Journal of Proteomics & Computational Biology
Download PDF
Research Article
Peptides Digested from Some Allium Sativum and Solanum lycopersicum Proteins Serve as Antihypertensive Agents: Computational Analysis
Kanawati A1 and Al-Madhagi HA2*
1Division of Biochemistry, Chemistry Department, Aleppo University,
Syrian Arab Republic
2Biochemical Technology Program, Faulty of Applied Sciences,
Dhamar University, Yemen
*Address for Correspondence:
Al-Madhagi HA, Biochemical Technology Program, Faulty of Applied
Sciences, Dhamar University, Yemen, E-mail: bio.haitham@gmail.com
Submission: 21 September, 2022
Accepted: 25 October, 2022
Published: 29 October, 2022
Copyright: © 2022 Kanawati A, et al. This is an open access article
distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Abstract
Hypertension (HTN) still constitutes a worldwide problem to the
healthcare section and individual’s life. The current angiotensin
converting enzyme (ACE) inhibitors are first-choice option but pose
deleterious side effects upon prolonged administration. The goal of the
present in silico study is to evaluate the Allium sativum and Solanum
lycopersicum proteins as source for anti-hypertensive natural peptides.
The corresponding protein sequences were obtained from the UniProt
database and then inputted to antihypertensive peptides predictor
online tool. Using the same tool, 3 digestive enzymes (pepsin, trypsin
and elastase 1) were chosen for the digestion of proteins into small peptides
and subsequently assess their hypotensive activity. Some biochemical
characteristics of positive bioactive peptides were calculated via
Pepstats web interface while HAPPENN tool examined hemotoxicity.
Afterwards, the secondary structure of positive bioactive peptides was
de novo predicted through PEPstrMOD server prior to docking against
human ACE (PDB ID: 1o86) using HPEPDOCK and ClusPro platforms.
A.sativum peptides are more potent than S.lycopersicum ones in
terms of IC50 as well as docking score albeit neither of which showed
hemotoxicity. The present in silico work suggests the hypotensive
activity of A.sativum and S.lycopersicum as natural treatment option
of HTN.
Keywords
Hypertension; Anti-hypertensive peptide; Peptide-protein
docking; Allium sativum; Solanum lycopersicum
Introduction
Hypertension (HTN) continues to be the second main risk factor
of death cases from cardio- and cerebrovascular accidents as well as
end-organs damage globally [1]. It is estimated in the United States
(US) that one in every three persons suffers from HTN [2]. The recent
redefinition of HTN is blood pressure (BP) levels ≥ 130/80 mm Hg or
having HTN medicines. Accordingly, the percentage of prevalence
of HTN in US rises to 50% [3]. Moreover, about half of COVID-19-
hospitalized patients have HTN which increases the risk of mortality
even more [4].
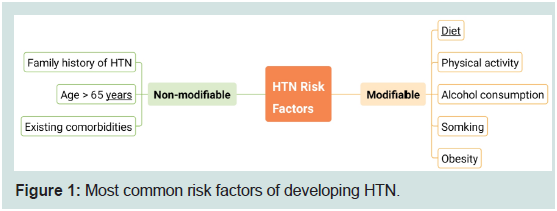
Various risk factors are traced to drive BP up that can be
categorized into modifiable and non-modifiable factors (Figure 1).
Modifiable factors include, but not limited to, diet (particularly Narich
fast foods), sedentary lifestyle and obese people. Non-modifiable
factors involve aging and the presence of chronic diseases besides
such as type 2 diabetes and kidney diseases [5,6]. In addition, genetic
inheritance and, more recently, epigenetics programming especially
in prenatal stage increases the incidence of HTN later in adulthood
[7].
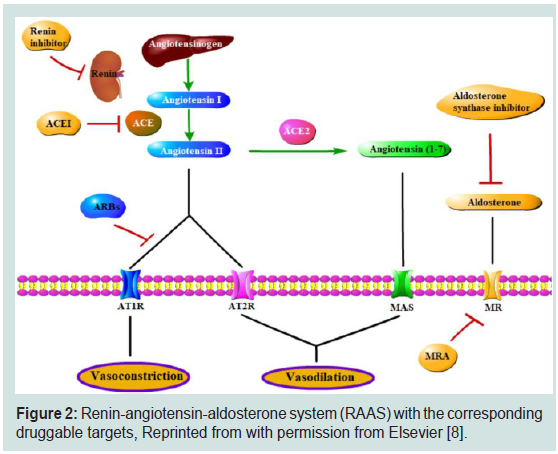
Once patient’s BP got elevated, therapeutic intervention should be introduced to restore the levels of BP to the accepted values.
