Journal of Orthopedics & Rheumatology
Download PDF
Research Article
Prevalence of Metabolic Syndrome in Patients with Ankylosing Spondylitis
El Hassani Sbai S*, Rostom S, Amine B and Bahiri R
Department of Rheumatology A (Pr R. Bahiri), University Hospital of Rabat-Sale, Morocco
*Address for Correspondence: El Hassani Sbai S, Department of Rheumatology A (Pr R Bahiri), El Ayachi
Hospital, 11000, Sale, Morocco, Tel: 00-212-537-78-29-19 / 17-14 Fax: 00-
212-537-88-33-27; E-mail: elhassanisalma1@gmail.com
Submission: 13 November 2019;
Accepted: 18 December 2019;
Published: 20 December 2019
Copyright: © 2019 El Hassani Sbai S, et al. This is an open access article
distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Abstract
Background: The aim of our study was to assess the prevalence of
metabolic syndrome in ankylosing spondylitis (As) and identify factors
that are associated with its presence.
Methods: A cross-sectional study including AS patients fulfilling
the modified 1984 New York criteria for AS criteria, and age- and sexmatched
controls.
MetS prevalence was assessed, using six MetS definitions (Joint
Consensus, National Cholesterol Education Program 2004 and 2001,
International Diabetes Federation, World Health Organization and
European Group for Study of Insulin Resistance). Physical activity was
assessed by the IPAQ (international physical activity questionnaire) short
version. Quality of life was assessed by the SF-36 (Short Form 36).
Results: One hundred and ten AS patients were included with 68%
male and median age 36 years [23-44]. The control group consisted
of 100 healthy control subjects with 67% male and median age 35
years [21-44]. MetS prevalence rates varied from 8.2% to 13.6% in AS
according to the definition used, when it was between 1 to 5% in the
control group( p=0,01). In multivariate analysis only persists a significant
association between MetS and patients ages (OR=1.12; IC [1.04-1.21];
p=0.003).
Conclusion: In this study, the frequency of MetS was observed to be
higher in patients with AS than the controls for every definition of MetS.
The occurrence of metabolic syndrome appears to be related to age
regardless to the activity, severity and treatment of AS.
Keywords
Ankylosing spondylitis; Metabolic syndrome; Prevalence
Introduction
Ankylosing Spondylitis (AS) is a chronic inflammatory disease
which especially involves the axial skeletal system although it may
also affect the peripheral joints and the extra-articular structures
[1,2]. The definite pathogenesis of AS has not yet been determined
[3].
Excess mortality has been documented in patients with
Ankylosing Spondylitis (AS). The overall mortality rate in AS
patients is 1.6- to 1.9-fold that in the general population, and the
excess cardiovascular mortality has been estimated at 20 to 40% [4-6].
Some studies have noted an increased morbidity and mortality in AS
patients compared with the general population and Epidemiological
studies have produced sound evidence that the risk of cardiovascular
disease is increased in patients with AS [3]. However, they cannot
determine whether the risk increase is due to an independent effect of
the AS or to an increase in the prevalence of conventional risk factors
[6].
Metabolic syndrome main components are dyslipidemia
(elevated triglycerides and apolipoprotein B (apoB) containing
lipoproteins, and low High-Density Lipoproteins (HDL), elevation of
arterial Blood Pressure (BP) and dysregulated glucose homeostasis,
while abdominal obesity and/or Insulin Resistance (IR) have gained
increasing attention as the core manifestations of the syndrome [7].
The metabolic syndrome is recognised as a cluster of cardiovascular risk factors [8]. Metabolic Syndrome (MS) is the clinical condition
where risk factors for the development of cardiovascular diseases
and diabetes mellitus accumulate [9]. Certain studies point to the
relationship between the metabolic syndrome and the inflammation
[10,11]. It has been reported that the prevalence of MS is significantly
increased in patients with inflammatory diseases like rheumatoid
arthritis, psoriasis or AS than in the general population [12-17]. There
is still little information regarding the prevalence of MetS in patients
with AS. Few studies were conducted in European countries but no
one in African countries where genetics, comorbidities and toxic
habits are different. MetS has not yet been studied among patients
with AS in Morocco. Therefore, the present study was designed to
assess the prevalence of MetS according to all definitions currently
used, in order to compare between other studies and identify the
potential factors that associate with its presence.
