Journal of Nutrition & Health
Download PDF
No formal sample size calculation was performed, as this was an exploratory consumer-use study intended to generate preliminary descriptive real-world data on tolerability, acceptability, and self reported symptom evolution. A sample of 40 participants was considered appropriate for the exploratory objectives of the study. The study population consisted of American women aged 20 to 45 years (mean age 33), with a balanced distribution between the 20- 33 and 34-45 age groups. Inclusion required a baseline PMS-related discomfort score ≥ 4/10 and a baseline PMS-related pain score ≥ 3/10 on a VAS, as well as written informed consent prior to participation.
Women who were pregnant or breastfeeding, who had given birth within the previous year, who reported endometriosis, who were perimenopausal or menopausal, or who has used PMS-targeted products (dietary supplements, medications, antispasmodics, antiinflammatory, etc) within 30 days prior to baseline assessment were not eligible.
At Q2, a high proportion of participants reported improvement versus baseline for several physical outcomes, including water retention (100%), digestive discomfort (100%), physical fatigue (95%), menstrual pain (83%), and breast discomfort (60%). This pattern suggests perceived improvement across multiple somatic symptoms [Figure 4].
At baseline, mean scores for psychological symptoms ranged from 6.9 to 8.0 out of 10, indicating a moderate-to-high self-reported symptom burden.
After the first supplemented cycle (Q1), mean scores were lower than baseline across all psychological items, suggesting an early favorable trend. After two supplemented cycles (Q2), mean scores decreased further, ranging from 5.3 to 6.6 out of 10. The largest relative reductions were observed for emotional imbalance (-32.1%), irritability (-30.0%), sensitivity (-25.6%), and nervousness (-23.7%).
Most participants reported a positive overall experience with the supplement, together with favorable perceptions of usability and acceptability. These findings are presented as supportive secondary observations rather than efficacy outcomes. Notably, 90% of participants reported a rapid onset of action, with noticeable benefits perceived within an average of four days of use.
Research Article
Effects of a Standardized Saffron Extract on Premenstrual Symptoms: An Open-Label, Exploratory, Real-World Consumer Study
Emmanuelle Arnaud*, Ilona Gille, and Maïté Jeanroy
Activ’Inside, Beychac-et-Caillau, France
*Address for Correspondence:Emmanuelle Arnaud, Activ’Inside, Beychac-et-Caillau, France,
Email: e.arnaud@activinside.com
Submission: 14 April, 2026
Accepted: 05 May, 2026
Published: 08 May, 2026
Copyright: ©2026 Arnaud E, et al. This is an open access
article distributed under the Creative Commons Attribution
License, which permits unrestricted use, distribution, and
reproduction in any medium, provided the original work is
properly cited.
Keywords:Premenstrual syndrome; Women’s health; Crocus sativus; Open study; Saffron extract
Abstract
Background: Premenstrual syndrome is a common cyclical condition characterized by emotional and physical symptoms that may affect daily functioning and quality of life. Because many women rely on analgesics to manage these symptoms, interest is growing in complementary approaches that may help support both psychological-related and physical complaints.
Objective: To assess the tolerability and perceived effects of a patented standardized saffron extract (Safr’Inside™) on premenstrual symptoms under real-life conditions.
Methods: An open-label, exploratory, real-world consumer study with medical oversight was conducted in California in women aged 20-45 years reporting recurrent premenstrual symptoms. Participants consumed one capsule containing 30 mg of a standardized saffron extract during a 10-day perimenstrual window over two consecutive cycles. PMS-related symptoms were assessed at baseline and after each supplemented cycle using an identical structured questionnaire (Q0, Q1 and Q2). The questionnaire captured 16 psychological and physical symptoms rated on a 0-10 Visual Analogue Scale (VAS), as well as treatment use, satisfaction, tolerability, and compliance.
Results: Across both supplemented cycles, Safr’Inside™ use was associated with self-reported improvements in emotional and physical symptoms. Participants reported improvements in both psychological-related outcomes (including quality of life impact, emotional imbalance, irritability, and nervousness) and physical symptoms (including appetite changes, digestive discomfort, physical fatigue, and water retention). Compliance was high and no adverse effects were reported.
