International Journal of Otorhinolaryngology
Download PDF
Case Report
Rhino-pulmonary Mucormycosis in Post COVID-19 patient
Nagi K1*, Manners S2, Alexander A2 and Saxena SK2
1Oking Hospital and Research Centre, Kohima, Nagaland, India
2Department of Otorhinolaryngology, Jawaharlal Institute of Postgraduate
Medical Education & Research, Puducherry, India
*Address for Correspondence
Nagi K, Oking Hospital and Research Centre, Kohima,
Nagaland, India, Email: kezenagi77@gmail.com
Submission: 03 March, 2022
Accepted: 25 April, 2022
Published: 28 April, 2022
Copyright: © 2022 Nagi K, et al. This is an open access article
distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction
in any medium, provided the original work is properly cited
Abstract
Invasive fungal sinusitis is a rising concern among individuals with
COVID 19, as the recent trend shows a three-fold increase in cases
during the second wave of the pandemic. Mucormycosis caused
by the fungi of the Mucorales group can manifest with florid clinical
presentations depending on the various sites it can involve, the
most common being Rhino-orbital-cerebral Mucormycosis. Current
literature review reports few cases of pulmonary mucormycosis and
even rare is the presentation with concurrent sinonasal involvement.
Here we report a case of Rhino-pulmonary mucormycosis in a Post
Covid Diabetic patient.
Keywords
Mucormycosis; COVID-19; Diabetes
Case Report
62-year-old male, with a history of Diabetes, Hypertension&
Coronary artery disease for 7 years, presented to our tertiary centre
with chief complaints of right-sided chest pain for 3 days which was
dull aching, moderate in severity increased on inspiration, nonradiating
and not associated with palpitation or perspiration, with
one episode of blood-tinged sputum and occasional cough. No c/o
breathlessness. No headache or nasal complaints. He had COVID-19
disease two weeks prior for which he received Inj methylprednisolone
30mg for 10 days, Injpiptaz 4.5gm TID for 3 days, Tab fluconazole
200mg BD for 6 days & Tab Levofloxacin 250mg BD for 5 days,
during the course of his hospital stay. He was not treated with antivirals
or antibodies.
On examination, the patient was conscious. Vitals were
stable. On auscultation, coarse crepitations were heard in the right
infrascapular and interscapular areas. ENT examination was normal.
Blood investigations revealed Blood Glucose 358mg/dl, Hemoglobin
12.5g/dl, White blood cell count 18,250/microlite, Neutrophil
88%, Lymphocyte 8% platelet count 2.6 Lac, Hemoglobin A1c was
9%. Urine Ketones was negative, C-Reactive protein 22.3mg/L &
Viral markers were negative. Chest X-ray revealed right pleural
thickening with surrounding homogenous opacity. Sputum acidfast
bacilli (AFB) and culture were negative. High-resolution CT of
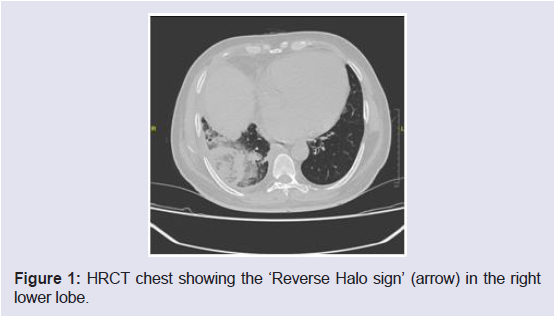
the chest revealed central ground-glass opacity surrounded by dense
consolidation suggestive of reverse Halo sign in the right lower lobe
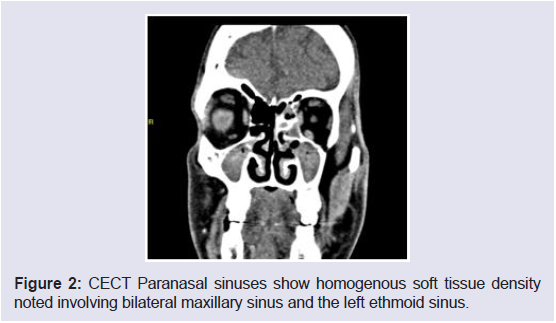
[Figure 1]. Following which a CT of the Nose & Paranasal sinus was
taken to rule out ENT foci of fungal infection i/v/o high suspicion
for mucormycosis, it revealed a homogenous soft tissue density in
the bilateral maxillary sinus and left ethmoid with no intraorbital or
intracranial extension [Figure 2].
Figure 2: CECT Paranasal sinuses show homogenous soft tissue density
noted involving bilateral maxillary sinus and the left ethmoid sinus.
A diagnostic nasal endoscopy revealed blackish necrotic middle
turbinate and crust along the left middle meatus, which was sent for
KOH fungal mount that reportedbroad aseptate hyphae suggestive of
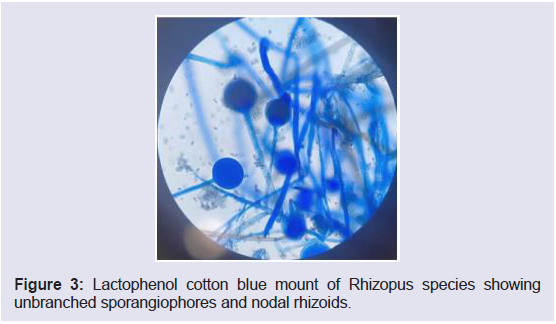
Mucormycosis. Subsequently, Sputum fungal culture grew Rhizopus
species [Figure 3].
