalence, Aetiology and Toxicology. J Toxins. 2014;1(1): 7.
Copyright © 2014 Quinn et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use,distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Toxins | ISSN: 2328-1723 | Volume: 1, Issue: 1
Submission: 05 August 2014 | Accepted: 03 September 2014 | Published: 08 September 2014
Reviewed & Approved by: Dr. Guangming Xiong, Head of the Pharmacology and Toxicology, University Kiel, Germany.
P. aquatica actively grows between autumn and spring and is dormant during the summer months [1]. It has high biomass production, is tolerant of heavy grazing and can persist in Australian conditions where water logging can be a significant issue [4]. The deep-rooted growth habit of phalaris is associated with its significant drought tolerance [6]. The success of phalaris as a pasture species in Australia’s challenging landscape has also resulted in its adoption in New Zealand, the USA, Canada, South Africa, Northern Africa, and South America [1]. In addition to P. aquatica, there are a number of other species of the genus Phalaris which are used in grazing systems worldwide including P. angusta [7], P. arundinacea [8], and P. caroliniana [9]. All of these species are found in Australia but even after its first commercialisation 100 years ago, P. aquatica is still the most prominent pasture species grown in Australian production systems [3]. Currently, there are estimated to be 2.5 million hectares of P. aquatica sown in Australia with most of that production being located in higher rainfall zones within New South Wales (NSW) [4] as well as South Australia (SA), Western Australia (WA) and Victoria (VIC) [5]. Phalaris aquatica is an autumn, winter and spring growing perennial which adapts well to dryland or irrigated pastures being tolerant of both salinity and water-logging [10]. P. aquatica performs best in highly fertile, deep soils but shows good persistence in a wide range of soil types. It requires more than 500mm mean annual rainfall to persist [11], making it a common choice of pasture species in the medium to high rainfall zones in Australia. Therefore, phalaris has proven particularly valuable in Australian pasture systems as it provides high quality winter forage, is highly tolerant of drought, shows resistance to insects, can survive flooding, and is persistent enough to be used to control invasive weeds.
Phalaris Toxicoses in Australian Livestock Production Systems: Prevalence, Aetiology and Toxicology
Rowan Alden1, Belinda Hackney1, Leslie A Weston3 and Jane C Quinn2*
- 1 School of Agricultural and Wine Sciences, Graham Centre for Agricultural Innovation, Charles Sturt University, Australia
- 2 School of Animal and Veterinary Sciences, Graham Centre for Agricultural Innovation, Charles Sturt University, Australia
- 3 Graham Centre for Agricultural Innovation, Charles Sturt University, Australia.
*Address for Correspondence: Jane C Quinn, Graham Centre Plant and Animal Toxicology Group, School of Animal and Veterinary Science, Charles Sturt University, Boorooma Street, Locked Bag 588, Wagga NSW 2678, Tel: +61-2-6933-4208; Fax: +61-2-6933-1991; E-mail: jquinn@csu.edu.au
Citation: Alden R, Hackney B, Weston LA, Quinn JC. Phalaris Toxicoses in Australian Livestock Production Systems: Prevalence, Aetiology and Toxicology. J Toxins. 2014;1(1): 7.
Copyright © 2014 Quinn et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use,distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Toxins | ISSN: 2328-1723 | Volume: 1, Issue: 1
Submission: 05 August 2014 | Accepted: 03 September 2014 | Published: 08 September 2014
Reviewed & Approved by: Dr. Guangming Xiong, Head of the Pharmacology and Toxicology, University Kiel, Germany.
Abstract
The perennial grass Phalaris is a valuable pasture species which features predominantly in Australian and North American grazing systems. Its usefulness, however, has been tempered by the presentation of sporadic outbreaks of severe toxicoses associated with its ingestion by grazing livestock. Phalaris toxicoses present as one of two clinical entities: phalaris ‘staggers’ or ‘sudden death’. Although alkaloid toxins have been implicated in both syndromes their identification and mechanism of action are yet to be fully defined, and the specific nature of phalaris toxicity is currently poorly understood. Mitigation strategies currently centre on changed management practices, such as sowing low alkaloid cultivars and supplementation with cobalt. This review examines the prevalence, aetiology and pathogenesis of phalaris ‘staggers’ and ‘sudden death’ syndromes as well as current knowledge of environmental and agronomic interactions associated with the production of nitrogenous compounds and bioactive secondary metabolites in the plant.Introduction
Phalaris aquatica L., previously also known as Phalaris tuberosa L. [1], originates from the Mediterranean and Middle East [2]. It was first introduced in Australia soon after European colonisation [1] as reported in 1877 and has since proven to be an important pasture species for the Australian climate [3]. The introduction that gave rise to the first Australian cultivar, named ‘Australian’, was developed at Toowoomba in 1883, and was released for commercialisation in 1906 [4]. ‘Australian’ spread throughout the southern region of the continent and for 60 years was the only commercially available cultivar [2]. The success of ‘Australian’ in the challenging environmental and agronomic climate in Australia prompted the development of other commercial cultivars such as ‘Seedmaster’, ‘Sirocco’ and ‘El Golea’, which were developed for greater seed retention or higher winter growth and summer dormancy. These cultivars were released in the 1960s and 1970s (Figure 1) [5].Figure 1: Phalaris aquatica cultivars in current using in Australia pasture systems. A) Phalaris aquatica cv. ‘Australian’, mature plants at full height showing flowering heads. B) P. aquatica cv. ‘Holdfast’ and C) P. aquatica cv. ’Sirocco’ as grazing pastures. Images reproduced by permission of R. Culvenor (CSIRO).