One of the mainstay pathways regulating BP is renin-angiotensinaldosterone
system (RAAS) (Figure 2). As the name implies, there
are 3 cooperating hormones to control BP, renin (also known as
angiotensinogenase), angiotensin and aldosterone. Driven by the
HTN, juxtaglomerular cells of kidney sense this rise in BP and respond
by secreting renin to the bloodstream. Therein, it proteolytically activates angiotensinogen produced from liver forming angiotensin
I and the latter catalyzed by the enzyme angiotensin-converting
enzyme 1 (ACE1) is cleaved further to angiotensin II. The produced
angiotensin II acts as powerful vasoconstrictor by activating its
corresponding receptors ATR (angiotensin II receptors) on smooth
muscle cells. With the assistance of ATR binding by angiotensin
II, aldosterone is secreted from adrenal gland to enhance Na
reabsorption contributing further to the establishment of HTN [8].
Therefore, interrupting ACE1 action cancels the vasoconstriction as
well as Na reabsorption actions reducing BP to the normal set. ACE1
inhibitors are considered first-choice treatment of HTN [9].
Figure 2: Renin-angiotensin-aldosterone system (RAAS) with the corresponding
druggable targets, Reprinted with permission from Elsevier [8].
Not only small organic molecules can achieve ACE1 inhibition.
Many food proteins upon enzymic digestion in the gut releases
bioactive peptides exhibiting inhibitory action toward different
targets of RAAS including ACE1 [10,11]. So, we purposed to explore
the anti-hypertensive activity of peptides digested from some
A.sativum proteins in silico. The present study provides a probable
means to nutraceutically treat HTN through the food taken on a
regular daily routine.
Materials & Methodology
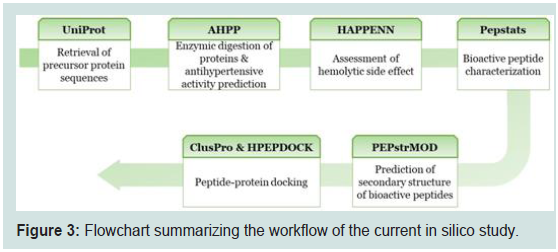
Workflow:
The Repeated workflow strategy employed in this study is
illustrated in Figure 3. However, the details of each step are explained
below.Source protein sequences retrieval:
The primary structure (amino acid sequence) of the examined
proteins was retrieved from UniProt database (https://www.
uniprot.org/) [12]. Three proteins were chosen randomly: Alliin
lyase 1 (Uniprot ID#Q01594), mannose-specific lectin (Uniprot
ID#P83886) and peroxidase (Uniprot ID#H2CLW6) limiting the
organism to Allium sativum only. Similarly, three proteins from
Solanum lycopersicum were selected for further analysis (Uniprot
ID#P27260, G1JUH1 & C1K5M2).Proteins digestion and prediction of antihypertensive activity:
The 3 mentioned proteins were subjected to three digestive
enzymes (randomly chosen) namely: Pepsin, Trypsin and Elastase
1 in combination. Afterwards, the anti-hypertensive activity of the
generated peptides were evaluated using anti-hypertensive peptide
predictor (AHPP) server [13]. This server enables enzymatic digestion
and activity assessment simultaneously. Only the peptide showed
positive anti-hypertensive activity and having ≥ 7 AA were selected
for further analysis.Examining hemolytic activity of the bioactive peptides:
Despite of posing biological activity, some peptides have a
coinciding side effects including hemotoxicity and many neglect
such aspect [14]. HAPPENN web server was utilized to examine the
hemotxicity of the positive peptides [15]. Results are expressed as
probability score between 0 (non-hemolytic) to 1 (highly hemolytic).Biochemical features of the bioactive peptides:
The peptides succeeded in achieving positive antihypertensive
activity and had at least seven AA were uploaded to pepstats web
server (https://www.ebi.ac.uk/Tools/seqstats/emboss_pepstats/) for
detecting some characteristics such as molecular weight, isoelectric
point and charge, among others. The obtained results were collected
and tubulated.Docking analysis:
The secondary structure of the best 3 peptides were predicted
de novo via PEPstrMOD online tool [16] (https://webs.iiitd.edu.
