Journal of Nutrition & Health
Download PDF
Eligible participants were Chinese children aged 7 to 12 years, with a balanced ratio between the 7-9 and 10-12 age groups and an equal gender distribution. Children had to be declared healthy by their parents or legal guardians, with no diagnosis of ADHD, autism, or other neurological disorders, and no food allergies or dietary restrictions. They could not have participated in any market research activity in the past three months. Both children and parents had to agree to comply with the study instructions, including daily consumption of a fruit-flavored stick supplement at breakfast for 28 days, and completion of cognitive and satisfaction questionnaires at specified timepoints. Children were also required to complete online cognitive mini-games (Cognifit Kids platform) at four key timepoints under standardized conditions at home.
Exclusion criteria included any diagnosed neurological condition such as ADHD, autism, or learning disabilities, as well as chronic disease, food allergy, ongoing medication or food supplement use, and participation in another clinical or open trial within the previous three months. Refusal to comply with study instructions or the product use protocol also led to exclusion.
The high adherence rate, coupled with positive parental feedback, reinforces the acceptability of Memophenol™ in real-world conditions and supports its feasibility for long- term use in children.
Conflict of Interest: All authors are full-time employees of Activ’Inside. This affiliation did not influence the design, analysis, or interpretation of the study results.
Research Article
Evaluation of Acute and Sustained Cognitive Effects of a Grape and Blueberry Polyphenol-Rich Extract in Healthy 7-12 Years Old Children: A Home-Based, Real-World Consumer Study
Ilona Gille, Emmanuelle Arnaud*, Maïté Jeanroy, Benoit Lemaire, David Gaudout
Activ’Inside, Beychac-et-Caillau, France
*Address for Correspondence:Emmanuelle Arnaud, Activ’Inside, Beychac-et-Caillau, France,
Email: e.arnaud@activinside.com
Submission: 03 February, 2026
Accepted:10 March, 2026
Published: 14 March, 2026
Copyright: © 2026 Gille I, et al. This is an open access article
distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction
in any medium, provided the original work is properly cited.
Keywords:Cognition; Executive function; Polyphenols; Children; Open study; Memophenol™
Abstract
Background: Executive functions such as attention, working memory, and emotional regulation undergo rapid development during school age, a period marked by increasing academic demands. This creates a specific need to support cognitive efficiency in children, whose regulatory capacities are still maturing. Polyphenol-rich nutritional interventions have previously shown benefits on executive processes in adults, but evidence
in younger children remains limited, justifying investigation in this age group.
Objectives: This open trial aimed to evaluate both the acute and chronic effects of daily supplementation with Memophenol™ on cognitive performances in children under real- world conditions.
Methods: A 28-day medically supervised real-world open trial was conducted in 41 healthy children (aged 7–12 years) from Singapore. Participants daily consumed one red-berry flavored stick powder containing a polyphenols-rich extract from grape and blueberry (150 mg of Memophenol™), each morning with breakfast. Children’s cognitive performance was assessed using the CogniFit Kids digital platform at four points (D0, D0+2h, D14, D28). Parents assessed their child’s behavioral and executive functions using an adapted version of the validated BRIEF-2 questionnaire (D0, D14, D28), followed by a satisfaction survey at D28.
Results: Children’s cognitive performance improved significantly by +9.5% 2 hours after Memophenol™ intake compared to baseline, as well as by +13.9% after 14 days, and +17.1% after 28 days of supplementation (p < 0.001). Executive function, as assessed by the BRIEF-2 Global Executive Composite (GEC), decreased by –8.3% and –12.9%, respectively after 14 and 28 days of supplementation (p < 0.001), reflecting better overall executive control. In addition, improvements in the Behavior Regulation (–11.5%), Emotion Regulation (–14.4%), and Cognitive Regulation (–12.8%) indices were observed after 28 days. Parents reported noticeable improvements in attention (63%) and overall efficacy (66%). The formulation was well tolerated.
Conclusions: These preliminary findings suggest that daily intake of 150 mg Memophenol™ may improve cognitive functions in children, with both acute and cumulative benefits after 28 days, observed in a real-world setting. The formulation demonstrated good safety and acceptability, supporting its potential as a natural strategy to enhance cognitive functions and behavioral regulation in school-aged children. These results highlight the potential of polyphenol supplementation to support learning performance and behavioral regulation in children under high academic demand. However, given the absence of a control group and the potential for expectancy and learning effects, these results should be interpreted with caution. Further randomized, controlled, and blinded studies are warranted to confirm these promising effects.
Objectives: This open trial aimed to evaluate both the acute and chronic effects of daily supplementation with Memophenol™ on cognitive performances in children under real- world conditions.
Methods: A 28-day medically supervised real-world open trial was conducted in 41 healthy children (aged 7–12 years) from Singapore. Participants daily consumed one red-berry flavored stick powder containing a polyphenols-rich extract from grape and blueberry (150 mg of Memophenol™), each morning with breakfast. Children’s cognitive performance was assessed using the CogniFit Kids digital platform at four points (D0, D0+2h, D14, D28). Parents assessed their child’s behavioral and executive functions using an adapted version of the validated BRIEF-2 questionnaire (D0, D14, D28), followed by a satisfaction survey at D28.