A limiting factor to the widespread use of phalaris in grazing systems worldwide is the sporadic, but significant, incidence of toxicoses in livestock grazing upon this genus [12-16]. Phalaris toxicoses typically presents as one of two clinical entities: phalaris ‘staggers’ [16,17] and also as the two potentially related forms of phalaris ‘sudden death’ syndrome [16,18]. In Australia, phalaris toxicity is estimated to cost producers approximately $1.7 million annually with the average farmer experiencing an outbreak between one in four and one in twenty years [19]. Significant stock losses, particularly in sheep, have been reported [20]. However, the full economic costs to the livestock industry are unclear. In their report [19], Sackett and colleagues do not address each of the syndromes separately, nor do they take into account losses in cattle or other species, nor estimate the cost of preventative cobalt supplementation and other pasture mitigation strategies. For these reasons, it is likely that this figure significantly underestimates the cost of these syndromes to Australian farming systems.
Prevalence of phalaris toxicoses in grazing livestock
McDonald first described toxicity in grazing livestock resulting from ingestion of phalaris in 1942 [21]. Subsequently, outbreaks were reported in New Zealand [22], America [8], and Argentina [23] with most reported in livestock grazing P. aquatica. However, toxicity has also been documented in stock grazing P. angusta [7], P. arundinacea [8], and P. caroliniana [9]. In Australia, a higher prevalence of phalaris toxicity has been reported in NSW than other states [24], possibly due to the more frequent use of this pasture species in this state than in others across Australia [25].
Ruminants are most commonly reported to be affected by phalaris toxicoses. Outbreaks in both cattle and sheep have been documented [9,16,21, 24] with sheep appearing to be more susceptible to phalaris toxicoses than cattle [26]. Goats can also be affected by phalaris toxicity although they appear to be less vulnerable than their ovine counterparts [27]. Horses have also been reported to be affected [18], but appear to be impacted more by ingestion of P. coerulescens and P. paradoxa than P. aquatica [28] which may explain why horses have been known to graze the same pastures as affected sheep without presenting any clinical signs of toxicity [21]. However, to date, no completely ‘safe’ or non-toxic phalaris cultivar has been identified.
Sheep of any age, breed or sex can develop clinical signs of phalaris toxicosis [21,27], although some breed specific susceptibility has been reported with Romney Marsh weaners appearing to be more susceptible than Merinos of the same age [29]. Gallagher and colleagues (1966) observed that incidence of phalaris ‘staggers’ in sheep decreased with increasing stocking rates [24]; conversely, Bourke et al. found that the incidence of polioencephalomalacia-like (PE-like) sudden death increased with higher stocking densities so effect of stock ratios is unclear on the aetiology of the disorder. Neither age nor cultivar of phalaris pasture has been shown to mitigate phalaris ‘staggers’ as both young and well-established pastures have been associated with neurotoxicity [30].
Grazing management has been suggested as a strategy to mitigate toxic outbreaks. Rapid digestion of phalaris cultivars by hungry sheep without time to adjust to high alkaloid and / or nitrogen (N) levels in the plant has been suggested as a factor associated with higher incidence of P. aquatica polioencephaliomalacia-like (PE-like) sudden death syndrome; a rapid, lethal toxicosis resulting from significant neurological damage [15,18,31,32]. Both supplemental feeding of sheep prior to introduction to high risk pastures, or slow introduction of a mob to a paddock, have been suggested as methods of reducing toxic outbreaks [27]. Joint sowing of other pasture species with phalaris varieties can also potentially reduce the risk of phalaris toxicity [26]; however, mixed pastures containing ryegrass, clovers, native grasses and lucerne have all been reported to be associated with development of phalaris toxicoses in livestock [30,33]. Testing paddocks with a small group of less valuable stock and closely monitoring their health prior to introduction of the mob can potentially reduce losses but is a time consuming and potentially costly exercise. As such, no effective mitigation strategy has yet been identified to completely eradicate risks associated with phalaris toxicoses when grazing livestock on this pasture species.
Clinical presentation of phalaris toxicoses -phalaris ‘staggers’
Phalaris toxicoses presents as two key clinical entities commonly known as phalaris ‘staggers’ and phalaris ‘sudden death’ [16]. Phalaris ‘sudden death’ usually occurs within 48 hours of entry to phalaris pastures and has two associated syndromes: one a cardiac syndrome and the other a neurological PE-like toxicosis [14,18,32,34]. Sheep within the same mob have been reported to present with both phalaris ‘staggers’ and ‘sudden death’ syndromes [24] but the reason why some animals present with one syndrome and others with another is currently unclear.
