Journal of Clinical and Investigative Dermatology
Download PDF
Case Report
Pyodermatitis-Pyostomatitis Vegetans: A Cutaneous Clue for Crohn’s Disease
Tallman R1*, Beatty C1, Ghareeb E1, Gayam S2 and Zinn Z2
1Department of Dermatology, West Virginia University Medicine, USA
2Department of Gastroenterology and Hepatology, West Virginia
University Medicine, USA
*Address for Correspondence: Tallman R, Department of Dermatology, West Virginia University Medicine,
Morgantown, Medical Center Drive, HSC P.O. Box 9158, Morgantown, WV
26506-9158, Fax: 304-293-8055, Tel: 855-988-273; Email: rmtallman@mix.
wvu.edu
Submission: 30 July, 2021;
Accepted: 06 September, 2021;
Published: 10 September, 2021
Copyright: © 2021 Tallman R, et al. This is an open access article
distributed under the Creative Commons Attribution License, which permits
unrestricted use, distribution, and reproduction in any medium, provided the
original work is properly cited.
Abstract
Pyodermatitis-pyostomatitis vegetans is a neutrophilic dermatosis
that is classically associated with inflammatory bowel disease. It is
rare with less than 40 cases reported in the literature within the last 10
years. We discuss the case of a 30-year-old male who presented with
pyodermatitis-pyostomatitis vegetans and previously undiagnosed
Crohn’s disease. We aim to emphasize the importance of prompt
recognition of pyodermatitis-pyostomatitis vegetans order to
commence a thorough investigation of underlying inflammatory bowel
disease, and initiate appropriate therapy. Because no standardized
treatment regimens have been established, we also describe the
variability in treatment responses seen with pyodermatitis-pyostomatitis
vegetans with a review of the literature.
Abbreviations
Pyodermatitis-pyostomatitis vegetans (PPV); Inflammatory
bowel disease (IBD); Non-steroidal anti-inflammatory (NSAID);
Direct immunofluorescence (DIF); Gastrointestinal (GI); Ulcerative
colitis (UC)
Introduction
Pyodermatitis-pyostomatitis vegetans (PPV) is a neutrophilic
dermatosis characteristically associated with underlying inflammatory
bowel syndrome (IBD) [1]. The first reported case of PPV
was in 1898 by Francois Hallopeau [2], and only 37 cases of PPV
were reported in the literature between 1979 and 2011 [3]. Diagnosis
can be challenging due to its resemblance to autoimmune blistering
disorders such as pemphigus vegetans, and infectious etiologies such
as Staphylococcus aureus and blastomycosis. Recognizing the signs
and symptoms of PPV is critical in order to prompt further work up
for IBD and begin appropriate management, especially considering
that responses to treatments can be variable among patients and often
require trial and error to maintain remission. Here, we report a case of
PPV that highlights the importance of prompt diagnosis, especially in
cases where underlying IBD has yet to be uncovered. We also perform
a literature review of treatment responses in patients with PPV.
Case Presentation
A 30-year-old Caucasian male with fever, hypotension, and
tachycardia was admitted for one-week history of abdominal pain,
diarrhea, rash, and concern for sepsis. Past medical history revealed
another recent admission for melena, diarrhea, and abdominal pain
which he had self-managed with 1 gram of ibuprofen per day. At
that time, EGD and colonoscopy revealed edematous and ulcerated
tissue with mucopurulent material. Biopsy revealed ulceration
and granulation tissue, which was diagnosed as non-steroidal
anti-inflammatory drug (NSAID)-induced colitis. NSAIDs were
discontinued, however diarrhea and melena had since persisted Additional past medical history included intravenous drug abuse
managed with buprenorphine and naloxone, and recent unintentional 35-pound weight loss.
On admission, he was noted to have an elevated white blood cell
count of 12,800 /UL with 87% polymorphonuclear lymphocytes.
C-Reactive Protein was elevated at 189.5 mg/L (normal <8 mg/L).