Results: Children’s cognitive performance improved significantly by +9.5% 2 hours after Memophenol™ intake compared to baseline, as well as by +13.9% after 14 days, and +17.1% after 28 days of supplementation (p < 0.001). Executive function, as assessed by the BRIEF-2 Global Executive Composite (GEC), decreased by –8.3% and –12.9%, respectively after 14 and 28 days of supplementation (p < 0.001), reflecting better overall executive control. In addition, improvements in the Behavior Regulation (–11.5%), Emotion Regulation (–14.4%), and Cognitive Regulation (–12.8%) indices were observed after 28 days. Parents reported noticeable improvements in attention (63%) and overall efficacy (66%). The formulation was well tolerated.
Conclusions: These preliminary findings suggest that daily intake of 150 mg Memophenol™ may improve cognitive functions in children, with both acute and cumulative benefits after 28 days, observed in a real-world setting. The formulation demonstrated good safety and acceptability, supporting its potential as a natural strategy to enhance cognitive functions and behavioral regulation in school-aged children. These results highlight the potential of polyphenol supplementation to support learning performance and behavioral regulation in children under high academic demand. However, given the absence of a control group and the potential for expectancy and learning effects, these results should be interpreted with caution. Further randomized, controlled, and blinded studies are warranted to confirm these promising effects.
Introduction
The school-age period is marked by rapid neurodevelopment and
the maturation of core executive functions such as attention, working
memory, reasoning, and emotional regulation. These cognitive
processes are essential for learning efficiency, behavioral selfregulation,
and adaptation to increasingly demanding educational
environments [1,2].
In Asia, academic pressure has become a significant societal concern. Students from Singapore and Japan have high expectations of their own academic achievement but perceive that they are unable to attain those standards. In addition to academic stress arising from self-expectations, Asian adolescents also experience academic stress arising from the need to excel academically to fulfill parental expectations and to avoid the loss of face. [3]. Alarmingly, recent data show that self-harm behaviors are increasingly common among young people in Singapore, with one in four youths reporting having engaged in non-suicidal self-injury (NSSI) at least once [4]. Media have also reported extreme cases, such as a child being hospitalized after spending 14 consecutive hours on homework [5].
In Asia, academic pressure has become a significant societal concern. Students from Singapore and Japan have high expectations of their own academic achievement but perceive that they are unable to attain those standards. In addition to academic stress arising from self-expectations, Asian adolescents also experience academic stress arising from the need to excel academically to fulfill parental expectations and to avoid the loss of face. [3]. Alarmingly, recent data show that self-harm behaviors are increasingly common among young people in Singapore, with one in four youths reporting having engaged in non-suicidal self-injury (NSSI) at least once [4]. Media have also reported extreme cases, such as a child being hospitalized after spending 14 consecutive hours on homework [5].
This intense pressure drives many parents to seek solutions to
support their children’s cognitive performance and emotional wellbeing.
In Singapore, a substantial proportion of families invest in
academic enrichment resources such as private tuition, reflecting the
strong societal emphasis placed on scholastic achievement [6]. Some
parents also turn to digital cognitive training tools or nutritional
supplements, although the efficacy and safety of these interventions
remain variable and are often debated in the scientific literature [7,8].
In recent years, polyphenols – naturally occurring compounds
found in fruits, vegetables, tea, and wine – have attracted considerable
interest due to their potential to enhance cognitive performance
and brain health [9]. Beyond their classical antioxidant properties,
polyphenol- rich foods and extracts, such as those derived from
tea, grapes, blueberries, and cocoa, have been suggested to
modulate cognitive performance through multiple complementary
mechanisms. These mechanisms include acute effects on endothelial
function and cerebral blood flow via nitric oxide-related pathways,
as well as longer-term effects on neuroinflammation and/or synaptic
plasticity [10]. Such mechanisms may contribute to improvements in
several cognitive domains, notably memory, attention, and executive
functions, in healthy adults and older individuals. Consistent
with these mechanistic hypotheses, randomized controlled trials
have investigated a range of polyphenol-rich foods and extracts in
relation to cognitive outcomes. Cocoa flavanols are among the most
extensively studied polyphenol sources in human cognition research.
Acute and short-term randomized controlled trials in adults have
suggested an association between the consumption of flavanol-rich
cocoa and changes in neurovascular markers and brain function, as
well as, in some contexts, improvements in cognitive performance.
For example, cocoa flavanols have been associated with increased
task-related brain activation measured by fMRI in healthy young
adults [11] and with enhanced cerebral perfusion in older adults in
a placebo-controlled crossover design [12]. More broadly, reviews
synthesizing intervention trials conclude that cocoa-derived
flavanols represent a polyphenol source with overall favorable
effects on cognition [13]. Within this broader landscape, and of
particular relevance here, randomized controlled trials have shown
that supplementation with flavonoid-rich blueberry or grape extracts
can lead to improvements on certain tests of working memory,
information-processing speed, and/or verbal learning in adults,
although the magnitude and reproducibility of effects vary according
to populations, doses, matrices, and outcome measures [14,15].