There are also two forms of phalaris ‘staggers’: acute and chronic. Acute cases appear to occur when sheep are actively grazing phalaris pastures and the onset is rapid and recovery can be complete whereas onset of chronic phalaris ‘staggers’ presents more slowly, as a chronic degenerative disorder, and cases are usually fatal [14,15,35]. Fatalities from acute staggers are rare [20,21,24] and the clinical signs can resolve after sheep are removed from the pasture, although this resolution can take many months [24,36]. Recovery from chronic staggers is unlikely due to the degenerative nature of the changes in the central nervous system finally resulting in weight loss and death [15,22,30,33].
Neurological signs associated with acute phalaris ‘staggers’ first appear between six hours to 21 days after stock are placed on phalaris pastures [21,22,24] with chronic changes requiring a minimum of three to four months to present clinically [35]. Acute signs of toxicity include hyper-excitability, muscle tremors, head nodding; an ill-coordinated ‘proppy’ gait, fore and hind limb paresis, convulsions and, in the most severe cases, death [14-16,22,33]. Other less commonly reported signs have included twitching ears, lethargy, recumbency, falling, heavy panting and loud heart sounds [21] possibly suggestive of commonality of clinical changes between the cardiac and ‘staggers’ presentations. The duration of, and combinations of, different clinical signs can be highly variable and in some cases can subside and then cattle, clinical signs of phalaris ‘staggers’ are not well reported as the incidence is relatively rare compared to that in sheep [26]; however, neurological changes also include ill-coordination, muscle tremors, excessive salivation, lethargy, failure to thrive, a reduced ability to swallow and falling [8,21,23]. There have been some attempts to administer pharmacological agents to control this syndrome and both ‘Myanesin’ and barbiturates have been shown to be an effective treatment for acute phalaris ‘staggers’ in livestock [30].
Pathological lesions associated with phalaris toxicoses
A variety of post-mortem changes have been reported to be associated with clinical cases of phalaris ‘staggers’ in livestock. These include pleural oedema, a green discolouration of the kidneys and peripheral and central neuropathy. Deposits of golden brown to green pigment have also been noted in the cell bodies of neurons of the central nervous system, in particular in the brain stem and cerebellum (Figure 2), as well as in motor neurons in the spinal cord [16,21,26,33]. These pigment deposits have been suggested to caused by accumulation of indoles, formed by oxidative deamination of alkaloid toxins, or direct pigment precipitation of 5-5-hydroxy- N,N-dimethyltryptamine (HDMT) in affected neurons [24]. Pigment deposition in neuronal cell bodies has been suggested to be indicative of a neuronal storage syndrome [16]. Few post-mortem reports have been published on the pathological changes observed cattle but Binder et al. describe necrotic neurons in the spinal cord with accumulation of intracytoplasmic granular pigment in many cases, with regions affected including the spinal cord, brain stem, pons, medualla and midbrain. A green-brown pigment accumulation was also noted in tubular epithelial cells of the kidney [16].Phalaris ‘sudden death’ syndrome: two clinical entities, one common outcome
As described above, phalaris ‘sudden death’ in sheep typically appears as one of two clinical entities: a cardiac form and a neurological form, PE-like sudden death syndrome [15,18,31]. The cardiac presentation of this disorder gives rise to multi-factorial cardio-respiratory cessation resulting in the sudden death of the animal, often within hours of introduction to phalaris pastures. Cardiac sudden death syndrome in sheep grazing phalaris has been suggested to be caused by acute hydrocyanic acid toxicity in combination with nitrate poisoning and other potentially toxic compounds [32], but definitive proof of the presence of hydrocyanic acid in plants and afflicted animals has not been obtained, likely due to the transient nature of this molecule. Interestingly, the geographical location of a pasture appears to influence the type of sudden death syndrome occurring with the neurological form being more common than the cardiac form in South-East Australia [31].
Sheep with the PE-like syndrome which do not die often exhibit blindness, wandering behaviour, head pressing, tremors, excess salivation and general depression, all suggestive of widespread neurological dysfunction [32]. PE–like sudden death has been reported 12 to 48 hours after hungry sheep are given access to the phalaris pasture [18] and has been suggested to be caused by peracute ammonia toxicity of unknown aetiology [31,37]. It has been suggested that thiamine or pyridoxine administration may protect sheep from PE-like sudden death although this anecdotal treatment remains to be validated [37]. Cattle have also been reported to die from PE-like death, with fatalities continuing for up to four days after being removed from the toxic pasture [31]. The symptoms of sudden death in cattle with PE-like sudden death syndrome have been described by Bourke et al. as similar to those in sheep and include wandering behaviour and excess salivation.
Plant secondary metabolites implicated in phalaris toxicoses in grazing livestock
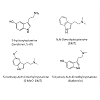
A number of plant compounds have been suggested to be causal in cases of phalaris toxicoses. Culvenor and colleagues first described investigations to identify the causal compounds in 1964 where they observed high levels of the psychoactive tryptamines N,N-dimethyltryptamine (DMT), 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) and the related metabolite 5-hydroxy-N,N-dimethyltryptamine (5-HDMT, also known as Bufotenin) in P. aquatica [38]. Tryptamine alkaloids are structurally similar to serotonin (Figure 3) and can affect both cardiac and neurological function [24]. DMT, 5-MeO-DMT and 5-HDMT were found to produce symptoms similar to acute phalaris ‘staggers’ when injected into sheep with 5-MeO-DMT producing the most severe clinical effects [39], suggesting a causal role for these compounds in the presentation of phalaris ‘staggers’ in livestock., Gramine (a tryptamine) and hordenine (a tyramine) and 5-MeO-DMT have also been implicated in cases of phalaris ‘sudden death’, although symptoms of ‘staggers’ could only be produced by experimental administration of 5-MeO-DMT [34]. The indole alkaloid N-methyltyramine (NMT) was also suggested to cause phalaris PE-like sudden death [40]; however, this could not be confirmed by clinical trials and the putative causal compound, if there is a single compound at all, remains elusive.