Figure 3: Lactophenol cotton blue mount of Rhizopus species showing
unbranched sporangiophores and nodal rhizoids.
A diagnosis of Pulmonary with concomitant sino-nasal
mucormycosis was made. The patient was started on Injection Amphotericin therapy & was scheduled for Endoscopic surgical
debridement. However, his general condition deteriorated, and he
developed sudden refractory hypotension with respiratory failure and
succumbed to it.
Discussion
Among the risk factors for mucormycosis, he had uncontrolled
diabetic status and a history of corticosteroid therapy for COVID
19 illness. Literature review reports that the incidence of pulmonary
mucormycosis relative to other clinical forms is 25 % [1]. It has a
fatality rate of > 50% [2].
Pulmonary mucormycosis results from the inhalation of
sporangiospores or by hematogenous or lymphatic spread and
presents with nonspecific symptoms like cough, dyspnea, chest pain,
and fever [2,4]. Mani et al reported a case of isolated pulmonary
mucormycosis which initially mimicked malignancy, this patient had
uncontrolled diabetic status but without ketoacidosis and prolonged
corticosteroid therapy for intractable asthma concluding that isolated
pulmonary mucormycosis is extremely rare and carries a poorer
prognosis than rhino-orbital-cerebral disease [3].
The pathogenesis of fungal infection depends on the interaction
of host factors (epithelial defence mechanism) & virulence of the
organism. Hyperglycemic state and in particular ketoacidosis impairs
the ability of neutrophils to destroy the Mucorales hyphae. Also,
ketoacidosis increases the availability of free iron which is used as a
growth factor by Mucorales. Pulmonary mucormycosis may induce
pulmonary embolism because Mucorales species tend to invade the
elastic intima of large and small vessels, causing thrombosis, bleeding,
and infarction, which should be considered a differential in patients
presenting with pulmonary embolism of unknown cause [5,6].
Imaging and nasal endoscopy revealed stage 2 ROCM (Rhinoorbital-
cerebral Mucormycosis) with pulmonary involvement in our
patient. The reverse halo sign which is a ground glass lesion with
a peripheral rim of consolidation is a specific sign of pulmonary
mucormycosis on a high-resolution CT scan which was also seen in
our patient [7,8]. Immunocompromised state being a prerequisite
for invasive fungal infection, the sequelae includes angioinvasion
and direct tissue injury of the respiratory tract and extension to great
vessels, invasion from the paranasal sinuses into the orbit and brain,
and hematogenous dissemination [2].
Histopathology and Fungal mount microscopy along with culture
for various clinical specimens are the major diagnostic modalities
for mucormycosis. Sputum and BAL cytology are unpredictable
and may be negative [2]. In our patient, the KOH mount of the
middle turbinate tissue grew broad aseptate hyphae and sputum
fungal culture revealed Rhizopus species. In the current COVID 19
pandemic era, there has been a three-fold increase in the cases of
invasive fungal infection attributed to the wide use of steroid therapy
and concurrent uncontrolled diabetic status.
The differential diagnosis of pulmonary mucormycosis involves
bacterial, viral and other fungal pneumonia which becomes a
diagnostic challenge [9,10]. Aspergillosis remains a very close
differential and hence in a case of suspected fungal infection
unresponsive to voriconazole strongly favours mucormycosis [4].
In cases where there is a high suspicion of a fungal aetiology
inpost-COVID patients who are diabetic and have received steroid
therapy, nose and paranasal foci of infection should be investigated
even though clinically they are asymptomatic as seen in our case
which will expedite in confirming the diagnosis and early initiation of
antifungal therapy and surgical debridement.
Treatment should be continued for a minimum of twelve
weeks but especially until all the clinical and radiological symptoms
are resolved and cultures have become negative. Strengthening
immunity along with risk factors such as diabetes and protein-energy
malnutrition should be treated and kept under control. Surgery
should always be considered in the management of mucormycosis
more so in the case of localised diseaseas it allows systemic treatment
to reach the infected areas isolated by vascular thrombotic events, and
reduces the fungal burden [4].
Few learning points through this case study:
➢ Pulmonary mucormycosis can be a devastating and lifethreatening
infection if not correctly diagnosed and treated.
➢ ConcomitantSinonasal and pulmonary involvement should
be kept in mind during diagnostic dilemmas and hence promoting
early specific management of mucormycosis.
➢ Invasive fungal sinusitis is a rising concern among
individuals with COVID 19, as the recent trend shows a three-fold
increase in cases during the second wave of the pandemic.
Conclusion
Mucormycosis is a life-threatening infection mostly occurring
in the immunocompromised host. The risk is many including
haematological malignancies, uncontrolled diabetes mellitus,
immunocompromised states, and Steroid therapy. Early diagnosis
and treatment which includes surgery and antifungal drugs can
improve outcomes and survival. Definitive diagnosis requires
pathologic demonstration of the organism in affected tissue.
Concurrent Sinonasal and pulmonary involvement should be kept in
mind during diagnostic dilemmas and hence promoting early specific
management of mucormycosis.
References
Citation
Nagi K, Manners S, Alexander A, Saxena SK. Rhino-pulmonary Mucormycosis in Post COVID-19 patient. Inter J Otorhinolaryngology. 2022;8(1): 3.