Conclusions: In this open-label, exploratory, real-world consumer study, 10-day supplementation with a standardized saffron extract, administered 5 days before the onset of menstruation, was associated with self-reported improvements in both emotional and physical premenstrual symptoms under everyday conditions. These findings should be interpreted cautiously and considered as preliminary complementary real-world evidence.
Objective: To assess the tolerability and perceived effects of a patented standardized saffron extract (Safr’Inside™) on premenstrual symptoms under real-life conditions.
Methods: An open-label, exploratory, real-world consumer study with medical oversight was conducted in California in women aged 20-45 years reporting recurrent premenstrual symptoms. Participants consumed one capsule containing 30 mg of a standardized saffron extract during a 10-day perimenstrual window over two consecutive cycles. PMS-related symptoms were assessed at baseline and after each supplemented cycle using an identical structured questionnaire (Q0, Q1 and Q2). The questionnaire captured 16 psychological and physical symptoms rated on a 0-10 Visual Analogue Scale (VAS), as well as treatment use, satisfaction, tolerability, and compliance.
Results: Across both supplemented cycles, Safr’Inside™ use was associated with self-reported improvements in emotional and physical symptoms. Participants reported improvements in both psychological-related outcomes (including quality of life impact, emotional imbalance, irritability, and nervousness) and physical symptoms (including appetite changes, digestive discomfort, physical fatigue, and water retention). Compliance was high and no adverse effects were reported.
Conclusions: In this open-label, exploratory, real-world consumer study, 10-day supplementation with a standardized saffron extract, administered 5 days before the onset of menstruation, was associated with self-reported improvements in both emotional and physical premenstrual symptoms under everyday conditions. These findings should be interpreted cautiously and considered as preliminary complementary real-world evidence.
List of abbreviations:
EFSA: European Food Safety Authority; 5-HIAA:
5-Hydroxyindoleacetic Acid; PMDD: Premenstrual Dysphoric
Disorder; PMS: Premenstrual Syndrome; SERT: Serotonin
Transporter; SSRI: Selective Serotonin Reuptake Inhibitor; UHPLC:
Ultra-High-Performance Liquid Chromatography; UV: Ultraviolet;
VAS: Visual Analogue Scale.Introduction
Premenstrual syndrome (PMS) is a common cyclical condition
characterized by the recurrence of emotional and physical symptoms
during the late luteal phase of the menstrual cycle, typically resolving
with the onset of menstruation or shortly thereafter
[1]. Symptom onset generally occurs within the 2 to 7 days preceding menses,
although in some women symptoms may begin earlier, up to
approximately 14 days before menstruation. PMS encompasses
a broad spectrum of manifestations that vary in intensity and may
substantially affect daily functioning and quality of life. In its most
severe form, symptoms may meet criteria for premenstrual dysphoric
disorder (PMDD), a condition associated with marked distress and
functional impairment.
Premenstrual symptoms are highly prevalent worldwide. It
has been estimated that 70% to 90% of women of reproductive age
experience at least some premenstrual symptoms, while 20% to 40%
report symptoms sufficiently bothersome to interfere with daily
activities. PMDD is estimated to affect approximately 3% to 8% of
women [1]. In the United States, up to three out of four women
report having experienced PMS symptoms at some point in their
lives, highlighting the broad relevance of this condition in routine
practice [2]. PMS symptomatology is heterogeneous and includes
both psychological and physical manifestations, such as irritability,
emotional lability, anxiety, headaches, abdominal cramps, breast
tenderness, fatigue, digestive discomfort, and changes in appetite or
eating behavior. Consistent with this multidimensional presentation,
an online quantitative study conducted in France in 2022 among
146 women reporting PMS found that 98% experienced physical
symptoms and 97% psychological symptoms, with abdominal pain,
fatigue, emotional swings, and lack of energy among the most
frequently reported complaints [3].
Given the frequency and multidimensional nature of PMS,
symptom management often relies on repeated use of conventional
treatments, particularly analgesics for pain-related symptoms.