Patients and Methods
Patients:
110 consecutive patients with AS fulfilling the 1984 Modified
New York Criteria who participated in the study were included [18].Patients with other inflammatory articular diseases, malignancies,
diseases of the central nervous system, chronic kidney disease, chronic
liver disease besides AS, were excluded from the study.
Informed consent was obtained from all subjects and the study
was approved by the ethics committee of our university.
Clinical assessments:
Demographic characteristics (age, sex, weight and height and
level of education), disease- specific variables (disease duration,
duration of morning stiffness, the number of nocturnal awakenings,
tender and swollen joint count,…) drug use (all anti-rheumatic
drugs, glucocorticoid use, cardiovascular drugs and analgesics among
others), comorbid conditions, and family history of rheumatic and
cardiovascular diseases were documented for each patient.Disease activity and function:
The disease activities of our patients were evaluated based on the
Bath AnkylosingSpondylitis Disease Activity Index (BASDAI) [19]. Functional
status was assessed by Bath Ankylosing Spondylitis Functional Index
(BASFI) [20,21]. Axial pain was assessed by VAS (visual analogue
scale) and the mini-BASDAI (Question (Q)1 + Q2)+(Q5+Q6) /2]/
3 [22].
Physical activity:
The short, last-week Arabic version of the International Physical
Activity Questionnaire (IPAQ) was administered, asking about the
time spent being physically active in the last 7 days [23]. Minutes
of sitting, walking, moderate-intensity (walking not included), and
vigorous- intensity activities were counted for the last week. The
items in the short IPAQ form were structured to provide separate
scores on walking, moderate-intensity and vigorous-intensity
activity. Computation of the total score for the short form requires
summation of the duration (in minutes) and frequency (days) of
walking, moderate-intensity and vigorous-intensity activities.Quality of life:
The Arabic version of the Short-Form 36 of the medical Outcome
Study (SF-36), validated previously [24], was completed. The SF36
is a generic instrument providing information about eight different
aspects of subjective health. The items of the SF-36 are grouped
into eight subscales: physical function, limitations due to physical
health problems, bodily pain, general health, vitality, social function,
limitation due to emotional health problems, mental health.Ascertainment of depression and anxiety:
The Hospital Anxiety and Depression Scale (HADS) was used
in patients to assess depression and anxiety. The Hospital Anxiety
and Depression Scale (HADS) is a 14-item scale designed to detect anxiety and depression, independent of somatic symptoms. It
consists of two 7- item subscales measuring depression and anxiety.
A 4- point response scale (from 0 representing absence of symptoms,
to 3 representing maximum symptomatology) is used, with possible
scores for each subscale ranging from 0 to 21[25].Body composition:
Body Mass Index (BMI) was calculated from weight/ height2 (kg/m2). BMI values < 18.5 kg/m2 are considered underweight, between
18.5-24.9 as normal, 25-29.9 as overweight and values greater than
30 indicate obesity [26]. Waist Circumference (WC) was measured
to the nearest 0.5 cm midway between the iliac crest and the lower
rib margin. According to the International Diabetes Federation
(IDF) a waist circumference value less than 80 cm indicate low risk
of type 2 diabetes, coronary heart disease or hypertension [27]. The
systolic and diastolic blood pressure was measured by a mercury
sphygmomanometer in the sitting position after five minutes of rest.Metabolic syndrome:
The metabolic syndrome, is a cluster of classical cardiovascular
risk factors (obesity, glucose intolerance, dyslipidemia, and
hypertension) thought to associate with cardiovascular risk beyond
the sum of its individual components [28], although this has recently
been questioned.Currently used criteria to define MetS:
No consensus has been reached regarding the definition of MetS.
