Journal of Clinical and Investigative Dermatology
Download PDF
Research Article
Serum Amyloid A as an Inflammation Marker in Lichen Planus
Metwalli M1, Ibraheem AH2, Abu bakr H3 and Fathia MK1
1Department of Dermatology, Zagazig University, Egypt
2Department of Clinical Pathology, Faculty of Medicine, Egypt
3Department of Medicine, Zagazig University, Egypt
*Address for Correspondence: Fathia M. Khattab, Department of Dermatology, Venereology and Andrology,Faculty of Medicine, Zagazig University, Egypt, Tel: 00201111108729;
Email: fathiakhattab@ yahoo.com
Submission: 22 June, 2021
Accepted: 20 July, 2021
Published: 24 July, 2021
Copyright: © 2021 Metwalli M, et al. This is an open access article
distributed under the Creative Commons Attribution License, which permits
unrestricted use, distribution, and reproduction in any medium, provided the
original work is properly cited.
Abstract
Background: Lichen planus (LP) is a chronic T cell-mediated
inflammatory disorder that can affect skin, mucosa, hair, and nails.
Serum amyloid A (SAA) is a conserved acute phase protein in response
to trauma, infection, malignancy, and severe stress. SAA may have homeostatic role rather than a pro-inflammatory or anti-inflammatory
one. Serum levels of SAA were demonstrated to be raised in several
inflammatory systemic and skin diseases as rheumatoid arthritis,
systemic lupus erythematosus, and psoriasis.
Aim: This study aimed to evaluate serum levels of SAA, and IL 6 in
a sample of Egyptian LP patients and to estimate its correlation with
disease severity.
Patients and methods: We included 21 adult patients with LP
and 21 healthy adults as control. The total score of LP severity was
measured for all patients through measurement of the affected body
surface area in cutaneous LP patients while using a semi-quantitative
clinical scoring system for oral LP together with the visual analog scale
for pain assessment in OLP. Serum levels of SAA were estimated in all
participants using enzyme-linked immunosorbent assay.
Results: The expression levels of SAA and IL 6 in peripheral blood
of patients in the two groups were detected. Pearson analysis was
used in the correlation between SAA and IL 6 and Receiver operating
characteristic (ROC) curve was employd to analyze the predictive
value of SAA and IL 6 for LP severity. Logistic regression analysis was
used to analyze the risk factors of LP patients. The expression levels of
SAA and IL 6 of patients in sever form were significantly higher than
those in mild form (P<0.05). Pearson analysis showed that SAA was
positively correlated with IL 6 expression (P<0.05). ROC curve analysis
showed that AUC predicted by SAA and IL 6 for LP severity was 0.789
and 0.762 (P<0.05). Logistic regression analysis showed that SAA and IL
6 were prediction indexes of LP severity.
Conclusion: The levels of SAA and IL 6 were significantly increased
during LP and effectively predicted the severity of LP and is a risk
factor affecting LP patients. Further studies are needed to establish
this association then it might be used for the evaluation of therapeutic
outcomes.
Introduction
Lichen planus (LP) is a chronic T-cell mediated mucocutaneous
disorder of multifactorial pathogenesis. It is characterized by
pruritic, purplish, flat, polygonal papules and plaques with a white
lacy surface. It is more predominant in adults of middle-aged with slight increase in female patients [1]. it is characterized by local and
systemic inflammation. Because inflammation plays a key role in the
severity, course and severity of LP, inflammatory markers have the
potential to improve current diagnosis and prognosis methods [2].
Serum amyloid A (SAA) is a conserved 12‐kDa protein produced
by hepatic and extrahepatic tissues. It is significantly related to the
acute phase response being higher in serum by up to 1000 folds
within 24 hours of the start of inflammation [3].
Various chronic inflammatory diseases, like rheumatoid arthritis
and metabolic syndrome, prolonged elevation of SAA may contribute to tissue damage and degradation and eventually can lead to the
development of secondary amyloidosis [4].
Several biological roles of SAA are shown by different studies
including immunological, inflammatory, and homeostatic functions.