In this context, there is a growing interest in evidence-based
nutritional interventions, such as polyphenol-rich extracts, to support
cognitive development in children. Clinical reviews indicate that
several nutritional strategies, may influence cognitive development
and cognitive performance in school-aged children. For instance, the
landmark review by Bryan et al. synthesizes evidence linking specific
nutrients to cognitive development and cognitive performance
in children [16]. While most clinical evidence on polyphenols
and cognition comes from studies conducted in adults and older
individuals, these promising findings have prompted investigations
into whether similar cognitive benefits might be observed in younger
populations, such as children and adolescents. To date, only a limited
number of randomized controlled trials have been conducted in
this population. For example, a study by Whyte et al. assessed the
acute effects of a wild blueberry drink in 7- to 10-year-old children
and reported improvements in memory and attention tasks shortly
after consumption [17]. Similarly, a randomized trial by Barfoot et
al. demonstrated that daily supplementation with blueberry powder
over 4 weeks led to enhanced executive function and verbal memory
in school-aged children [18]. Other study by Whyte et al. showed that
polyphenols from blueberry powder consumption enhance executive
function such as reaction time [19].
While current findings are promising, there is a need for
larger, well-controlled studies to confirm the cognitive benefits of
polyphenol supplementation in children and to better understand
the underlying mechanisms. Our study aims to address these gaps
by evaluating the effects of Memophenol™, a standardized grape
and blueberry polyphenol extract, in a larger cohort of school-aged
children, using robust cognitive endpoints and a longer intervention
period. Memophenol™ is a patented formulation (WO/2017/072219
and WO/2020/104533) developed by Activ’Inside (Beychac et
Caillau, France) that combines standardized grape and wild blueberry
extracts). Clinical studies in both young and older adults have
demonstrated that Memophenol™ supplementation can improve
memory, learning, and executive functions, with measurable effects
observed as early as 90 minutes post-intake and sustained benefits after
six months of continuous use [20-22]. In addition to these cognitive
benefits, preclinical and clinical data indicate that Memophenol™
stimulates neurogenesis, and improves synaptic plasticity, all of
which are key mechanisms underlying cognitive performance
[23-25]. Building on these findings, the present real-world open study
was designed to assess whether these cognitive benefits could also
be observed in a younger population. Specifically, we evaluated the
effects of a 28-day supplementation with 150 mg of Memophenol™ on
cognitive performance and executive function in healthy school-aged
children living in Singapore.
Materials and Methods
Study design and participants:
This real-world open trial was conducted under medical
supervision by Syres-Asia (Singapore), a sensory and consumer
research institute specializing in consumer testing for efficacy and
tolerance, in Singapore between July 6th and August 2nd, 2025.
The study aimed to evaluate the efficacy and tolerance of a dietary
supplement containing Memophenol™ in healthy school-aged
children. Participants were recruited from the SYRES Singapore
database of panelists, using a combination of phone screening and an
online questionnaire to ensure completeness of information.Eligible participants were Chinese children aged 7 to 12 years, with a balanced ratio between the 7-9 and 10-12 age groups and an equal gender distribution. Children had to be declared healthy by their parents or legal guardians, with no diagnosis of ADHD, autism, or other neurological disorders, and no food allergies or dietary restrictions. They could not have participated in any market research activity in the past three months. Both children and parents had to agree to comply with the study instructions, including daily consumption of a fruit-flavored stick supplement at breakfast for 28 days, and completion of cognitive and satisfaction questionnaires at specified timepoints. Children were also required to complete online cognitive mini-games (Cognifit Kids platform) at four key timepoints under standardized conditions at home.
Exclusion criteria included any diagnosed neurological condition such as ADHD, autism, or learning disabilities, as well as chronic disease, food allergy, ongoing medication or food supplement use, and participation in another clinical or open trial within the previous three months. Refusal to comply with study instructions or the product use protocol also led to exclusion.
The intervention period lasted 28 days, during which each child
consumed one red berry- flavored stick containing Memophenol™
dissolved in water at breakfast every day. Assessments were
conducted at four timepoints: baseline (D0, before the first
intake), two hours after the first intake (D0+2h, to assess acute
effects), day 14 (D14), and day 28 (D28). The two-hour post-intake
assessment (D0+2h) was specifically chosen based on published
pharmacokinetic data indicating that the peak plasma concentration
of key polyphenolic compounds, such as flavanol monomers
((−)-epicatechin and (+)-catechin), typically occurs within 1-2 hours
after oral administration [26,27]. This timing was selected to coincide
with the expected maximal bioavailability of the active compounds
and to optimize the detection of any acute cognitive effects. At each
timepoint, children completed online cognitive mini- games using the
Cognifit Kids platform under standardized home conditions, while
parents completed the BRIEF-2 executive function questionnaire at
D0, D14, and D28, as well as a satisfaction questionnaire at D28. All
children were instructed to maintain their usual diet and physical
activity throughout the study, and medical supervision was available
for safety monitoring.