A causal link between the development of phalaris ‘staggers’, resulting from ingestion of high levels of 5-MeO-DMT, and the poten¬tial presence of other currently unidentified bioactive indole alkaloids which could affect accumulation of this and other neuroactive com¬pounds in the central nervous system of affected animals, remains to be fully determined . The compound 5-MeO-DMT and its metabolite bufotenine are potent non-selective serotonin agonists with activity both in the central nervous system and systemic tissues including the intestinal tract [41]. Administration of 5-MeO-DMT and other serotonin agonists can cause multiple physiological and behavioural effects [41]. The differential effects observed in livestock after ex¬perimental administration of NMT [40], and 5-MeO-DMT [39] may be due to the relative high affinity of 5-MeO-DMT to the serotonin 5HT-1A receptor in comparison to other members of this chemical family [41]. Inhibition of 5-MeO-DMT activity in the central nervous system is modulated by deamination of 5-MeO- DMT to bufotenin, a liver-based reaction, with bufotenin showing a preferential affinity for the 5-HT-2A receptor and rapid elimination from the systemic cir¬culation [41]. Exposure of animals to 5-MeO-DMT, in the presence of monoamine-oxidase inhibitors, such as the as indole alkaloid har¬maline or its derivatives which can be found in some plant and seeds [42-44], can result in fatal 5-MeO-DMT toxicity [45] not dissimilar to that observed in phalaris toxicoses. This combination of 5-MeO-DMT and monoamine oxidase inhibitors which extend 5-MeO-DMT activity in the central nervous system, have also been shown to have beneficial effects in combating addictive behaviours associated with serotonergic hyperstimulation in humans[46,47] suggesting that the balance between these compounds is important for maintenance of normal neurological function.
The foundation P. aquatica high alkaloid varieties, ‘Australia’ and ‘Seedmaster’, are now associated with more frequent occurrence of toxicity [48]. The identification of a critical role for alkaloid toxins in the clinical presentation of phalaris toxicoses has also resulted in considerable efforts to produce ‘safer’ commercially available cultivars with reduced alkaloid production. ‘Holdfast’, ‘Sirosa’, ‘Sirocco’ and ‘Sirolan’ are commercially available cultivars that have been bred for low alkaloid content [25] with ‘Holdfast’ and ‘Sirolan’ selected particularly for low DMT and related alkaloid concentrations. Although Holdfast also has low DMT, it does contain very high concentrations of tyramine- related alkaloids and was associated with a toxic outbreak in South Australia [49]. Despite their ‘low’ alkaloid contents, there have been cases of phalaris ‘staggers’ in sheep and cattle grazing ‘Sirocco’ and ‘Seedmaster’ varieties [49], and numerous cases of reported toxicity with ‘Sirolan’ and other cultivars in Western Australia [49] and other locations. This clearly suggests two possibilities: either these cultivars contain levels of alkaloids above the toxic threshold or other metabolites are also potentially responsible for these outbreaks. ‘Landmaster’ and ‘Atlas PG’ were bred for static levels of dimethyltryptamine, but reduced levels of tyramine-related alkaloids in comparison to ‘Holdfast’ [50], a ratio thought to be beneficial in reducing alkaloid-related deaths. The importance of this ratio is not currently known. The lack of successful production of non-toxic varieties of P. aquatica is likely due to the complex interactions governing alkaloid production in these graminaceous plants, given that a multitude of factors including drought or heat stress, soil composition, phenology and herbivory can all influence production [51,52]. Environmental factors are clearly associated with production of DMT and tyramine-related alkaloids in all phalaris cultivars [50], similar to results obtained when studying Echium plantagineum and production of lycopsamine alkaloids [52] although exact regulation of their production in phalaris is currently less well understood.
Cardiac and PE-like sudden death: a variation in presentation of one clinical syndrome?
We now understand that phalaris ‘sudden death’ in grazing livestock has been associated with all commercially available cultivars including the low alkaloid biotypes [4]. The chemical constituents resulting in ‘sudden death’ are still unidentified. Recent research in Australia has focused on PE –like sudden death as it is the more prevalent of the two forms of toxicity [18]. Bourke et al. found that deceased cattle affected with PE–like ‘sudden death’ exhibited hyperammonemia showing high ammonia concentrations in the aqueous humour of the eye post- mortem in comparison to normal reference level ranges. Similar occurrences have also been reported in sheep [31].
