Journal of Clinical Trials & Patenting
Download PDF
Review Article
Phenytoin: Repurposing an Old Molecule and Patent Strategies for Neuropathic Pain
Keppel Hesselink JM*
- Institute of Neuropathic Pain, the Netherlands; University of Witten/Herdecke, Germany
*Address for correspondence: Keppel Hesselink JM, Institute for Neuropathic Pain, the Netherlands and Faculty of health, University of Witten/Herdecke, Germany; E-mail: jan@neuropathie.nu
Citation: Keppel Hesselink JM. Phenytoin: Repurposing an Old Molecule and Patent Strategies for Neuropathic Pain. J Clin Trials Pat 2018;3(1): 3.
Journal of Clinical Trials & Patenting | ISSN: 2573-3834 | Volume: 3, Issue: 1
Submission: 07 March, 2018| Accepted: 10 April, 2018 | Published: 14 April, 2018
Copyright: © 2018 Keppel Hesselink JM. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Phenytoin was introduced in the clinic in 1938, and due to its broad mechanism of action it has become a clear prototype of a repurposed or repositioned drug in a variety of indications, from bipolar disorders up to wound healing. Some years ago, we identified phenytoin as a co-analgesic with optimal properties to put into a topical formulation for the treatment of peripheral neuropathic pain. Phenytoin is the archetype of a sodium channel blocking drug and the main target for its efficacy as a neuropathic analgesic resides in the epidermis: the nociceptor. The development however of an old drug in a new off-label indication can only be attractive in case of a well-balanced patent strategy.
Introduction
Old molecules may have many unexpected promises for the treatment of different disorders, and repurposing or repositioning such drugs is hot. Especially hot, since the clinical development leading to registration for old drugs in principle can be cheap and quick. However, for such development financial incentives are needed, preferably based on a patent strategy.
We started the develop a new topical formulation of an old molecule, phenytoin, in a new indication, peripheral neuropathic pain, and want to share our strategy related to patenting and clinical development of a repurposed drug.
History of phenytoin as a multi-purpose drug
Phenytoin was first introduced in the clinic in 1938 as a new anti-epileptic, devoid of the side effects of the barbiturates [1]. The compound was never protected by any patent, and thus interest exploring its putative clinical value in many indications was long absent. Since the first use of phenytoin in the neurological clinic, various observations suggested its clinical relevance in a great number of indications, and the driving factor for this repurposing of phenytoin was the Wall Street tycoon Jack Dreyfus (August 28, 1913 - March 27, 2009) [2].
Jack Dreyfus is probably the first non-scientist who can be qualified as a champion for the repurposing of old drugs. In 1963 he took up the courage to ask his treating physician to write him a prescription for phenytoin for his depressed mood. First they believed Dreyfus his remarkable recovery should be attributed to luck, but Dreyfus soon became an ambassador for phenytoin and in a short period of time many others started to benefit from taking phenytoin as an antidepressant, as Dreyfus pointed out at the age of 90 [3].
Phenytoïne (diphenylhydantoine; 5,5-diphenylimidazolidine-2,4-dione) was synthesized in 1908 by the German chemist professor Heinrich Biltz [4]. He sold the molecule to Parke-Davis, who had the molecule resting on their shelves for nearly 30 years. After being introduced in 1938 in the clinic by the neurologists Merritt and Putnam, one of the key opinion leaders at that time, William Gordon Lennox (1884-1960), expressed great enthusiasm for the new drug: “The big news of the year is the discovery and clinical use of sodium diphenyl hydantoinate (Dilantin Sodium). Merritt and Putnam, working at the Neurological Unit of the Boston City Hospital, report the results of treating 200 non-institutionalized cases of epilepsy” [5].
‘Dilantin sodium’ was subsequently added to the catalog price list of Parke-Davis in June 1938, but was never patented. Phenytoin sodium Extended Capsule, for oral use, was subsequently developed and approved in the USA in 1953. Up to now the listed indication for phenytoin is still the treatment of tonic-clonic (grand mal) and psychomotor (temporal lobe) seizures and prevention and treatment of seizures occurring during or following neurosurgery. In 1988 Dreyfus already complained: “today, 50 years after its first use, phenytoin’s only listed indication is still ‘anticonvulsant’. There is a flaw in our system of bringing prescription medicines to the public” [6]. In the same book containing this quotation, ‘ The broad range of clinical use of phenytoin’, published in 1988, a great number of indications for phenytoin were listed: aggression, depression, bipolar disorders, wound healing, pain syndromes and tinnitus, among many others.