Several groups have attempted to establish diagnostic criteria and
the most widely used have been provided by the many international
organizations and expert groups, such as the World Health
Organization (WHO) [29], the European Group for the study of
Insulin Resistance (EGIR) [30], the National Cholesterol Education
Program Adult Treatment Panel III (NCEP:ATPIII) [31,32], the
International Diabetes Federation (IDF) and the Joint Consensus (JC)
[33,34], have attempted to incorporate all the different parameters
used to define MetS.In this study, the prevalence of the MetS was analysed according
to all existing definitions (JC, NCEP 2004, NCEP 2001, WHO, IDF,
EGIR) in order to establish the range of discrepancy between them.
For further analysis of the predictors of the metabolic syndrome only
the NCEP 2004 definition is presented, as this is most widely used
definition reported in the literature, thus allowing comparisons to be
drawn with other studies [32].
Biochemical measures:
Venous blood samples were drawn after an overnight fast.
C-reactive protein (CRP), Erythrocyte sedimentation rate (ESR),
plasma glucose, total cholesterol, low-density lipoprotein (LDL) and
high-density and lipoprotein (HDL) were determined by standard
laboratory methods. Concentrations of total cholesterol > 5.0
mmol/L, LDL ≥ 3.0 mmol/L, HDL < 1.3 mmol/L were considered
pathologic [32].Control group:
The control group consisted of 100 healthy individuals of
matching age and sex groups.Statistical analysis:
Statistical analysis was performed with SPSS 10, 0 statistical
software packages. The statistical analysis of patient questionnaire data
involved computation of means, medians, standard deviations, and
ranges for quantitative variables; and numbers and percentages for
qualitative variables. Sample t test were used to compare quantitative
data and the chi-square test for qualitative data. Multivariate logistic
regression models were constructed and Odds Ratios (OR) and
95% Confidence Interval (CI) were calculated to investigate the
independent of the predictors of individual AS-related characteristics
and MetS.Results
Description of the AS patients:
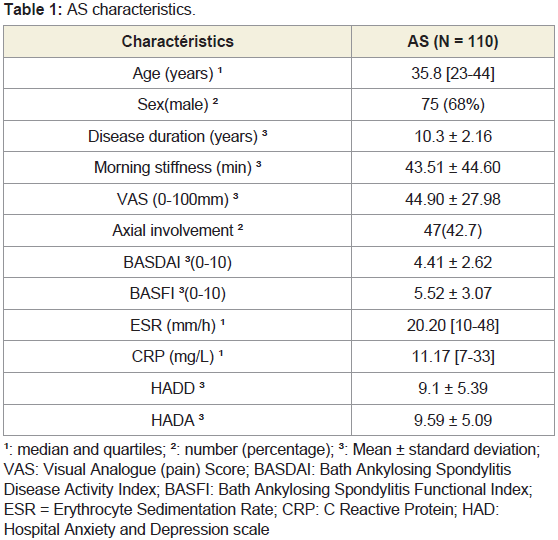
Characteristics of patients with AS are summarised in (Table 1).