A total of 52 children were enrolled, and 41 completed the study
per protocol and were included in the analysis. The final sample
consisted of 21 girls and 20 boys, with a mean age of 9.5 years,
all of Chinese ethnicity, living in Singapore, with no allergies or
neurological conditions, and regular breakfast habits.
Intervention:
The intervention consisted of a red-berry–flavored powder
stick containing 150 mg of Memophenol™, which is a proprietary
polyphenol-rich extract made from whole grape (Vitis vinifera L.)
and blueberry (Vaccinium spp.) extracts. Each stick was dissolved
in a glass of water and consumed at breakfast every day for a 28-
day period. Memophenol™ contains at least 75% total polyphenols
measured as catechin equivalents, at least 43% total flavonoids
including flavan-3-ols, flavonols, and anthocyanins, at least 20%
flavan-3-ols monomers, at least 22% flavan-3-ols oligomers with a
degree of polymerization less than or equal to four, at least 0.10%
anthocyanins including malvidin-3-glucoside, and at least 300 ppm
stilbenes including resveratrol.The selected dosage of 150 mg was determined based on two main
criteria, the first being published clinical data assessing the efficacy
of polyphenol- and anthocyanin-rich food intake in children, which
indicate that similar amounts are found in dietary interventions using
blueberries, mulberry powder, or polyphenol-rich supplements,
and the second being extrapolation from previous clinical studies
in adults, with the dose adjusted according to body weight and
established safety margins for daily intake. This approach resulted in
a target range of approximately 102 to 164 mg for children aged 7 to
12 years, and the 150 mg dose was chosen to align with both efficacy
data and international safety recommendations.
Children were instructed not to consume polyphenol-rich foods
such as grape, berries, cocoa, and tea during breakfast throughout the
intervention period, and each participant received at home a doypack
containing 30 sticks. According to the Memophenol™ specification
sheet, the product complies with international regulations regarding
contaminants such as heavy metals, polycyclic aromatic hydrocarbons,
and pesticides, as well as microbiological safety.
Cognitive assessment:
Cognitive performance was assessed using the CogniFit Kids
digital platform (CogniFit Inc., Tel Aviv, Israel [28]), a computerized
cognitive testing tool specifically adapted for children. The
assessments were conducted at four time points: at baseline (D0), two
hours after the first intake (D0+2h), and after 14 (D14) and 28 (D28)
days of supplementation. The CogniFit Kids platform was accessed
online from the participants’ homes, under the supervision of a
parent or legal guardian to ensure standardized testing conditions and
minimize distractions. Each cognitive session lasted approximately
15 to 20 minutes and included a battery of six interactive mini-games
designed to evaluate key cognitive domains relevant to school-aged
children. The tasks assessed attention, working memory, processing
speed, cognitive flexibility, inhibition, and reasoning abilities, in
accordance with validated neuropsychological models [29,30].
The platform automatically calculated individual scores for each
cognitive domain, as well as a global cognitive performance index, by
aggregating performance metrics such as accuracy, reaction time, and
task completion. These scores were standardized according to age appropriate
normative data provided by CogniFit, allowing for the monitoring of cognitive changes over the course of the intervention [30,31].Executive functions were evaluated using the standardized and
validated BRIEF-2 (Behavior Rating Inventory of Executive Function,
Second Edition) parental questionnaire [32], which was administered
in an English-adapted version suitable for the Singaporean context.
Parents completed the questionnaire at baseline (D0), day 14 (D14),
and day 28 (D28) of supplementation. The BRIEF-2 consists of
63 items and explores three main executive function domains:
behavioral regulation, emotional regulation, and cognitive regulation,
as perceived in daily life situations. Each item is rated on a threepoint
Likert scale (Never, Sometimes, Often), and the responses
are used to calculate composite scores for each domain as well as
a global executive function score. Higher scores indicate greater
executive dysfunction. The BRIEF-2 is widely used in both clinical
and research settings for the assessment of executive functions in
children and has demonstrated robust psychometric properties [33].
The use of the English version ensured that parents could accurately
and independently report on their child’s executive functioning in a
language they were comfortable with.
Satisfaction, Tolerability and compliance:
At the end of the study, parents completed a satisfaction and
acceptability questionnaire regarding perceived changes in their
children’s attention, concentration, memory, and overall well-being.
In addition to the satisfaction and acceptability questionnaire, a
medical assessor supervised the study to collect any adverse events.Statistical analysis:
Baseline demographic variables (sex, age) were described.
Categorical data were summarized as percentages, while continuous
variables were reported as mean ± standard deviation (SD).