Phenytoin as a co-analgesic
Recently we surprisingly found that there is no difference in the amount of evidence to support carbamazepine and phenytoin in the treatment of trigeminal neuralgia [7]. Already as early as 1942, three patients were reported suffering from trigeminal neuralgia; 200-300 mg phenytoin daily was effective in reducing the pain [8]. These data were drivers for us to design a patented strategy around topical phenytoin in the treatment of peripheral neuropathic neuropathic pain syndromes.
Most patents follow the strategy of broadly covering a great number of compounds and derivatives thereof, as well as a wide range of indications. However, patent protection lasts for 20 years, and many compounds and indications remain unexplored after expiry of the patent, due to the absence of an economic stimulus. Phenytoin is a good example. Although a great number of indications have been explored, clinical development leading to the registration of new indications for this multipurpose drug remain absent. One basically needs patents to make development attractive [9]. In the absence of a patent, not much development work is done, and physicians will always need to prescribe the repurposed drug ‘off-label’ , with all its problems.
Topical phenytoin: its mechanism of action
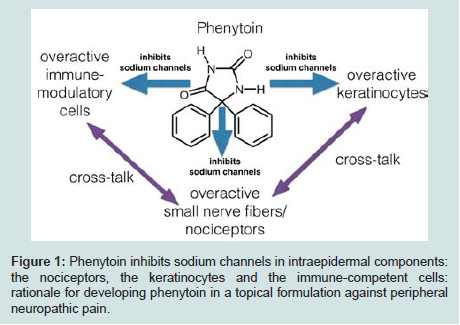
In the period 1972-1980 it was established that phenytoin inhibits sodium permeability via its blocking effect on these ion channels [10]. Several studies in the early 1980s described the purification of the sodium channel protein, and initial studies using synaptosomesystems pointed out that phenytoin blocked voltage-gated sodium channels [11]. To date there is clear consensus that sodium channels are the most important targets of phenytoin [12]. Phenytoin is the archetypical unselective sodium channel blocker [13]. Phenytoin’s binding-site is situated at the inner cytoplasmic membrane, at the inner vestibule of the pore of the ion channel. This most probably explains its broad activity for many if not all of the sodium channels, as this binding site can be found in all members of this channel family [14]. Sodium channels of various types have been characterized on the nociceptors in the skin, the keratinocytes and the immunecompetent cells [15]. These 3 elements cross-talk and induce overactive nociceptors, leading to peripheral sensitization (Figure 1). This is exactly the reason why we selected the non-selective, broad acting sodium channel blocker phenytoin in a topica l formulation for the treatment of peripheral neuropathic pain. As we have seen recently that a selective NaV1.7 blocker in a topical formulation failed to prove efficacy, this would indirectly support our choice for a broad acting blocker [16].
Figure 1: Phenytoin inhibits sodium channels in intraepidermal components: the nociceptors, the keratinocytes and the immune-competent cells: rationale for developing phenytoin in a topical formulation against peripheralneuropathic pain.
Patent and development strategy: phenytoin in a topical formulation for the treatment of neuropathic pain
To date we have gathered and published some clinical data, supporting that topical applied phenytoin, in a concentration range of 5-20%, is safe in a number of neuropathic pain syndromes. We also detected and describedfirst indicators for efficacy [17-20]. Based on these data we filed 2 patents on specific topical formulations of phenytoin for the treatment of neuropathic pain: ‘topical phenytoin for use in the treatment of peripheral neuropathic pain’ and ‘topical pharmaceutical composition containing phenytoin and a (co-) analgesic for the treatment of chronic pain’. Both patents entered the PCT phase earlier this year (2018).
These patents will become cornerstones for a development strategy to change the current situation of off-label use of compounded phenytoin creams. Phenytoin creams are currently prescribed for neuropathic pain, in the Netherlands and the USA, based on our recent findings [16-20]. An investigator driven development plan is under discussion, focused on painful diabetic neuropathy as a first indication. End of phase IIa data, including the proof of concept based on the results of a placebo-controlled single-blind treated cohort of 20 neuropathic pain patients, are available and will soon be published. Consultation with an EU competent authority is planned and we anticipate a quick and lean development up to end of phase III, based on data gathered in the past and published in peer reviewed papers. Especially in the fields of preclinical data and proof of principle, toxicology, side-effects, plasma-levels, pharmacokinetics and mechanism of action we expect to be able to extract sufficient data from the past for supporting the submission related to the registration of a topical formulation in painful diabetic neuropathy. It is our belief that especially the facts that we did not detect any phenytoin plasma levels after administration, and we did not see any serious adverse event in our first 100 patients treated, together with the intraepidermal mechanism of action, will support an uncomplicated and expedited development.