However, recurrent use of such medications may raise tolerability
concerns in some women. Self-medication with painkillers for
PMS-related symptoms is common, and survey data indicate
substantial rates of analgesic use among women experiencing
premenstrual discomfort. In parallel, selective serotonin reuptake
inhibitors (SSRIs), such as fluoxetine, have demonstrated efficacy
for both psychological and physical symptoms of PMS and may
act rapidly in this context, sometimes within days. Together, these
observations support the relevance of serotonergic pathways in PMS
and underscore the interest of complementary, well-tolerated, nonpharmacological
approaches that may be suitable for recurrent use
and broader symptom management [4-7].
The pathophysiology of PMS is considered multifactorial, with
converging evidence implicating ovarian hormone fluctuations and
downstream modulation of neurotransmission systems involved
in mood regulation, particularly the serotonergic pathway. PMS is
closely linked to ovulatory cycles and appears to emerge during the
late luteal phase, when progesterone and estrogen levels decline in the
absence of pregnancy [8].
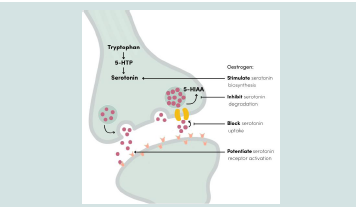
Estrogen has been described as supporting serotonergic neurotransmission through modulation of serotonin synthesis, receptor signaling, reuptake, and degradation. Consequently, the late-luteal decline in estrogen may contribute to serotonergic vulnerability and to the emergence of emotional symptoms such as irritability, anxiety, emotional instability, and sleep disturbances [Figure 1] [9]. This mechanistic framework is further supported by observations in PMDD, where increased serotonin transporter (SERT) activity near menstruation has been proposed as one contributor to reduced synaptic serotonin availability and symptom exacerbation. Beyond mood regulation, serotonin also contributes to pain modulation, suggesting that altered serotonergic signaling may be involved not only in the emotional manifestations of PMS, but also in its physical symptom dimension. In addition to hormonal and
Estrogen has been described as supporting serotonergic neurotransmission through modulation of serotonin synthesis, receptor signaling, reuptake, and degradation. Consequently, the late-luteal decline in estrogen may contribute to serotonergic vulnerability and to the emergence of emotional symptoms such as irritability, anxiety, emotional instability, and sleep disturbances [Figure 1] [9]. This mechanistic framework is further supported by observations in PMDD, where increased serotonin transporter (SERT) activity near menstruation has been proposed as one contributor to reduced synaptic serotonin availability and symptom exacerbation. Beyond mood regulation, serotonin also contributes to pain modulation, suggesting that altered serotonergic signaling may be involved not only in the emotional manifestations of PMS, but also in its physical symptom dimension. In addition to hormonal and
neurochemical factors, other contributors such as stress, smoking,
and nutritional or dietary factors may further influence symptom
expression, reinforcing the multifactorial nature of PMS and the need
for multidimensional management strategies [10,11].
In this context, saffron (Crocus sativus L.) has attracted increasing scientific interest as a potential nutraceutical approach for emotion and stress-related conditions. Its main bioactive compounds, notably crocins and safranal, have been associated with effects on neurotransmission pathways involved in emotional regulation, including possible interactions with the serotonergic system, as well as antioxidant and anti-inflammatory activities. This multimodal profile is of interest in PMS, where psychological and physical symptoms coexist [12-14].
In this context, saffron (Crocus sativus L.) has attracted increasing scientific interest as a potential nutraceutical approach for emotion and stress-related conditions. Its main bioactive compounds, notably crocins and safranal, have been associated with effects on neurotransmission pathways involved in emotional regulation, including possible interactions with the serotonergic system, as well as antioxidant and anti-inflammatory activities. This multimodal profile is of interest in PMS, where psychological and physical symptoms coexist [12-14].
Several randomized, double-blind, placebo-controlled studies
have reported improvements in both psychological and physical
symptoms following saffron supplementation. In one trial including 50
women aged 20-45 years, supplementation with 30 mg/day of saffron
extract (2% safranal, UV) for two months significantly improved
both physical and emotional symptoms from the first month onward.
After two months, PMS severity was reduced by at least 50% in 76%
of supplemented participants, and depressive symptoms decreased
by at least 50% in 60% [15]. A second randomized, double-blind,
placebo-controlled trial in 78 women aged 18-35 years using the same
daily dose for two months also reported significant improvements
in PMS severity and symptom dimensions compared with placebo
[16]. Finally, a third randomized, double-blind, placebo-controlled
study investigated saffron’s volatile fraction in 35 women aged 19-
26 years. Participants were exposed to saffron stigma oil odor for
20 minutes, resulting in a significant reduction in salivary cortisol.