Differences in Cognifit Z-scores (mean) and BRIEF-2 scores (mean)
between baseline (D0) and follow-up (D+2h, D14 and D28) were
analyzed using two-tailed paired Student’s t-tests, and a two-sided
P value < 0.05 was considered statistically significant. Statistical
analyses were performed using Excel (Microsoft Corporation) with
the Analysis ToolPak add-in.Results
Participant characteristics:
The selection of study participants followed a multi-step process,
Children aged 7 to 12 years, of both sexes (male and female), were
eligible for participation and children with a diagnosed neurological
disease were excluded from the study. Initially, 670 potentially eligible
children were identified from the database. After applying inclusion
and exclusion criteria, 52 participants were enrolled in the study.
During the course of the trial, 11 participants were excluded due to
non-compliance with study instructions, resulting in a final sample of
41 children who completed the study.Cognifit tests:
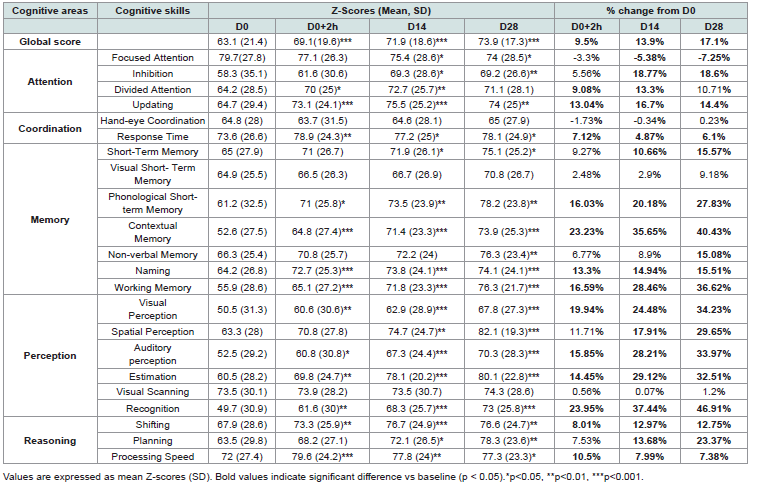
Cognitive improvements assessed with Cognifit tests were evident
as early as two hours following the first intake of Memophenol™ (Table
1). The Global Cognitive Score (Z score) increased significantly by
+9.5% at D0+2h (p < 0.001), indicating an acute enhancement of
cognitive processing efficiency and attentional control. Continuedsupplementation further amplified this effect, reaching +13.9% after
14 days and +17.1% after 28 days (p < 0.001), demonstrating both
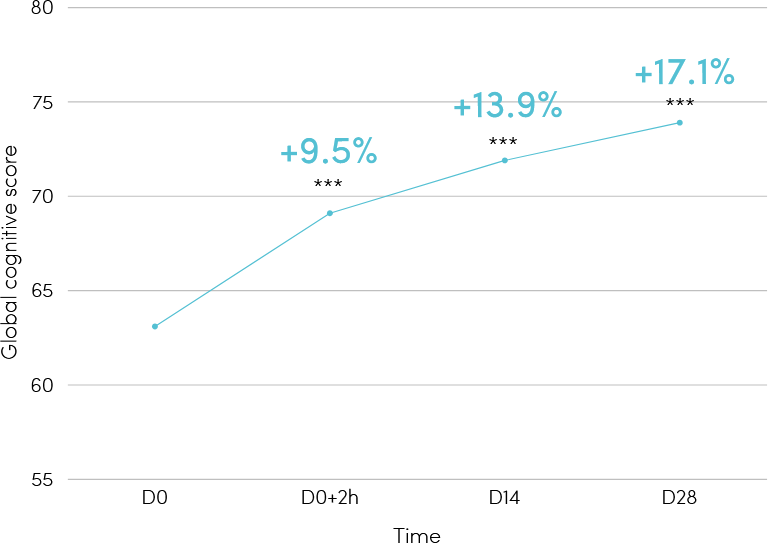
rapid and sustained benefits on global cognitive functioning [Figure 1].
Analysis of individual cognitive subdomains confirmed that
improvements were broad and not limited to a single process.
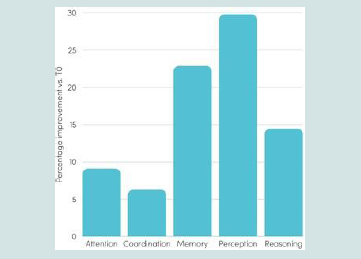
Significant increases were observed across multiple domains,
including memory (+22.9%) and perception (+29.8%), which
represented the strongest increases. Reasoning also improved
substantially (+14.5%), whereas attention (+9.1%) and coordination
(+6.3%) showed moderate but consistent enhancement over the 28-
day period [Figure 2].