Patients had mean disease duration of 10.3 years ± 2.16, had mean morning stiffness of 43.51 min ± 44.60 and had mean axial pain of
44.90 mm ± 27.98 ; 42.7% of our patients had a pure axial AS. They
had moderate disease activity (the mean BASDAI 4.41 ± 2.62), the
mean BASFI was 5.52 ± 3.07. Regarding inflammation the median
of ESR was 20.20 mm/h [10-48] and median of CRP was 11.17 mg/l
[7-33].Characteristics of the two groups:
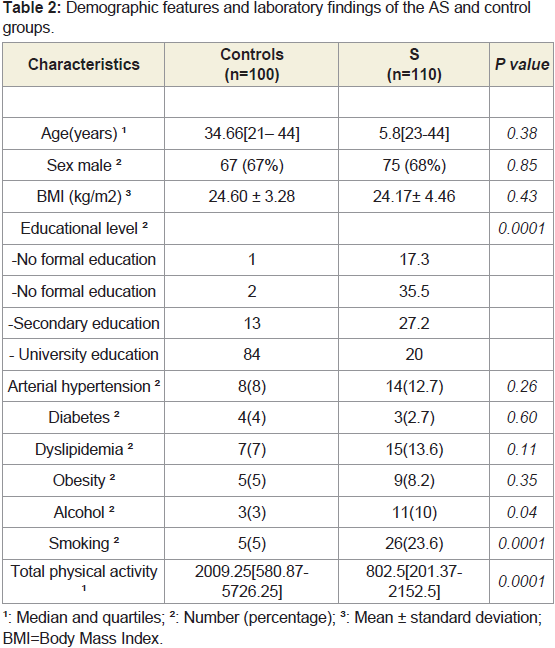
A summary of the socio-demographic and clinical characteristics
of the two groups is presented in (Table 2). Both the AS and the
control groups were similar in terms of age distribution (P =0.38),
with median age in AS group 35.8 [23-44] and in control group 34.66
[21-44]. Most of the patients were male (68% in AS group and 67%
in controls). Also there was no difference statistically significant
between the two groups concerning the body mass index. Educational
level was much higher in the control group compared to AS group
(P=0.0001).In the AS group there were 12.7% hypertensive patients,
2.7% diabetics, 13.6% with dyslipidemia and 8.2% with obesity,
whereas in control group 8% were hypertensive, 4% diabetics, 7%
with dyslipidemia and 5% with obesity. There were more alcohol
consumption and tobacco use in the AS group compared to controls
(P<0.05). Concerning physical activity it was more reduced in AS
group than controls (p=0.0001).Prevalence of the metabolic syndrome in study population according to definition used:
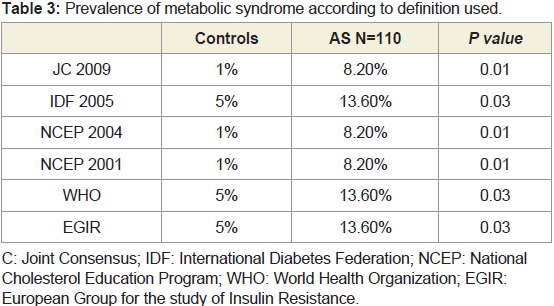
There was great diversity in the reported prevalence rates
according to the definition used (Table 3). The prevalence of MS
ranged from 8.2% to 13.6% in AS group, whereas it ranged from 1%
to 5% in control group. The difference between them was statistically
significant (P < 0.05).Associations of the metabolic syndrome in patients with AS:
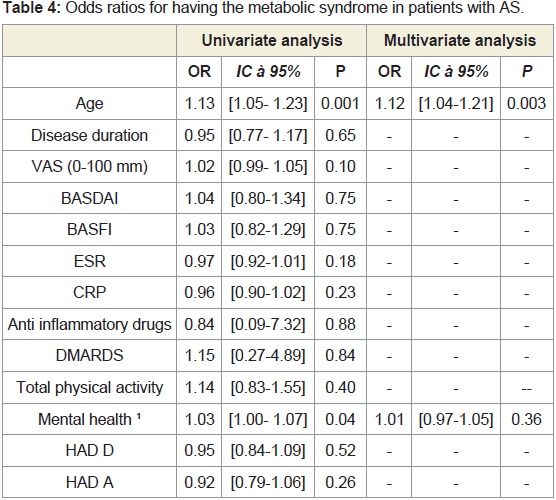
Using the definition of metabolic syndrome according to NCEP
2004 we assessed parameters of AS that were associated with MetS in
univariate and multivariate analysis (Table 4).In Univariate analysis, it has been found association between
MetS, age of patients (OR=1.13; IC [1.05-1.23]; P=0.001) and their
mental health (OR= 1.03; IC [1.00- 1.07]; P= 0.04). We did not found
any association with disease duration, disease activity or severity, ESR
or CRP, taken disease-modifying anti-rheumatic drugs (DMARDS)
or Anti inflammatory drugs (P>0.05). There was no association
between MetS and total physical activity of patients and depression
or anxiety (P>0.05).