These findings support a possible role of saffron volatiles, largely
attributed to safranal, in stress-related and emotional outcomes
[17]. Mechanistically, safranal has been described as a potential
modulator of serotonin reuptake, while crocin may interact with
other monoaminergic pathways, providing biological plausibility for
the observed clinical effects [18]. Furthermore, ex vivo data suggest
that Safr’Inside™ may modulate serotonin-related pathways under
stress-related conditions [19].
Although these controlled studies support the potential of saffron for the management of premenstrual symptoms, additional data obtained with a well-characterized and standardized saffron extract may help further document its use in this setting. Complementary evidence generated under real-world conditions may also be informative, particularly regarding adherence, tolerability, perceived symptom evolution, and acceptability in routine use. Such data are of interest given that PMS is commonly managed through everyday self-care strategies and that dietary supplement use is widespread, particularly among women in the United States [2].
The aim of this open-label, exploratory, real-world consumer study with medical oversight was therefore to evaluate the tolerability and self-reported effects of Safr’Inside™, a standardized saffron extract (0.2% safranal, 3% crocins, analyzed by HPLC, and 12% safromotivines analyzed by UV), in women experiencing recurrent premenstrual symptoms, by documenting changes in psychological and physical symptom dimensions across two consecutive supplemented menstrual cycles under real-life conditions.
Although these controlled studies support the potential of saffron for the management of premenstrual symptoms, additional data obtained with a well-characterized and standardized saffron extract may help further document its use in this setting. Complementary evidence generated under real-world conditions may also be informative, particularly regarding adherence, tolerability, perceived symptom evolution, and acceptability in routine use. Such data are of interest given that PMS is commonly managed through everyday self-care strategies and that dietary supplement use is widespread, particularly among women in the United States [2].
The aim of this open-label, exploratory, real-world consumer study with medical oversight was therefore to evaluate the tolerability and self-reported effects of Safr’Inside™, a standardized saffron extract (0.2% safranal, 3% crocins, analyzed by HPLC, and 12% safromotivines analyzed by UV), in women experiencing recurrent premenstrual symptoms, by documenting changes in psychological and physical symptom dimensions across two consecutive supplemented menstrual cycles under real-life conditions.
Materials and methods
Study design and participants:
This open-label, exploratory, real-world consumer study with
medical oversight was conducted in California (USA) in 40 women
reporting premenstrual symptoms. The study aimed to evaluate the
tolerability and self-reported effects of a standardized saffron extract
consumed during a targeted 10-day perimenstrual window over two
consecutive menstrual cycles.No formal sample size calculation was performed, as this was an exploratory consumer-use study intended to generate preliminary descriptive real-world data on tolerability, acceptability, and self reported symptom evolution. A sample of 40 participants was considered appropriate for the exploratory objectives of the study. The study population consisted of American women aged 20 to 45 years (mean age 33), with a balanced distribution between the 20- 33 and 34-45 age groups. Inclusion required a baseline PMS-related discomfort score ≥ 4/10 and a baseline PMS-related pain score ≥ 3/10 on a VAS, as well as written informed consent prior to participation.
Women who were pregnant or breastfeeding, who had given birth within the previous year, who reported endometriosis, who were perimenopausal or menopausal, or who has used PMS-targeted products (dietary supplements, medications, antispasmodics, antiinflammatory, etc) within 30 days prior to baseline assessment were not eligible.
Intervention:
The intervention consisted of one daily capsule providing 30
mg of Safr’Inside™, a full-spectrum standardized saffron extract
(Crocus sativus L.) developed by Activ’Inside (Beychac-et-Caillau,
France). This saffron extract is produced according to patent
WO2018/020013A1 and is standardized to contain >3% crocins
and >0.2% safranal, quantified by UHPLC. Participants consumed
the supplement over two consecutive menstrual cycles, during a
10-day peri-menstrual period in each cycle, from five days before
menstruation onset to five days after menstruation onset
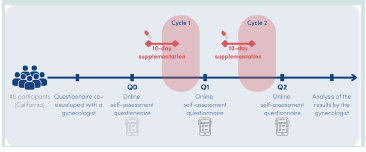
[Figure 2].Evaluation protocol:
PMS-related symptoms were assessed using a structured online
questionnaire developed by a gynecologist and intended as a
pragmatic, non-validated tool to capture the consumer experience
under real-world conditions. Evaluations were performed atthree time points: baseline (Q0, before supplementation), after
the first supplemented menstrual cycle (Q1), and after the second
supplemented cycle (Q2) [Figure 2].