Although rare, hyperammonemia, or non-protein nitrogen poisoning, is reported in domestic livestock [53]. Clinical signs associated with hyperammonemia develop rapidly, often presenting with 30 minutes of ingestion, with progression to death within as little as four hours. This presentation shows marked similarities to phalaris cardiac ‘sudden death’ syndrome and related syndromes. The sequence of cellular events causing death in these cases is thought to be multi-factorial, potentially via ammonia perturbation of the lactic acid cycle [54], or inhibition of sodium-potassium ATPase pump recycling of extracellular potassium causing systematic metabolic acidosis [55] resulting in subsequent cardiac arrest [53]. Interestingly, altered lactic acid cycling can also result in altered neurological signs, coincident with changes observed in livestock with a PE-like syndrome, suggesting that these two syndromes may not be totally unrelated. It has been postulated that the alkaloid that causes PE-like sudden death may act as an inhibitor of the urea cycle, thereby slowing the rate of removal of ammonia from the bloodstream, resulting in hyperammonaemia [31].
The greatest period for risk of phalaris toxicoses occurs when phalaris plants are undergoing rapid vegetative growth [49], coincident with stages when nitrogen (N) levels are typically highest in the plant [25,31]. As inhibition of key enzymes required for the metabolic conversion of ammonia to less toxic forms of N in plants has been suggested to be a possible mechanism of the hyperammonaemia observed in clinical cases of phalaris toxicoses [31], ingestion of high levels of N in the diet would be contraindicated when grazing phalaris varieties. Cattle were observed to suffer from PE-like sudden death after grazing actively growing lucerne, a pasture species which naturally contains high levels of protein N [31]. This high level of nitrogen intake prior to exposed to potentially toxic phalaris pasture may have contributed to the deaths of these cattle, but further investigations are warranted to answer these questions.
A grazing animal’s susceptibility to hyperammonaemia can be potentially mitigated by slow introduction of the animal to a high non-protein N diet, (including actively growing, young phalaris plants), providing optimal balances of rumen and gut microflora to be established for conversion of excess N to digestible proteins. Rumen pH and the rumen microflora environment can be influenced by many factors including a low carbohydrate diet, stress, metabolic challenge due to disease, hepatic dysfunction, high ambient temperatures or dehydration; all of which can all affect the animal’s ability to effectively metabolise exogenous nitrogen sources. An accumulation of such factors might influence a predisposition to ammonia sensitivity, and therefore ultimately toxicity, in naive animals introduced to rapidly growing phalaris pastures.
Environmental factors influencing outbreaks of phalaris toxicoses in ruminants
Phalaris toxicoses has been reported in animals grazing phalaris pastures on soils with cobalt deficiency [18,30]. This has been particularly evident in South Australia [33]. Soil cobalt levels have been associated with outbreaks of phalaris ‘staggers’ [56]. However soil cobalt does not seem to correlate well with cobalt levels within the plant; therefore it has been suggested that sheep generally obtain their dietary cobalt by eating soil [56]. Clearly, there is not a simple correlation between cobalt deficient soils and phalaris toxicoses, as affected animals have also been observed on Australian soil types that are not deficient in this trace mineral, including Tablelands basalt, Cowra red loam and andesite soils [36] suggesting once again that other environmental factors are also at play. Equally, the mechanism of action of cobalt administration to livestock as a prophylactic treatment for phalaris toxicoses is also questionable despite being a treatment modality for many years [33,57], with cobalt bullets suggested as an effective prophylactic to prevent chronic phalaris ‘staggers’ in sheep [24,26]. Interestingly, administration of cobalt has been found to be less effective in mitigating for the acute form of phalaris toxicoses; in this case administration of cobalt had limited or no effect at all on the incidence of mortality resulting from phalaris sudden death syndrome [24,58].
A link between cobalt deficiency and alkaloid production in the plant has been hypothesized. Cobalt has been found to have a protective role against drought stress in higher plants [59] and it has been shown that alkaloid levels are highest in phalaris plants under the greatest moisture stress [18,60,61]. Therefore, soil cobalt deficiency could reduce the plant’s drought tolerance causing enhanced alkaloid toxin production due to stress, thereby increasing the likelihood of incidence of phalaris toxicity in deficient soils. Other environmental factors have also been reported to increase levels of alkaloids in plants; these include frost events [18], shading [24,60], spacing between plants or inter-plant competition, temperature and drought [50,52]. Sheep deaths associated with phalaris have also been suggested to be more prevalent early in the morning and when there is cloud cover or fog [24] although the direct correlation between these findings and toxicity remains unclear. High N content in soils, linked to widespread application of superphosphate fertilisers stimulating legume production and therefore nitrogen fixation prior to the 1970s and the associated growth of clover, may also have historically increased the concentrations of N-containing compounds within phalaris stands. Fertiliser use on pastures has declined since 1973 when the superphosphate bounty was removed [62]. As legumes such as subterranean clover (Trifolium subterraneum L.) have a higher phosphorus requirement for optimal production than grasses, and as such the loss of the superphosphate bounty resulted in a decline in legume production in grazing pastures and therefore availability of nitrogen to non-leguminous pasture species [63]. This change has resulted in pastures that have become more grass dominant across Australia, and therefore encouraged growth of species such as phalaris in gazing systems. Whether this long-term change in pasture composition as a result of lower superphosphate use, and the associated loss of legumes in Australian grazing systems has had any impact on prevalence of phalaris toxicoses remains unclear but is an interesting question.