Due to concern for sepsis secondary due to bacterial endocarditis,
vancomycin, cefepime, and metronidazole were started. Examination
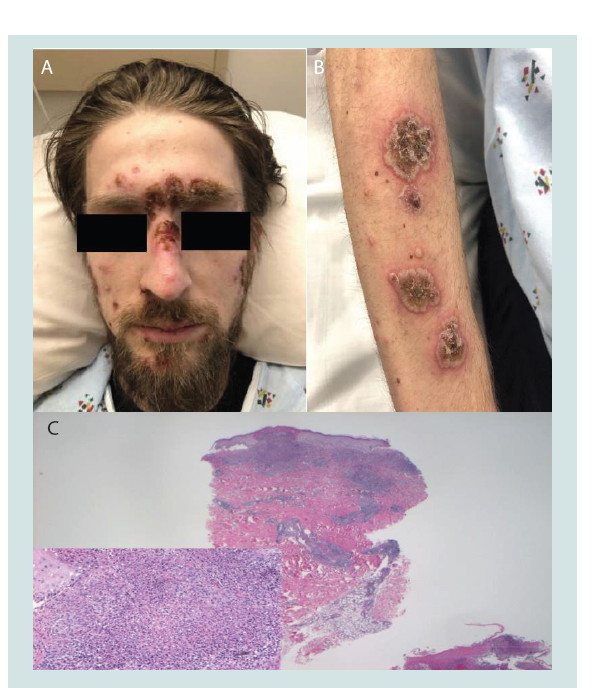
of the rash revealed crusted vegetative plaques with a rim of pustules
localized to the face, arms, and legs (Figure 1A,Figure 1B), and multiple
erosions on the hard palate. Dermatologic differential diagnosis
included deep fungal infection including blastomycosis, staphylococcal
infection, pemphigus vegetans, and PPV. Biopsyrevealed necrotic
epidermis, ulcerations, and neutrophilic infiltration (Figure 1C) with
negative direct immunofluorescence (DIF). Tissue cultures were
negative. Histologic diagnosis was compatible with a neutrophilic dermatosis including PPV.
Figure 1: Pyodermatitis vegetans lesions and histopathology. A. Multiple
erosions and crusts were observed on the face. B. On the extensor surfaces
were multiple erythematous papules and plaques with pustules studding the
borders and verrucous crust in the center of the lesions. C. Hematoxylin and
eosin at x 2, inset at x10; Punch biopsy from left arm showed an ulcerated
lesion with underlying neutrophilic abscesses and a neutrophilic interstitial
infiltrate. PAS, GMS, AFB Fite stains were all negative for microorganism.
Direct immunofluorescence was negative. Bacterial and fungal tissue cultures
were all negative for growth.
Given ongoing diarrhea, melena, and skin biopsy compatible with
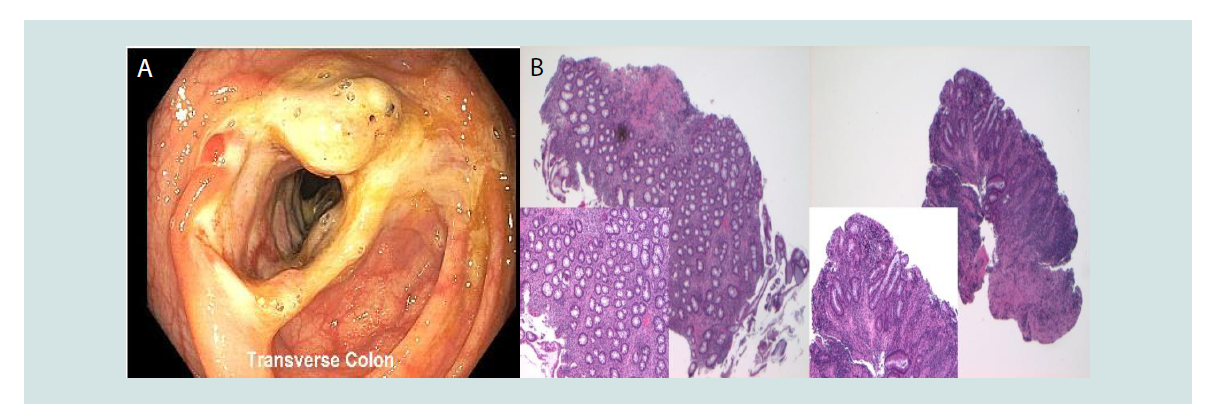
PPV, gastroenterology was consulted. Colonoscopy revealed multiple
strictures and large ulcers throughout the colon (Figure 2A). Biopsy of
the colonic lesions revealed focal chronic active colitis with ulceration
and granulation and no granulomas present (Figure 2B). IBD panel
was consistent with Crohn’s disease. Given his history and skin
biopsy results, it was suspected that the lesions were PPV secondary
to IBD. The patient was started on prednisone 40 mg daily with
significant improvement of skin and gastrointestinal (GI) symptoms.