The detailed evolution of each cognitive skill, as assessed by the
CogniFit Kids platform, is presented in [Table 1]. Most sub-scores
demonstrated progressive improvement over time, particularly in
contextual memory (+40.4%, p < 0.001), working memory (+36.6%,
p < 0.001), and visual perception (+34.2%, p < 0.001). These results
underline a global enhancement of higher-order cognitive abilities
related to attention Brief-A Scores
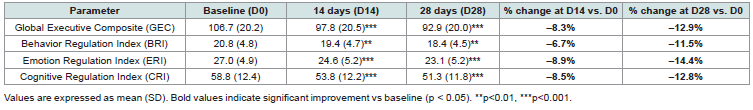
Behavioral and emotional regulation, assessed via the BRIEF-2 parental questionnaire, also improved significantly compared to baseline during the 28-day supplementation [Table 2]. The Global Executive Composite (GEC) decreased by -8.3% at D14 and -12.9% at D28 (both p < 0.001), reflecting better overall executive control of children.
All three sub-indices followed this trend: the Behavior Regulation Index (BRI) declined by –11.5%, the Emotion Regulation Index (ERI) by -14.4%, and the Cognitive Regulation Index (CRI) by -12.8% (all p < 0.001). These consistent decreases indicate marked improvements in behavioral flexibility, emotional stability, and cognitive organization – key dimensions of executive functioning essential for classroom attention and learning adaptability.
Collectively, these findings confirm that Memophenol™ supplementation was associated with enhanced self-monitoring, emotional control, and cognitive regulation in children, consistently with the objective improvements measured through the digital cognitive tasks.
Behavioral and emotional regulation, assessed via the BRIEF-2 parental questionnaire, also improved significantly compared to baseline during the 28-day supplementation [Table 2]. The Global Executive Composite (GEC) decreased by -8.3% at D14 and -12.9% at D28 (both p < 0.001), reflecting better overall executive control of children.
All three sub-indices followed this trend: the Behavior Regulation Index (BRI) declined by –11.5%, the Emotion Regulation Index (ERI) by -14.4%, and the Cognitive Regulation Index (CRI) by -12.8% (all p < 0.001). These consistent decreases indicate marked improvements in behavioral flexibility, emotional stability, and cognitive organization – key dimensions of executive functioning essential for classroom attention and learning adaptability.
Collectively, these findings confirm that Memophenol™ supplementation was associated with enhanced self-monitoring, emotional control, and cognitive regulation in children, consistently with the objective improvements measured through the digital cognitive tasks.
Parental feedback and satisfaction:
At D28, 63% of parents reported noticeable improvement in
their child’s attention, while 66% perceived the supplementation as
Figure 2:Percentages of improvement in cognitive subdomains after a 28-
day Memophenol™ intake vs. T0
globally effective and 61% expressed their intention to continue or
purchase the product.
These high levels of satisfaction demonstrate strong acceptability and perceived benefit, suggesting that the cognitive and behavioral changes were both observable and meaningful in daily life contexts.
These high levels of satisfaction demonstrate strong acceptability and perceived benefit, suggesting that the cognitive and behavioral changes were both observable and meaningful in daily life contexts.
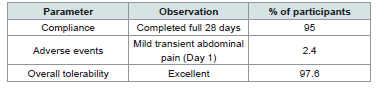
Tolerability and compliance:
Compliance throughout the 28-day intervention was excellent,
with 80.5% of participants completing the entire supplementation
period without missing a single dose, and 19.5% forgetting to take the
supplement 1 or 2 times. Only one mild and transient gastrointestinal
event (2.4%) was reported on Day 1, which resolved spontaneously
without treatment discontinuation. Overall tolerability reached
97.6%, confirming the product’s safety and suitability for pediatric
use [Table 3].The high adherence rate, coupled with positive parental feedback, reinforces the acceptability of Memophenol™ in real-world conditions and supports its feasibility for long- term use in children.
Discussion
This real-world open trial provides new evidence that daily
supplementation with 150mg Memophenol™ significantly enhances
both cognitive and executive functions in school-aged Singapore
children aged 7-12 years. Notably, improvements were observed as
early as two hours after intake, a timing that was deliberately chosen
based on pharmacokinetic data indicating that the key polyphenolic
compounds in Memophenol™, such as (−)-epicatechin and
(+)-catechin, reach their peak plasma concentrations (Tmax) within
1-2 hours after oral ingestion [26,27]. These flavanol monomers are
rapidly absorbed in the small intestine, undergo extensive phase II
metabolism (glucuronidation, sulfation, methylation), and their
conjugated metabolites are detectable in plasma and urine shortly
after intake. This acute bioavailability likely underlies the rapid
neuromodulatory effects observed in our study [24].