The same questionnaire was administered at Q0, Q1, and Q2. It was designed to capture the most common and clinically relevant manifestations of PMS encountered in routine gynecological practice. It included items related to symptom-management behaviors, as well as psychological and physical symptom domains.
At each time point, participants rated 16 PMS-related symptoms on a 0-10 Visual Analogue Scale (VAS), where 0 corresponded to the absence of the symptom and 10 to the highest perceived intensity/ frequency depending on the item. The 16 items were: impact of PMS on quality of life, stress, nervousness, emotional imbalance, sensitivity, irritability, lack of enthusiasm, menstrual pain, appetite changes, physical fatigue, breast discomfort, muscle discomfort, digestive discomfort, weight gain, water retention, and headache. No global score was calculated. Improvement was defined as a lower score compared with Q0.
All questionnaire data were reviewed and interpreted by the medical assessor.
The baseline questionnaire (Q0) was sent on Day 0 (D0) and asked participants to retrospectively report PMS symptoms experienced during unsupplemented menstrual cycles preceding study entry. As a result, Q0 reflects recalled pre-intervention symptom experience rather than prospectively recorded baseline data. Responses were collected again after the first supplemented cycle (Q1) and after the second supplemented cycle (Q2), allowing within-subject comparison over time.
The same questionnaire was administered at Q0, Q1, and Q2. It was designed to capture the most common and clinically relevant manifestations of PMS encountered in routine gynecological practice. It included items related to symptom-management behaviors, as well as psychological and physical symptom domains.
At each time point, participants rated 16 PMS-related symptoms on a 0-10 Visual Analogue Scale (VAS), where 0 corresponded to the absence of the symptom and 10 to the highest perceived intensity/ frequency depending on the item. The 16 items were: impact of PMS on quality of life, stress, nervousness, emotional imbalance, sensitivity, irritability, lack of enthusiasm, menstrual pain, appetite changes, physical fatigue, breast discomfort, muscle discomfort, digestive discomfort, weight gain, water retention, and headache. No global score was calculated. Improvement was defined as a lower score compared with Q0.
All questionnaire data were reviewed and interpreted by the medical assessor.
The baseline questionnaire (Q0) was sent on Day 0 (D0) and asked participants to retrospectively report PMS symptoms experienced during unsupplemented menstrual cycles preceding study entry. As a result, Q0 reflects recalled pre-intervention symptom experience rather than prospectively recorded baseline data. Responses were collected again after the first supplemented cycle (Q1) and after the second supplemented cycle (Q2), allowing within-subject comparison over time.
Statistical analysis:
No formal statistical analysis was performed. Only descriptive
summary measures were planned for this exploratory study. No
inferential analyses were performed. Measures of dispersion were
not available for reporting in the present manuscript, which limits
the reader’s ability to assess inter-individual variability in symptom
ratings. Given the exploratory and descriptive nature of this openlabel
real-world study, results were summarized using mean scores,
percentage changes versus baseline, and proportions of participants
reporting improvement. No hypothesis-testing framework was prespecified,
and no imputation procedure was applied for missing data.Satisfaction, tolerability and compliance:
At the end of the study, participants completed an acceptability
questionnaire regarding perceived changes in their menstrual cycles
and premenstrual symptoms and reported on ease of use, tolerability,
and compliance.Results
Psychological outcomes:
Participants rated psychological symptoms on a 0-10 Visual
Analogue Scale (VAS) at Q0, Q1, and Q2. Mean ratings and
percentage changes versus baseline are presented in [Table 1].