Vedolizumab infusions successfully controlled GI symptoms after
prednisone was tapered, however cutaneous manifestations of PPV
recurred. He was eventually switched to infliximab with successful
remission of both Crohn’s disease and PPV.
Figure 2: Inflammatory bowel disease. A. Colonoscopy revealed large ulcers found at the cecum, in the ascending colon, transverse colon, descending colon,
and sigmoid colon. There was narrowing of the mucosa at several sites throughout the colon located near the ulcerations. B. Hematoxylin and eosin at x 4,insets at x10; On histology, there was focal active colitis with ulceration and granulation tissue. No granulomas were seen.
Discussion
PPV is a rare neutrophilic dermatosis that characteristically
presents with friable pustules of the oral cavity that form a “snailtrack”
pattern and papulopustular cutaneous lesions that give rise to
vegetating plaques [1]. PPV has a well-documented association with
IBD. Although it presents more often as a sequela of ulcerative colitis
(UC), it can be seen in Crohn’s disease as well, as in our patient [4].
Cases have also been reported with primary sclerosing cholangitis.
It is more common for patients to present with GI symptoms
prior to the onset of PPV, however skin lesions may appear first in
approximately 15% of cases [1]. Although classic PPV involves both
the mouth and the skin, involvement of both sites is not required to
render a diagnosis. The etiology of PPV remains uncertain, and males
between the ages of 20 and 50 are preferentially affected (3:1 male:
female ratio) [5].
PPV has characteristic histology including intraepithelial
and subepithelial splitting with neutrophilic and eosinophilic
microabscesses. It is important to distinguish PPV from auto-immune
blistering disorders, including the pemphigus vegetans subtype of
pemphigus vulgaris, IgA pemphigus, epidermolysis bullosa acquisita,
and dermatitis herpetiformis. Unlike these blistering disorders, DIF
is negative in PPV. Additional considerations include cutaneous
infections such as blastomycosis-like pyoderma due to S.aureus, and
endemic dimorphic fungal infections such as blastomycosis, which
can be differentiated by positive tissue cultures. Finally, clinical
findings of oral ulcers and documented IBD also support the diagnosis
of PPV [1,3]. Our patient exhibited characteristic histology of PPV
with negative DIF, negative bacterial and fungal tissue cultures, and
presence of oral ulcers diffusely on the hard palate with new diagnosis
of underlying IBD.
There is no standardized treatment for PPV, and responses