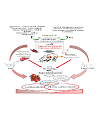
In addition to effects of soil mineral composition on plant metabolism, there is also evidence to suggest that autumn rains resulting in vigorous new growth of a phalaris dominant pasture can lead to an increased likelihood of phalaris toxicoses [21,22,24,33,49]. Culvenor et al. showed that the concentration of alkaloids within phalaris species were highest in autumn when new growth occurred following summer dormancy [64,65]. Further research investigating the impact of climate, rainfall, soil type and weather conditions on the physiological characteristics of the phalaris cultivars, and the associated incidence of outbreaks of phalaris toxicosis, is important in providing solutions for graziers attempting to graze livestock on phalaris species during periods of highest risk for livestock poisoning. A summary of the complex agronomic, phytochemical and physiological interactions associated with phalaris toxicoses is shown in (Figure 4).
Conclusion
Although incidence of phalaris toxicoses in Australia are consistently reported, the epidemiology of this disease remains unclear: Livestock grazing and toxicity surveys are critical to determine the incidence of these disorders which are undoubtedly multi-factorial in nature (Figure 4). Determination of the causal compound(s) associated with phalaris toxicoses, the mechanism by which hyperammonaemia is induced, and other factors affecting the metabolic breakdown of nitrogenous compounds, their catabolism in the plant and involvement in these complex clinical presentations are all areas in which further research is required. This information could enable the development of safe cultivars with associated metabolic profiles to assist in mitigation of toxic outbreaks. The mechanism by which cobalt and supplementation with other anecdotes prevent chronic phalaris ‘staggers’ remains unknown. However, knowledge of high risk soil types and increased awareness within the industry could reduce stock losses. The impact of environmental factors on toxicity requires additional investigation using controlled environment experimentation coupled to metabolic profiling for determination of bioactive inhibitors and discovery of the toxin(s) involved in these syndromes will also result in better industry awareness of risk. Optimal integration of pasture and animal management practices, including fertiliser application, grazing regimes, stocking rate and pasture composition, will also likely reduce the risk of phalaris toxicoses and possibly enhance the continued utilisation of this drought tolerant and versatile pasture grass globally as well as in Australian farming systems.Acknowledgements
The authors wish to thank R. Culvenor, C. Bouveret, T. Ross and M.A. Combs for helpful comments on this manuscript. RA is in receipt of a Graham Centre for Agricultural Innovation Internship Award. All authors are members of the Graham Centre for Agricultural Innovation; a collaboration between Charles Sturt University and the Department of Primary Industries, New South Wales, Australia and are funded from numerous sources to undertake research related to palant-related toxicities in livestock.References
- Oram RN, Ferreira V, Culvenor RA, Hopkins AA, Stewart A (2009) The first century of Phalaris aquatica L. cultivation and genetic improvement: a review. Crop and Pasture Science 60: 1-15.
- Culvenor RA (2009) Breeding and use of summer-dormant grasses in southern australia, with special reference to phalaris. Crop Science 49: 2335-2346.
- Jessop JP (2006) Grasses of South Australia: an illustrated guide to the native and naturalised species. Kent Town, South Australia: Wakefield Press.
- Smith K (2013) Developing a Phalaris pre-breeding plan: Phalaris 2020 vision - better cultivars faster. North Ryde, Sydney, Australia.
- Oram RN and Culvenor RA (1994) Phalaris Improvement in Australia. New Zealand Journal of Agricultural Research 37: 329-339.
- Seargent K, Watson D (2008) Growing Phalaris for production and persistence. 2008, Future Farm Industries CRC, Factsheet.
- Odriozola E, Campero C, Lopez T, Marin R, Casaro G, et al, (1991) Neuropathological effects and deaths of cattle and sheep in argentina from phalaris-angusta. Vet Hum Toxicol 33: 465-467.
- Binder EM, Blodgett DJ, Currin JF, Caudell D, Cherney JH, et al. (2010) Phalaris arundinacea (reed canarygrass) grass staggers in beef cattle. J Vet Diagn Invest 22: 802-805.
- Nicholson SS, Olcott BM, Usenik EA, Casey HW, Brown CC, et al. (1989) Delayed phalaris grass toxicosis in sheep and cattle. J Am Vet Med Assoc 195: 345-346.
- Nichols PGH, Rogers ME, Craig AD, Albertsen TO, Miller SM, et al. (2008) Production and persistence of temperate perennial grasses and legumes at five saline sites in southern Australia. Australian Journal of Experimental Agriculture 48: 536-552.
- Gallagher CH, Koch JH, Moore RM and Steel JD (1964) Toxicity of Phalaris tuberosa for sheep. Nature 204: 542-545.
- Camara F, Amaro MA (2003) Nutritional aspect of zinc availability. Int J Food Sci Nutr 54: 143-151.
- Bourke CA (1994) The Clinico-toxicological differentiation of phalaris spp toxicity syndromes in ruminants. Plant-Association Toxins: 523-528.
- Bourke CA, Carrigan MJ, Dixon RJ (1990) The pathogenesis of the nervous syndrome of Phalaris aquatica toxicity in sheep. Aust Vet J 67: 356-358.