References
- Keppel Hesselink JM, Kopsky DJ (2017) Phenytoin: 80 years young, from epilepsy to breast cancer, a remarkable molecule with multiple modes of action. J Neurol 264: 1617-1621.
- Keppel Hesselink JM (2018) Phenytoin repositioned in wound healing: clinical experience spanning 60 years. Drug Discov Today 23: 402-408.
- Biltz, H (1908) Constitution of the products of the interaction of substituted carbamides on Benzil and certain new methods for the preparation of 5, 5-diphenylhydantoin. Chem Ber 41: 1379-1393.
- Shorvon SD (2009) Drug treatment of epilepsy in the century of the ILAE: The first 50 years, 1909-1958. Epilepsia 50(Suppl): 69-92.
- Dreyfus J (1988) Brief history. In: Smith BH, Bogovh S, Dreyfus J (Eds), The broad range of clinical use of phenytoin. Bioelectrical modulator. Dreyfus medical foundation.
- Keppel Hesselink JM, Schatman ME (2017) Phenytoin and carbamazepine in trigeminal neuralgia: marketing-based versus evidence-based treatment. J Pain Res 10: 1663-1666.
- Bergouignan, M (1942) Successful cure of essential facial neuralgias by sodium diphenylhydantoinate. Rev Laryngol Otol Rhinol (Bord) 63: 34-41.
- Keppel Hesselink JM (2017) Amantadine and phenytoin: patent protected cases of drug repositioning. Clin Invest 7:11-16.
- Lipicky RJ, Gilbert DL, Stillman IM (1972) Diphenylhydantoin inhibition of sodium conductance in squid giant axon. Proc Natl Acad Sci U S A 69: 1758-1760.
- Willow M, Kuenzel EA, Catterall WA (1984) Inhibition of voltage-sensitive sodium channels in neuroblastoma cells and synaptosomes by the anticonvulsant drugs diphenylhydantoin and carbamazepine. Mol Pharmacol 25: 228-234.
- Qiao X, Sun G, Clare JJ, Werkman TR, Wadman WJ (2014) Properties of human brain sodium channel α-subunits expressed in HEK293 cells and their modulation by carbamazepine, phenytoin and lamotrigine. Br J Pharmacol 171: 1054-1067.
- Sills GJ (2013) Classical mechanisms of action of antiepileptic drugs. In: Potschka H, Lerche H (Eds.), Therapeutic Targets and Perspectives in the Pharmacological Treatment of Epilepsy. Bremen UNI-MED Verlag pp: 62-65.
- Mantegazza M, Curia G, Biagini G, Ragsdale DS, Avoli M (2010) Voltage-gated sodium channels as therapeutic targets in epilepsy and other neurological disorders. Lancet Neurol 9: 413-424.
- Keppel Hesselink JM, Kopsky DJ, Bhaskar AK (2016) Skin matters! The role of keratinocytes in nociception: a rational argument for the development of topical analgesics. J Pain Res 10: 1-8.
- Price N, Namdari R, Neville J, Proctor KJ, Kaber S, et al. Safety and efficacy of a topical Sodium Channel Inhibitor (TV-45070) in patients with Postherpetic Neuralgia (PHN). A randomized, controlled, proof-of-concept, crossover study, with a subgroup analysis of the Nav1.7 R1150W Genotype. Clin J Pain 33: 310-318.
- Keppel Hesselink JM, Kopsky DJ (2017) Topical phenytoin cream in small fiber neuropathic pain: fast onset of perceptible pain relief. Int J Pain Relief 1: 015-019.
- Keppel JM, Kopsky DJ (2017) Topical phenytoin cream reduces burning pain due to small fiber neuropathy in sarcoidosis. J Anesth Pain Med 2: 1-3.
- Kopsky DJ, Keppel Hesselink JM (2017) Topical phenytoin for the treatment of neuropathic pain. J Pain Res 10: 469-473.
- Jan MK Hesselink, David J Kopsky D (2017) Topical phenytoin in neuralgic pain, peripheral modulation of central sensitization: two case reports. J Pain Relief 6: 284.
- Keppel Hesselink JM, Kopsky DJ (2017) Burning pain in small fibre neuropathy treated with topical phenytoin: rationale and case presentations. J Clin Anesth Pain Med 1: 6.