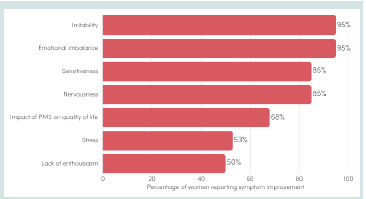
The proportion of participants reporting improvement versus
baseline at Q2 was high for several psychological outcomes, including
emotional imbalance (95%), irritability (95%), nervousness (85%),
and sensitiveness (85%). These findings suggest a consistent pattern
of perceived improvement across psychological domains [Figure 3].Physical outcomes:
Using the same 0-10 scale at Q0, Q1, and Q2, participants also
rated physical symptoms. Mean ratings and percentage changes
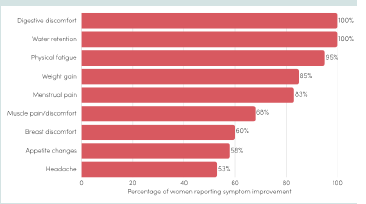
versus baseline are presented in [Table 2].At Q2, a high proportion of participants reported improvement versus baseline for several physical outcomes, including water retention (100%), digestive discomfort (100%), physical fatigue (95%), menstrual pain (83%), and breast discomfort (60%). This pattern suggests perceived improvement across multiple somatic symptoms [Figure 4].
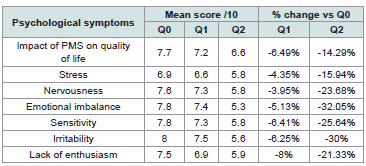
At baseline, mean scores for psychological symptoms ranged from 6.9 to 8.0 out of 10, indicating a moderate-to-high self-reported symptom burden.
After the first supplemented cycle (Q1), mean scores were lower than baseline across all psychological items, suggesting an early favorable trend. After two supplemented cycles (Q2), mean scores decreased further, ranging from 5.3 to 6.6 out of 10. The largest relative reductions were observed for emotional imbalance (-32.1%), irritability (-30.0%), sensitivity (-25.6%), and nervousness (-23.7%).
Table 1:Self-reported ratings of psychological symptoms across the study period
and percentage change vs. baseline.
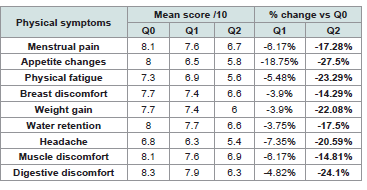
Table 2:Self-reported ratings of physical symptoms across the study period and
percentage change vs. baseline.
Figure 3:Proportion of participants reporting improvement in emotional
symptoms of PMS after two supplemented cycles vs. baseline
Figure 4:Proportion of participants reporting improvement in physical
symptoms of PMS after two supplemented cycles vs. baseline
At baseline, mean scores for physical symptoms ranged from 6.8
to 8.3 out of 10, indicating a high self-reported symptom burden.
After the first supplemented cycle (Q1), mean scores were lower
than baseline for all physical items, suggesting an early favorable
trend. After two supplemented cycles (Q2), mean scores ranged from
5.4 to 6.9 out of 10. The largest relative reductions were observed for
appetite changes (-27.5%), digestive discomfort (-24.1%), physical
fatigue (-23.3%), weight gain (-22.1%), and headache (-20.6%).
Participant satisfaction and acceptability:
In addition to symptom-related outcomes, qualitative questions
were used to document participant satisfaction, perceived usability,
and acceptability of Safr’Inside™ under real-world conditions.Most participants reported a positive overall experience with the supplement, together with favorable perceptions of usability and acceptability. These findings are presented as supportive secondary observations rather than efficacy outcomes. Notably, 90% of participants reported a rapid onset of action, with noticeable benefits perceived within an average of four days of use.
Tolerability and compliance:
Compliance was high throughout both intervention cycles: 95%
of participants reported taking the supplement in full without missing
any doses, while 5% reported missing 2 to 3 doses. No side effects
were reported during the study, supporting the product’s safety and
tolerability.Discussion
This open-label, exploratory, real-world consumer study with
medical oversight conducted over two consecutive menstrual cycles
suggests that daily supplementation with a standardized saffron extract
(Safr’Inside™), administered during a targeted 10-day perimenstrual
window, was associated with self-reported improvements in both
psychological and physical premenstrual symptoms under everyday
conditions.