- Finnie JW, Windsor PA, and Kessell AE (2011) Neurological diseases of ruminant livestock in Australia. II: toxic disorders and nutritional deficiencies. Australian Veterinary Journal 89: 247-253.
- Maxie MG, Youseff S (2007) Nervous System, in Jubb, Kennedy and Palmer’sPathology of Domestic Animals, MG Maxie, Editor, Saunders Elseiver.
- Mayhew J (2009) Large Animal Neurology, Wiley-Blackwell, ‘Chichester, United Kingdom’ 337-338.
- Bourke CA (1998) Factors affecting the incidence of the PE 'sudden death' form of Phalaris aquatica poisoning in sheep. in 9th Australian Agronomy Conference. Wagga Wagga, NSW, Australia: Australian Society of Agronomy Inc.
- Sackett D, Holmes P, Abbott K, Jephcott S, Barber M (2006) Assessing the economic cost of endemic disease on the profitability of Australian beef cattle and sheep producers. Meat and Livestock Australia. North Ryde, Sydney, Australia.
- Bourke CA, Colegate SM, Culvenor RA (2006) Evidence that N-methyltyramine does not cause Phalaris aquatica-related sudden death in ruminants. Aust Vet J 84: 426-427.
- McDonald IW (1942) A 'staggers' syndrome in sheep and cattle associated with grazing on Phalaris tuberosa. Australian Veterinary Journal 18: 182-189.
- Le Souef HD (1948) Poisoning of sheep by Phalaris tuberosa. Aust Vet J 24: 12-13.
- Germán Cantón, Carlos Campero, Matías Villa and Ernesto Odriozola (2010) Acute and chronic nervous signs in cattle associated with Phalaris angusta poisoning in Argentina. Pesquisa Veterinaria Brasileira 30: 63-66.
- Gallagher CH, Koch JH, Hoffman H (1966) Diseases of sheep due to ingestion of Phalaris tuberosa. Aust Vet J 42: 279-86.
- Broad M (2006) New light shed on Phalaris toxicity, in Farming Ahead, CSIRO.
- Hungerford TG (1990) Diseases of Livestock. Sydney, Australia. McGraw-Hill.
- Brightling A (2006) Livestock diseases in Australia: diseases of cattle, sheep, goats and farm dogs. Mt. Waverly, Victoria, Australia: C.H. Jerram and Associates.
- Bourke CA, Colegate SM, Slattery S, Oram RN (2003) Suspected Phalaris paradoxa (paradoxa grass) poisoning in horses. Aust Vet J 81: 635-637.
- Southcott WH (1956) Observations on Phalaris staggers in sheep. Aust Vet J 23: 225-228.
- Gallagher CH, Koch JH, Hoffman H (1967) Deaths of ruminants grazingPhalaris tuberosa in Australia. Aust Vet J 43: 495-500.
- Bourke CA, Colegate SM, Rendell D, Bunker EC, Kuhn RP (2005) Peracute ammonia toxicity: a consideration in the pathogenesis of Phalaris aquatica 'Polioencephalomalacia-like sudden death' poisoning of sheep and cattle. Aust Vet J 83: 168-71.
- Bourke CA1, Carrigan MJ (1992) Mechanisms underlying Phalaris aquatica "sudden death" syndrome in sheep. Aust Vet J 69: 165-167.
- Lee HJ and Kuchel RE (1953) The aetiology of phalaris staggers in sheep .1. preliminary observations on the preventive role of cobalt. Australian Journal of Agricultural Research 4: 88-99.
- Bourke CA, Carrigan MJ, Dixon RJ (1988) Experimental evidence that tryptamine alkaloids do not cause Phalaris aquatica sudden death syndrome in sheep. Aust Vet J 65: 218-220.
- Bourke CA, Carrigan MJ, Seaman JT, Evers JV (1987) Delayed development of clinical signs in sheep affected by Phalaris aquatica staggers. Aust Vet J 64: 31-32.
- Choi S, Bunker E, Watt B (2012) Phalaris staggers persisting for seven months post exposure in crossbred lambs.
- Bourke CA, Rendell D, Colegate SM (2003) Clinical observations and differentiation of the peracute Phalaris aquatica poisoning syndrome in sheep known as 'polioencephalomalacia-like sudden death'. Aust Vet J 81: 698-700.
- Culvenor CC, Smith LW and Dalbon R (1964) Occurrence of Indolealkylamine Alkaloids in Phalaris Tuberosa L+P Arundinacea L. Australian Journal of Chemistry 17: 1301.
- Gallagher CH, Koch JH, Hoffman H (1967) Electro-myographic studies on sheep injected with the N,N-dimethylated tryptamine alkaloids of Phalaris tuberosa. Int J Neuropharmacol 6: 223-228.
- Anderton NA, Cockrum PA, Walker DW, Edgar JA (1994) Identification of a Toxin Suspected of Causing Sudden Death in Livestock Grazing Phalaris Pastures. Plant-Association Toxins 269-274.
- Shen HW, Jiang XL, Winter JC, Yu AM (2010) Psychedelic 5-Methoxy-N,N-Dimethyltryptamine: metabolism, pharmacokinetics, drug interactions, and pharmacological actions. Curr Drug Metab 11: 659-666.