In a multivariate logistic regression model it persists association
between MetS and the age of patients with AS (OR= 1.12; IC [1.04-
1.21]; P= 0.003).
Discussion
In this cross-sectional observational study with case control where
we investigated the frequency of MetS in patients with AS, we have
observed the rate of MetS in the AS patients higher than the control
group, but its prevalence depends on the definition used (8.2% to
13.6% in AS group while from 1% to 5% in control group). It was
found a significant relationship between MetS and the age of patients
with AS but no significant relationship in term of disease duration
and functional, clinical activity, inflammation or taken drugs.
The prevalence of the metabolic syndrome has varied markedly
between different studies. Recently, Batmaz et al. in their study
including 50 AS patients and 44 controls found a prevalence of MetS
(defined with NCEP ATP III criteria) 12% in AS group against 4.5%
in control groups but it was statistically no significant (P>0.05) [16].
In the study conducted by Malesci et al. including 24 patients with AS
and 19 controls, the prevalence of metabolic syndrome, according to
the NCEP/ATPIII criteria, was found to be considerably higher than
that seen in the controls (45.8% vs. 10.5%) [15]. In another study,
involving 63 patients with spondylitis receiving anti-TNF therapy and
126 controls, the prevalence of metabolic syndrome (NCEP/ATPIII
criteria) was also higher among the patients than among the controls
(34.9% vs. 19%) [35]. In our study the prevalence of MetS according
to NCEP ATP III criteria was 8.2% in AS and 1% in control groups.
These results may be comparable to the study of Malesci despite the
small sample.
The factors found in this study to be independently associated
with the metabolic syndrome in AS, irrespective of the definition
used included older age, but there was no association with the disease
duration, disease activity or inflammation measured by ESR and C
reactive protein. The association with older age is not surprising,
because in the general population the MetS has been shown to affect
primarily older subjects, as a consequence of age-related modification
of some of its components [36]. Allowing comparison of our results
with those of other studies in AS and other conditions, in the literature,
Papadakis et al. have also observed that the AS patients with MetS are
older than the AS patients without MetS; their disease duration was
longer and they had higher BASDAI scores and cardiovascular risks
[14]. In the study of Batmaz et al. they did not observe any statistically
significant relationship between the presence of MetS and the disease
duration, the BASDAI and the BASFI values [16]. In Malesci study
the investigators have not detected any statistically significant
relationship between the presence of MS in the AS patients and their
ages, disease duration and BASDAI and BASFI results [15].
This study has several strengths. These include the use of all of the
existing MetS criteria for the first time in Morocco patients with AS,
it includes case-control. Despite this the most prominent limitation
of our study was the cross-sectional design and selection bias cause
Tertiary center that recruits the most severe forms of AS and this do
not reflect the reality of AS rheumatism in Morocco. Further and
wider ranging clinical studies are thus needed in order to evaluate the
presence of MetS in patients with AS.
Conclusion
In summary this study shows that the ankylosing spondylitis
has been associated with increased prevalence of MetS. Older age
was independent predictor associated with the presence of MetS in
patients with AS. These findings suggest that clinicians should screen
for MetS in patients with early AS to control its components and,
therefore, reduce their risk of cardiovascular diseases.
References
Citation
El Hassani Sbai S, Rostom S, Amine B, Bahiri R. Prevalence of Metabolic Syndrome in Patients with Ankylosing Spondylitis. J Orthopedics Rheumatol. 2019;
6(1): 5.