Serum amyloid A have also antimicrobial properties against a wide
spectrum of organisms as bacteria, viruses, and fungi [5]. Moreover,
it has been shown that SAA protein is involved in the pathogenesis
of several inflammatory diseases including rheumatoid arthritis [6]
psoriases, [7] coronary artery disease [8], and Urticaria [9]
Interleukin-6 (IL-6) is one of the most important inflammatory
cytokines. It was originally described as B-stimulating factor 2, which
induces B lymphocytes to produce immunoglobulin. It can be rapidly
synthesized in the case of infection and tissue damage. Relevant data
showed that IL-6 plays a key role in the pathogenesis of inflammatory
diseases [10].
This study was carried out to detect the expression of SAA and
IL-6 in the peripheral blood serum of LP patients through experiments
to provide accurate basis for future clinical diagnosis and treatment
of LP.
Patients and Methods
This study was approved by the Institutional Review Board of the
Faculty of Medicine, Zagazig University, Egypt. Informed consent
was obtained from each participant. All the data were kept private
after assigning a code number to every participant, only known by
the researchers.
This case-control study was conducted at Zagazig University
Hospital between February to December 2020. Data were collected
consecutively from 21 adults, clinically proven, lichen planus
patients admitted to the Dermatology, Venereology and Andrology
Department, Faculty of Medicine, Zagazig University, Egypt. The
control group involved 21 healthy individuals. Participants with
autoimmune disorders, infectious disease, hepatic, malignancy, those
treated for lichen planus during the previous 2 months of recruitment,
pregnant females as well as participants with positive covid-19 within
the past 3 months were excluded from the study.
All the study participants were subjected to detailed history
taking, thorough physical examination, and measurement of serum
SAA. In addition, LP severity was assessed for all LP patients.
Estimation of SAA levels, IL 6 by enzyme-linked immunosorbent assay:
Serum levels of SAA, IL 6 was estimated based on ELISA Kit for in
vitro quantitative measurement according to the producer protocol
(SunRedBio, Shanghai, China). Five-milliliter venous blood samples
were drawn into a sterile vial from all patients and controls. The
clotted blood is then centrifuged at 3,000 rpm for 10 minutes. Then
serum was transferred to labeled tubes and stored at -80 until assay.Assessment of disease severity for the involved cases:
Cutaneous LP: severity assessment was done through calculation
of the affected body surface area (BSA) which was measured by using
the total palmar surface of the hand, including the five digits, which
is approximately 1% of total BSA. Patients were classified on basis of
BSA into mild (<3% BSA affected), Moderate (3-10% BSA affected),
and severe (>10% BSA affected) categories. This scale is primarily
used for the classification of psoriasis and no similar scale was found
for LP, it was useful practically and clinically for this study as well [11]Severity assessment of OLP was done using a semiquantitative
scoring system based on the site, area, and presence of erosions,
known as the clinical scoring system of OLP. It divides the mouth
into eight areas with a score of severity between 0 and 2. Grading,
based on scores, is divided into grades 0,1,2, and 3. Determination of
severity, based on grade, is either mild, moderate, or severe disease
[12].
Assessment of LP activity was done by progression surveillance,
and it was classified according to the emergence of new lesions or
progression of current lesions over the last 6 weeks to 1 year [13].
Results
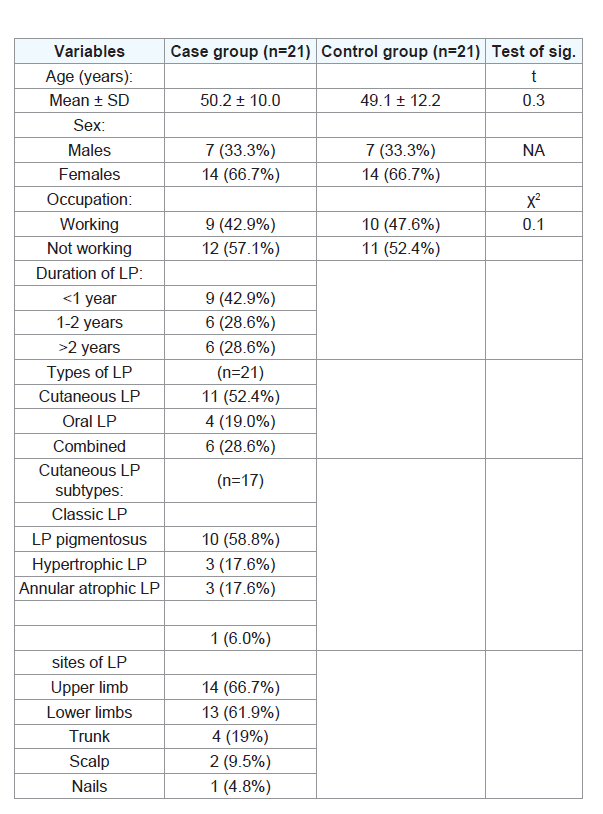
Various features of the study participants are shown in (Table 1).