- Lamchouri F, Zemzami M, Jossang A, Abdellatif A, Israili ZH, et al. (2013) Cytotoxicity of alkaloids isolated from Peganum harmala seeds. Pak J Pharm Sci 26: 699-706.
- Gambelunghe C, Aroni K, Rossi R, Moretti L, Bacci M (2008) Identification of N,N-dimethyltryptamine and beta-carbolines in psychotropic ayahuasca beverage. Biomed Chromatogr 22: 1056-1059.
- Herraiz T, González D, Ancín-Azpilicueta C, Arán VJ, Guillén H (2010) beta-Carboline alkaloids in Peganum harmala and inhibition of human monoamine oxidase (MAO). Food Chem Toxicol 48: 839-845.
- Sklerov J, Levine B, Moore KA, King T, Fowler D (2005) A fatal intoxication following the ingestion of 5-methoxy-N,N-dimethyltryptamine in an ayahuasca preparation. J Anal Toxicol 29: 838-841.
- Brierley D, Davidson C (2012) Developments in harmine pharmacology - Implications for ayahuasca use and drug-dependence treatment. Prog Neuropsychopharmacol Biol Psychiatry 39: 263-272.
- Liester MB and Prickett JI (2012) Hypotheses Regarding the Mechanisms of Ayahuasca in the Treatment of Addictions. J Psychoactive Drugs 44: 200-208.
- Kennedy DJ, Cregan PD, Glastonbury JR, Golland DT, Day DG (1986) Poisoning of cattle grazing a low-alkaloid cultivar of phalaris-aquatica, sirolan. Aust Vet J 63: 88-89.
- Bourke CA (1997) A comparison between 'sudden death' poisoning in sheep grazing phalaris pastures iat Dubbo, Naracoote and Esperance. in Twelfth Annual Conference of the Grasslands Society of NSW. Grasslands Society of NSW
- Culvenor RA, Reed KFM, and McDonald SE (2005) Comparative levels of dimethyltryptamine- and tyramine-related alkaloid toxins in Australian cultivars and some wild populations of Phalaris aquatica. Australian Journal of Agricultural Research 56: 1395-1403.
- Quinn JC, Kessell A, Weston LA (2014) Secondary plant products causing photosensitization in grazing herbivores: their structure, activity and regulation. Int J Mol Sci 15: 1441-1465.
- Weston PA, Weston LA, Hildebrand PA (2013) Metabolic profiling in Echium plantagineum: presence of bioactive pyrrolizidine alkaloids and naphthoquinones from accenssions across Australia. Phytochem Rev 12: 831-837.
- Gupta RC (2012) Veterinary Toxicology -Basic and Clinical Principles. 2nd Edition. Elseiver: London, UK 1300-1302.
- Hatch RC (1977) Veterinary toxicology, in veterinary pharmacology and therapeutics, Jones LM, Booth LF, Editor, Iowa State University Press: Ames, IA 1253-1259.
- Dinning JS, Briggs HM (1948) Effect of orally administered urea on the ammonia and urea concentration in the blood of cattle and sheep, with observations on blood ammonia levels associated with symptoms of alkalosis. Am J Physiol 153: 41-46.
- Bourke CA (1998) The association of soil cobalt with the incidence of the ‘staggers’ form of Phalaris aquatica poisoning in livestock. in 9th Australian Agronomy Conference. Wagga, NSW, Australia: Australian Society of Agronomy Inc.
- Reid RL and Horvath DJ (1980) Soil chemistry and mineral problems in farm livestock - a review. Animal Feed Science and Technology 5: 95-167.
- Moore RM, Hutchings RJ (1967) Mortalities among sheep grazing Phalaris tuberosa. Australian Journal of Experimental Agriculture and Animal Husbandry 7: 17-21.
- Palit S, Sharma A, and Talukder G (1994) Effects of cobalt on plants. The Botanical Review 60: 149-181.
- Marten GC, Simons AB and Frelich JR (1974) Alkaloids of reed canarygrass as influenced by nutrient supply. Agronomy Journal 66: 363-368.
- Majak W, McDairmid RE, Powell TW, Van Ryswyk AL, Stout DG, et al. (1979) Relationships between alkaloids in reed canary grass (Phalaris arundinacea), soil, moisture and nitrogen fertility. Plant, cell and environment 2: 335-340.
- Crofts F (1997) Australian pasture production: the last 50 years. Pasture Production and Management. Editors S Lovett JV, JS Inkata Press: Melbourne, Australia 1-16.
- Wheeler JL, Pearson CJ, Robards GE (1986) Temperate Pastures: Their Production Use and Management, 1986, CSIRO Publishing, East Melbourne, Australia.
- Culvenor RA and Boschma SP (2005) Evaluation of phalaris (Phalaris aquatica L.) germplasm for persistence under grazing on the North-West Slopes, New South Wales. Australian Journal of Agricultural Research 56: 731-741.
- Oram RN (1970) Genetic and environmental control of the amount and composition of toxins in Phalris tuberosa L. in XI International Grasslands Conference. Queensland: University of Queensland Press.