These observations are directionally consistent with previous randomized, placebo-controlled clinical studies in women with PMS, in which oral supplementation with 30 mg/day of saffron extract administered continuously over two menstrual cycles was associated with improvements in both psychological and physical symptoms. However, direct comparison should remain cautious given the important differences in study design, population, assessment tools, and supplementation regimen.
From a biological perspective, these self-reported improvements are compatible with current understanding of PMS pathophysiology and with the known properties of saffron bioactives. PMS has been linked to cyclical changes in ovarian hormone levels, which may influence serotonergic neurotransmission and thereby contribute to emotional symptoms such as irritability, mood instability, stress sensitivity, and nervousness.
Additional mechanistic plausibility comes from an ex vivo clinical approach performed with Safr’Inside™, in which serum collected after intake of the extract attenuated stress-induced changes in serotonin-related markers in a human neuron culture model, including serotonin release, serotonin transporter expression, and 5-HIAA levels [19]. While such ex vivo findings cannot be directly extrapolated to clinical efficacy, they support the hypothesis that circulating metabolites derived from Safr’Inside™ may help preserve serotonergic tone under stress-related conditions. In the context of PMS, where altered serotonergic regulation and stress reactivity have both been implicated in emotional symptom expression, these findings are biologically consistent with the improvements reported here, particularly in psychological symptom domains. This serotonergic hypothesis may also be relevant, at least in part, to the physical symptom improvements observed in the present study. Beyond its role in mood regulation, serotonin is also involved in nociceptive processing, with effects that vary according to receptor subtype, anatomical level, and pain context [10]. Because saffron has been proposed to influence serotonergic signaling, including pathways related to serotonin reuptake, it is plausible that some of the observed improvements in pain-related symptoms may reflect, indirectly, modulation of serotonin-related mechanisms. However, this interpretation remains hypothetical and was not specifically assessed in the present study.
This study was specifically designed to evaluate outcomes under real-life conditions, with minimal disruption to participants’ usual dietary and lifestyle habits. Such an approach enhances ecological validity and provides insight into how supplementation may be perceived and used in everyday settings. It should not, however, be considered as providing the same level of evidence as a randomized controlled trial.
Several important methodological limitations should nevertheless be acknowledged. First, the study used an open-label, non-controlled design, and therefore expectancy effects and natural cycle-to-cycle symptom variability may have contributed to the observed changes. Second, the sample size was limited. Third, the baseline assessment (Q0) was retrospective, whereas follow-up assessments referred to prospectively experienced supplemented cycles; therefore, recall bias may have inflated baseline symptom burden and amplified the apparent magnitude of change. Fourth, symptom evaluation relied on a pragmatic questionnaire that was not formally validated, although it was developed by a gynecologist to reflect common and clinically relevant PMS manifestations encountered in practice. Finally, no formal statistical analysis was performed in this exploratory study. Results are therefore presented descriptively as mean scores, percentage changes versus baseline, and proportions of participants reporting improvement. Accordingly, the findings should be interpreted as preliminary real-world observations rather than confirmatory evidence.
These observations are directionally consistent with previous randomized, placebo-controlled clinical studies in women with PMS, in which oral supplementation with 30 mg/day of saffron extract administered continuously over two menstrual cycles was associated with improvements in both psychological and physical symptoms. However, direct comparison should remain cautious given the important differences in study design, population, assessment tools, and supplementation regimen.
From a biological perspective, these self-reported improvements are compatible with current understanding of PMS pathophysiology and with the known properties of saffron bioactives. PMS has been linked to cyclical changes in ovarian hormone levels, which may influence serotonergic neurotransmission and thereby contribute to emotional symptoms such as irritability, mood instability, stress sensitivity, and nervousness.