to different therapies are extremely variable among patients. To
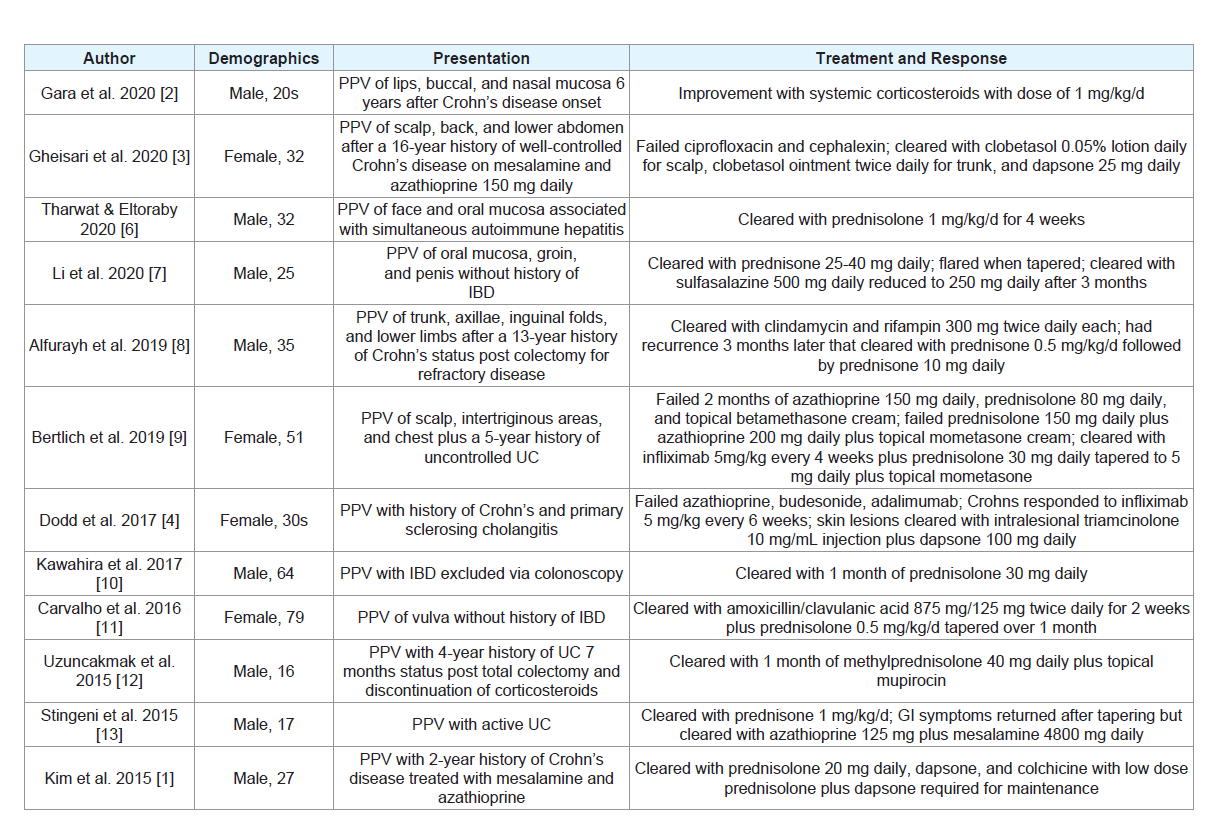
illustrate this, we performed a review of case reports and case series
of PPV since 2015 in PubMed detailing patient responses to varying
treatment regimens, which is summarized in Table 1. Although
there is no standard algorithm for management, corticosteroids are
considered efficacious first line therapy. They are used as systemic,
intralesional, and topical treatments depending on disease severity.
All the cases reviewed that used corticosteroids achieved clearance,
however relapse was common when tapering [1-4,6-13]. Other
systemic treatments for PPV include dapsone, azithromycin cyclosporine, methotrexate, and infliximab, although varying efficacy has been reported with these options. Other topical treatments
include tacrolimus, mupirocin, and antiseptic mouth washes for oral
involvement.
Often the clinical courses of PPV and IBD are parallel [1], and
successful management of the underlying IBD typically results in
resolution of PPV [2]. However, there have also been cases in which
PPV develops and persists despite well controlled IBD [1,3,4]. Our
patient was started on vedolizumab initially for Crohn’s disease due
to preference over infliximab, however he was eventually switched
to infliximab after his skin manifestations continued to flare. Both
his skin and GI symptoms have been well controlled with infliximab
infusions. Previous reports have shown successful responses of both
cutaneous and GI symptoms to infliximab [14], whereas vedolizumab
has shown less efficacy in treating cutaneous manifestations of IBD
[15].
Conclusions
In summary, we present a case of PPV that posed a diagnostic
challenge, given both the patient’s risk factors for infectious cause
and unknown underlying Crohn’s disease. Our case demonstrates
characteristic clinical and histological features of PPV and highlights
the necessity to recognize this condition to screen for underlying
IBD. A review of the literature revealed the majority of cases clear
with corticosteroid treatment, however relapse is common once
discontinued. Although PPV has been shown to resolve with
management of IBD, this was inconsistent among the reviewed
cases and with our patient as well. Responses to other treatments for
maintenance of PPV is variable and often requires trial and error to
achieve remission.
References
Citation
Tallman R, Beatty C, Ghareeb E, Gayam S, Zinn Z. Pyodermatitis-Pyostomatitis Vegetans: A Cutaneous Clue for Crohn’s Disease. J Clin Investigat Dermatol. 2021;9(2): 4