In line with this, Bensalem et al. demonstrated that supplementation with Memophenol™ led to a marked increase in the urinary excretion of specific polyphenol metabolites, including (+)-catechin, (−)-epicatechin, B-type procyanidin dimers, and their conjugated forms, as early as after the first dose and sustained over 6 months [20]. The progressive increase in performance over 28 days demonstrates the cumulative benefit of sustained supplementation,
In line with this, Bensalem et al. demonstrated that supplementation with Memophenol™ led to a marked increase in the urinary excretion of specific polyphenol metabolites, including (+)-catechin, (−)-epicatechin, B-type procyanidin dimers, and their conjugated forms, as early as after the first dose and sustained over 6 months [20]. The progressive increase in performance over 28 days demonstrates the cumulative benefit of sustained supplementation,
extending previous findings from adult and elderly populations
to a younger age group. The observed acute effects are consistent
with the rapid action of flavanol monomers on neurovascular and
neurometabolic pathways [36,37]. Previous human studies have
demonstrated that Memophenol™ can enhance attentional control
and information processing speed within 90 minutes post-intake
[21], and other polyphenol-rich interventions (e.g., flavanol-rich
cocoa beverages, grape-derived polyphenol drinks) have been shown
to acutely enhance cerebral perfusion and oxygen availability, thereby
improving attentional control and information processing speed
within 1-2 hours post- intake [38]. Thus, the fast cognitive response
observed in children after Memophenol™ intake in our study is
consistent with these pharmacokinetic and mechanistic findings, and
may operate through similar polyphenol-driven pathways shared
across flavonoid-rich products. This pattern aligns with mechanistic
frameworks summarized in clinical reviews, which highlight
improvements in endothelial function and hemodynamic responses
as plausible proximal drivers of rapid changes in cognitive efficiency
(e.g., processing speed and attentional control), while acknowledging
that not all acute trials show benefits depending on the cognitive
endpoints and the intervention dose/matrix [35].
In the present study, acute improvements in the Global Cognitive Score (+9.5% after 2 hours) were accompanied by early changes in executive regulation as perceived by parents. Because the Global Cognitive Score is a Z-score normalized to a reference population, these results indicate that children moved from slightly above or around average baseline performance toward higher-than-average levels after supplementation. According to Cognifit normative guidelines, a Z-score improvement of this magnitude reflects a meaningful shift relative to age-matched cognitive norms, supporting the real-world relevance of the effect. The longer- term improvements observed after 14 and 28 days (+13.9% and +17.1%, respectively) likely reflect a progressive neuroadaptive response induced by repeated exposure to polyphenols, including enhanced synaptic plasticity and antioxidant protection within hippocampal and prefrontal circuits. These results may be explained by the mechanisms described by Bensalem et al. [23-25], who demonstrated in aged mice that Memophenol™ promotes hippocampal neurogenesis and synaptic connectivity, leading to measurable gains in learning and memory. Similarly, clinical trials in older adults with mild cognitive impairment [22] and in healthy students under cognitive load [21] have reported significant improvements in attention, working memory, and learning efficiency following supplementation with Memophenol™. The consistent direction of effects across these studies strengthens the hypothesis that the ingredient acts on conserved pathways of neuronal signaling across different life stages.
Beyond cognitive enhancement, the significant reduction in BRIEF-2 indices (GEC: –12.9%, p < 0.001) highlights the impact of supplementation on self-regulatory abilities. Improvements in emotion regulation (–14.4%) and behavior regulation (–11.5%) suggest a broader influence of Memophenol™ on executive domains that integrate cognitive, emotional, and social functioning. These effects are particularly relevant in academic contexts where attention, self-control, and emotional stability strongly influence learning outcomes and well- being. Importantly, the formulation was well
In the present study, acute improvements in the Global Cognitive Score (+9.5% after 2 hours) were accompanied by early changes in executive regulation as perceived by parents. Because the Global Cognitive Score is a Z-score normalized to a reference population, these results indicate that children moved from slightly above or around average baseline performance toward higher-than-average levels after supplementation. According to Cognifit normative guidelines, a Z-score improvement of this magnitude reflects a meaningful shift relative to age-matched cognitive norms, supporting the real-world relevance of the effect. The longer- term improvements observed after 14 and 28 days (+13.9% and +17.1%, respectively) likely reflect a progressive neuroadaptive response induced by repeated exposure to polyphenols, including enhanced synaptic plasticity and antioxidant protection within hippocampal and prefrontal circuits. These results may be explained by the mechanisms described by Bensalem et al. [23-25], who demonstrated in aged mice that Memophenol™ promotes hippocampal neurogenesis and synaptic connectivity, leading to measurable gains in learning and memory. Similarly, clinical trials in older adults with mild cognitive impairment [22] and in healthy students under cognitive load [21] have reported significant improvements in attention, working memory, and learning efficiency following supplementation with Memophenol™. The consistent direction of effects across these studies strengthens the hypothesis that the ingredient acts on conserved pathways of neuronal signaling across different life stages.
Beyond cognitive enhancement, the significant reduction in BRIEF-2 indices (GEC: –12.9%, p < 0.001) highlights the impact of supplementation on self-regulatory abilities. Improvements in emotion regulation (–14.4%) and behavior regulation (–11.5%) suggest a broader influence of Memophenol™ on executive domains that integrate cognitive, emotional, and social functioning. These effects are particularly relevant in academic contexts where attention, self-control, and emotional stability strongly influence learning outcomes and well- being. Importantly, the formulation was well
tolerated, with no adverse effects reported other than one mild,
transient gastrointestinal event. This excellent safety profile aligns
with previous clinical data [20-22] in both young and older adults,
confirming that Memophenol™ supplementation at a daily dose of
150 mg is safe and acceptable for pediatric use. This is also consistent
with broader clinical trial evidence suggesting that polyphenol-rich
interventions are generally safe and well tolerated across a wide range
of doses and durations, with no consistent signal of excess adverse
effects compared with controls [34].