Additional mechanistic plausibility comes from an ex vivo clinical approach performed with Safr’Inside™, in which serum collected after intake of the extract attenuated stress-induced changes in serotonin-related markers in a human neuron culture model, including serotonin release, serotonin transporter expression, and 5-HIAA levels [19]. While such ex vivo findings cannot be directly extrapolated to clinical efficacy, they support the hypothesis that circulating metabolites derived from Safr’Inside™ may help preserve serotonergic tone under stress-related conditions. In the context of PMS, where altered serotonergic regulation and stress reactivity have both been implicated in emotional symptom expression, these findings are biologically consistent with the improvements reported here, particularly in psychological symptom domains. This serotonergic hypothesis may also be relevant, at least in part, to the physical symptom improvements observed in the present study. Beyond its role in mood regulation, serotonin is also involved in nociceptive processing, with effects that vary according to receptor subtype, anatomical level, and pain context [10]. Because saffron has been proposed to influence serotonergic signaling, including pathways related to serotonin reuptake, it is plausible that some of the observed improvements in pain-related symptoms may reflect, indirectly, modulation of serotonin-related mechanisms. However, this interpretation remains hypothetical and was not specifically assessed in the present study.
This study was specifically designed to evaluate outcomes under real-life conditions, with minimal disruption to participants’ usual dietary and lifestyle habits. Such an approach enhances ecological validity and provides insight into how supplementation may be perceived and used in everyday settings. It should not, however, be considered as providing the same level of evidence as a randomized controlled trial.
Several important methodological limitations should nevertheless be acknowledged. First, the study used an open-label, non-controlled design, and therefore expectancy effects and natural cycle-to-cycle symptom variability may have contributed to the observed changes. Second, the sample size was limited. Third, the baseline assessment (Q0) was retrospective, whereas follow-up assessments referred to prospectively experienced supplemented cycles; therefore, recall bias may have inflated baseline symptom burden and amplified the apparent magnitude of change. Fourth, symptom evaluation relied on a pragmatic questionnaire that was not formally validated, although it was developed by a gynecologist to reflect common and clinically relevant PMS manifestations encountered in practice. Finally, no formal statistical analysis was performed in this exploratory study. Results are therefore presented descriptively as mean scores, percentage changes versus baseline, and proportions of participants reporting improvement. Accordingly, the findings should be interpreted as preliminary real-world observations rather than confirmatory evidence.
Conclusion
In conclusion, this open-label, exploratory, real-world consumer
study conducted in California suggests that supplementation with
Safr’Inside™, a standardized saffron extract, taken once daily during
a targeted 10-day perimenstrual window over two consecutive cycles,
was associated with self-reported improvements in psychological and
physical premenstrual symptoms under everyday conditions.
Because baseline symptoms were retrospectively recalled and no
control group was included, the magnitude of the observed changes
should be interpreted cautiously.
Overall, these findings provide complementary preliminary real-world evidence that is directionally consistent with previously published randomized, placebo-controlled trials of saffron supplementation, while remaining distinct in design and evidentiary strength.
Future studies should confirm these observations in randomized, double-blind, placebo-controlled designs using validated PMS instruments and prospective symptom tracking across multiple cycles.
Overall, these findings provide complementary preliminary real-world evidence that is directionally consistent with previously published randomized, placebo-controlled trials of saffron supplementation, while remaining distinct in design and evidentiary strength.
Future studies should confirm these observations in randomized, double-blind, placebo-controlled designs using validated PMS instruments and prospective symptom tracking across multiple cycles.
Acknowledgements
The authors thank the participants for their time and cooperation.
Funding:
This study was funded by Activ’Inside (Beychac-et-Caillau,
France), the developer of Safr’Inside™. The sponsor provided the
study material and covered operational costs but had no role in data
collection, statistical analysis or interpretation of the results.Conflicts of Interest:
All authors are full-time employees of Activ’Inside, the company
that developed the product tested in this study. This affiliation did
not influence the objectivity of the research or the interpretation of
the results.Ethics approval and consent to participate:
This interventional study was based on the voluntary use of a food
supplement for 10 days during two menstrual cycles. All participants
provided informed consent before inclusion. In accordance with the
non-pharmacological and real-world nature of the study, no formal
ethics committee approval was obtained.Availability of data and materials:
All relevant data supporting the conclusions of this study are
presented in this article.Authors’ contributions:
E.A. and I.G. contributed to the development of the study protocol,
supervised the study, performed data analysis and interpretation, and
were involved in manuscript writing. M.J. contributed to its critical
revision, and approved the final version for publication.References
Citation
Arnaud E, Gille I, Jeanroy M. Effects of a Standardized Saffron Extract on Premenstrual Symptoms: An Open-Label, Exploratory, Real-World Consumer Study. J Nutri Health. 2026;9(1): 01.