in/raghava/pepstrmod/) in provision to be docked against human
ACE1(PDB ID: 1O86) after removal of water molecules, chloride
and zinc ions and the inhibitor lisinopril by UCSF Chimera v1.16
[17]. Then, peptides and receptor PDB files to uploaded to and
HPEPDOCK protein-protein docking platform [18].Results
Biochemical features and anti-hypertensive peptide prediction:
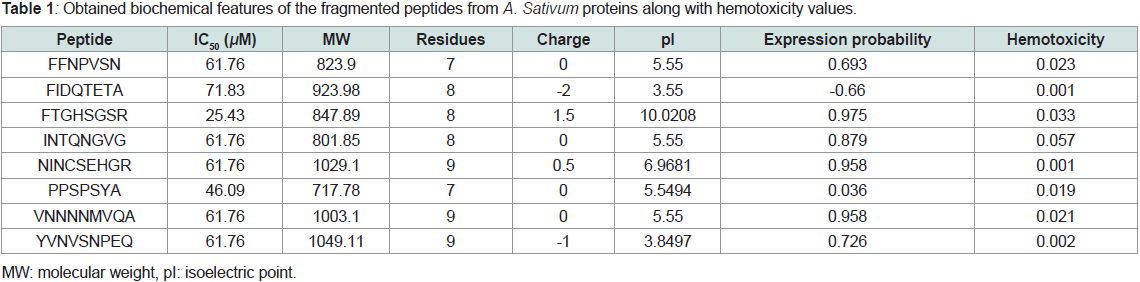
Table 1 Obtained biochemical features of the fragmented peptides
from A.sativum proteins along with hemotoxicity values.The 3 examined proteins of A.sativum, only 8 peptides found
positive (antihypertensive). As we chose 3 digestive enzymes (pepsin,
trypsin and elastase 1) the residues were short (<10 AA). Most of
the fragmented peptides had neutral charge and, hence, pI around
5. Furthermore, the digested peptides posed high probability of
being expressed in inclusion bodies which facilitates the large-scale
production and isolation of the examined peptides. Moreover, none
of the peptides showed hemolytic activity as reflected by values near
zero (indicative of non-hemolytic) (Table 1). Overall, the digested
peptides from A.sativum are both anti-hypertensive as well as nonhemolytic
peptides particularly 3rd peptide (FTGHSGSR). This
outcome is good for provision to molecular docking against human
ACE1 to further confirm the findings.
Table 1: Obtained biochemical features of the fragmented peptides from A. Sativum proteins along with hemotoxicity values.
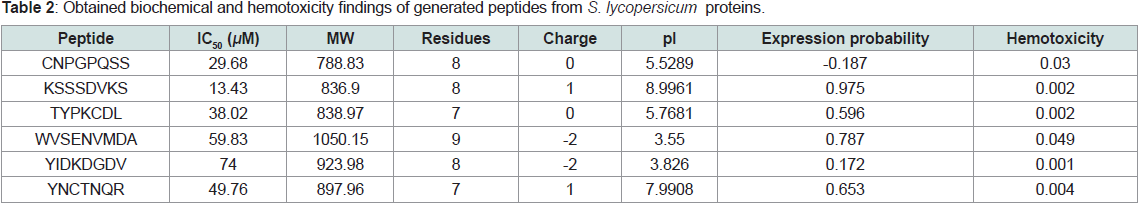
Only 6 positive peptides were obtained from precursor proteins
of Solanum lycopersicum. The fragmented peptides were significantly
potent in terms of anti-hypertensive activity as mirrored by the IC50
values. The digested peptides are composed of a combination of
neutral, acidic and basic amino acids with net charge between -2 to
1 and pI in the range 3.55 to 8.9. In addition, none of the obtained
peptides exhibited hemolytic activity (Table 2).
Table 2: Obtained biochemical and hemotoxicity findings of generated peptides from S. lycopersicum proteins.
Docking analysis:
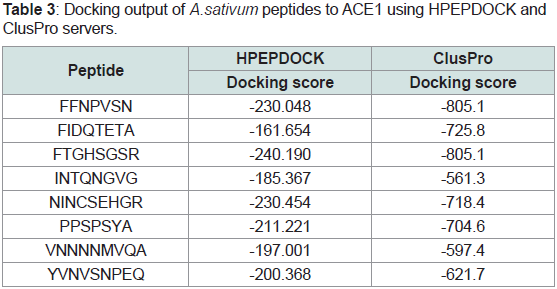
Table 3 Docking output of A.sativum peptides to ACE1 using
HPEPDOCK and ClusPro serversApparently from Table 3, the fragmented peptides from allium
sativum exhibited good inhibition profile toward ACE1, confirming
thus their anti-hypertensive activity predicted via AHPP server. The docking scores in HPEPDOCK ranged from -185 to -240 kcal/mole.