Beyond its combination of an acute (2h)) and a short-term (14- 28 days) assessment, this study presents several strengths. First, it combines objective cognitive assessments with subjective parental feedback, providing a comprehensive evaluation of the product’s effects. Second, the real-life setting enhances the ecological validity and relevance of the findings for end-users. Third, the longitudinal design allows for the monitoring of individual changes over time. Nevertheless, some limitations should be acknowledged. First, the repeated administration of the CogniFit Kids battery may have induced a potential learning effect, notably in healthy kids, potentially inflating cognitive scores independently of the intervention. Second, the lack of a control group prevents us from attributing the observed improvements solely to the intervention, as natural maturation or external factors cannot be ruled out. Third, the absence of a double-blind design may have introduced expectation or observer bias, particularly in subjective parental feedback. Additionally, the study population consisted exclusively of healthy children living in Singapore, which may limit the generalizability of the results to other socio-educational contexts. Taken together, these limitations suggest that the observed effects should be interpreted with caution, and further randomized, controlled, and blinded studies are warranted to confirm these preliminary findings.
Beyond its combination of an acute (2h)) and a short-term (14- 28 days) assessment, this study presents several strengths. First, it combines objective cognitive assessments with subjective parental feedback, providing a comprehensive evaluation of the product’s effects. Second, the real-life setting enhances the ecological validity and relevance of the findings for end-users. Third, the longitudinal design allows for the monitoring of individual changes over time. Nevertheless, some limitations should be acknowledged. First, the repeated administration of the CogniFit Kids battery may have induced a potential learning effect, notably in healthy kids, potentially inflating cognitive scores independently of the intervention. Second, the lack of a control group prevents us from attributing the observed improvements solely to the intervention, as natural maturation or external factors cannot be ruled out. Third, the absence of a double-blind design may have introduced expectation or observer bias, particularly in subjective parental feedback. Additionally, the study population consisted exclusively of healthy children living in Singapore, which may limit the generalizability of the results to other socio-educational contexts. Taken together, these limitations suggest that the observed effects should be interpreted with caution, and further randomized, controlled, and blinded studies are warranted to confirm these preliminary findings.
Conclusion
This real-world open trial provides preliminary evidence that
daily supplementation with 150 mg of a Grape and Wild Blueberry
Polyphenol-Rich Extract (Memophenol™) may significantly enhance
cognitive performance and executive functioning in healthy school aged
children aged 7-12 years old from Singapore. Improvements
were detectable as early as two hours after intake and continued to
increase over 28 days, suggesting a combination of rapid, exposuredriven
effects and cumulative benefits with repeated supplementation.
The rapid onset is consistent with previously reported acute cognitive
effects of Memophenol™ in healthy young adults under sustained
cognitive demand [21]. The continued improvement across the 14–
28-day period also fits with prior Memophenol™ findings in older
cohorts, including randomized controlled trials showing benefits
on information-processing speed, learning-related measures, and
executive-function indices (e.g., BRIEF-A) in adults with mild
cognitive impairment [22] as well as cognitive support in healthy
older adults [20], highlighting its potential as a natural and safe
nutritional strategy to help school-aged children sustain attention in
the classroom, strengthen day-to-day executive skills (e.g., working
memory, attention, etc.), and support learning efficiency and
behavioral self-regulation when facing homework load, exams, and
the routine cognitive demands of school life.
Notably, the agreement between objective task-based performance
(CogniFit) and real-life executive-function evaluations (BRIEF-2 and
parental feedback) supports the robustness and practical relevance
of the observed changes, suggesting they may extend beyond a
single assessment method and translate to everyday self-regulation.
However, because this study was open-label and lacked a control
group, the results should be considered exploratory; well-powered
randomized, double-blind, placebo-controlled pediatric trials
incorporating biochemical endpoints are needed.
Funding and Conflicts of Interest:
Funding: This study was funded by Activ’Inside (Beychacet-
Caillau, France). The sponsor provided the study material
Memophenol™ and covered operational costs but had no role in data
collection, statistical analysis, or interpretation of the results.Conflict of Interest: All authors are full-time employees of Activ’Inside. This affiliation did not influence the design, analysis, or interpretation of the study results.
Acknowledgements::
The authors thank the Syres team (Dr
David Tan, Marion Bousquet, and Séverine Freyssinet) for clinical
supervision and coordination of the study, as well as the participating
families for their time and commitment.References
Citation
Gille I, Arnaud E, Jeanroy M, Lemaire B, Gaudout D. Evaluation of Acute and Sustained Cognitive Effects of a Grape and Blueberry Polyphenol-Rich Extract in Healthy 7-12 Years Old Children: A Home-Based, Real-World Consumer Study. J Nutri Health. 2026;9(1): 01.