Similarly, from -561 up to -805 kcal/mole predicted against ACE1
via ClusPro platform. The ranking of peptides in the two servers are
almost the same which confirm the results of each other with the 3rd
peptide were the best in the two servers.
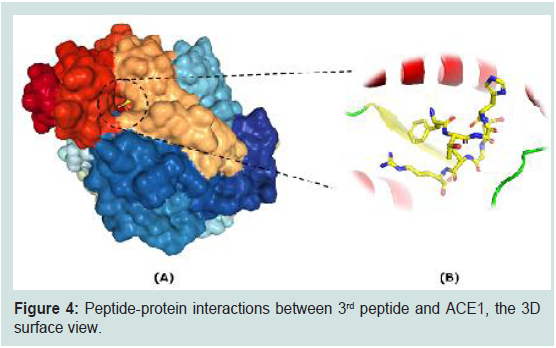
To further reveal the detailed peptide-protein interactions
between 3rd peptide and ACE1, the 3D surface view and indepthrepresentation
have been performed (Figure 4). It has been
figured out that peptides lie well within the active center of ACE1 as
shown in surface view (Figure 4A).
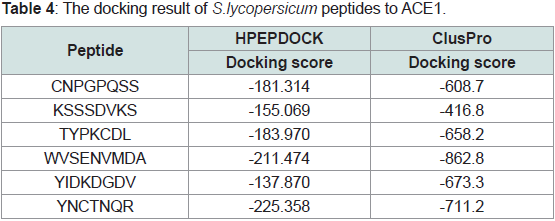
The docking profile of the digested peptides from S.lycopersicum
showed slightly weaker binding energy to ACE1 explored by
HPEPDOCK server in comparison to A.sativum peptides. While the highest docking score of S.lycopersicum peptides were peptide 6 which
gave inhibitory value of -225 kcal/mole, 3rd peptide of A.sativum
had a docking score of -240 kcal/mole, suggesting better potency.
Nonetheless, peptide 4 of S.lycopersicum showed best docking energy
compared to the 6th peptide in HPEPDOCK (-862 vs -711 kcal/mole)
(Table 4). This docking energy is even stronger than best docking
energy of 3rd peptide (best one) of A. sativum peptides.
Discussion
Developing HTN worsen individual’s health life since it is
accompanied by complications progression such as cardiovascular
diseases, stroke and dementia. Beyond 1970s, BP levels have been
declined in high-income nations whilst raised substantially in
Middle-east and Southeast regions [19]. In 2016, about third of adult
population worldwide are diagnosed with HTN [20]. Men on common
have age-standardized systolic BP higher than women counterparts
in several countries [21]. This data necessitates should urge to find
more potent therapeutics with minimized side effects because the
first-choice treatments (ACE1 inhibitors) exhibit deleterious side
effects involving headache, visual disturbances, cough, dizziness and
insomnia on prolonged use [22]. In recent years, the direction of HTN treatment has been transformed into food peptides including milk,
egg, meat and marine meats. The hypotensive peptides are released
upon hydrolysis through gastrointestinal tract enzymes [23]. Jogi
et al. reviewed long list of hypotensive peptides derived from food
sources [24]. However, garlic and tomato-hypotensive peptides were
not mentioned. The present in silico study was designed to simulate
the in vitro and in vivo works for assessment of hypotensive peptides
generated from food sources and predict their ACE1 inhibition. The
results of the current study suggest the hypotensive activity of both
garlic (A.sativum) and tomato (S.lycopersicum) as evidenced by IC50
values and validating molecular docking against ACE1 target albeit
garlic peptides are more potent. Moreover, the fragmented peptides
are non-hemolytic and had a high probability of being expressed
in inclusion bodies for large-scale production. Overall, the present
theoretical exploration provided a notable suggestion that should
be validated by in-vitro and in-vivo experiments
References
10. Aluko RE (2015) Antihypertensive peptides from food proteins. Annu Rev Food Sci Technol 6: 235-262.
Citation
Kanawati A, Al-Madhagi HA. Peptides Digested from Some Allium Sativum and Solanum lycopersicum Proteins Serve as Antihypertensive
Agents: Computational Analysis. J Proteomics Computational Biol. 2022;5(1): 4.