The cases group included 14 (66.7%) were females and 7 (33.3%) were
males. The range of age was between 29 and 68 years. The control
group had 14 females (66.7%) and 8 males (33.3%), and their ages
ranged between 28 and 65 years. The length of LP duration ranged
from 1 week to 5 years. Lesions were found on the upper extremities,
lower limb, trunk, scalp, nails, and oral mucosa in 14, 13, 4, 2, 1, 10
patients, respectively.
The mean serum levels of SAA were significantly higher in
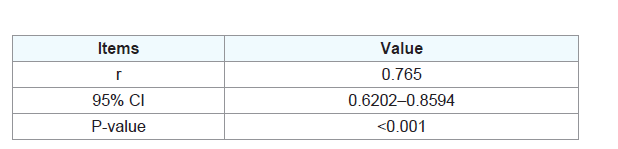
patients than their healthy controls (P < .02). Pearson correlation
analysis showed that SAA level in serum of patients was positively
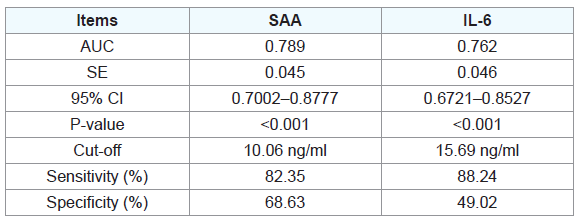
correlated with IL-6 .When the cut-off value was 10.06 ng/ml, the
sensitivity of SAA to LP severity was 82.35% and the specificity
was 68.63%. When the cut-off value was 15.69 ng/ml, the sensitivity
of IL-6 to LP severity was 88.24% and the specificity was 49.02%
(Table 2,3).
Discussion
Lichen planus skin inflammation enhances SAA expression, and
vice versa, which may contribute to the exacerbation of skin lesions,
the elevation of serum SAA levels, and the development of systemic
complications such as atherosclerosis in lichen patients.
SAA is a major acute plasma protein, which can regulate innate
immunity and cholesterol homeostasis. SAA has a significant
relationship with acute phase reaction. Serum level rises up to 1,000
times within 24 h [5. This is the same as C-reactive protein (CRP).
SAA can be used as a diagnostic, prognostic or therapeutic followup
marker for many diseases. Cytokines are effective inducers of
SAA in hepatocytes[6]. Relevant literature shows that the synthesis
of SAA is regulated by IL-6 [7]. IL-6 is a multi-effect cytokine with known multiple functions in immune regulation, inflammation, and
tumorigenesis [10]. Biological medicines for inflammatory cytokine
IL-6 are increasingly considered as treatment methods for chronic
diseases and cancers [2]. However, the role of IL-6 in LP is still less
elaborated. Therefore, analyzing the impact of SAA and IL-6 on LP,
is not only of great significance for future clinical screening of LP, but
also provides new ideas for potential therapeutic targets of LP in the
future.
The results of this study showed that the expression levels of SAA
and IL-6 were significantly up-regulated in LP patients, suggesting
that SAA and IL-6 may participate in the development and severity
of LP. According to Pearson correlation analysis, SAA level in serum
of patients was positively correlated with IL-6 (r= 0.765, P<0.001),
which shows serious tissue damage in LP severity. In psoriasis, SAA
was considered as a more specific marker of psoriasis rather than
C-reactive protein. 16 SAA protein levels in psoriasis patients’ sera
were reported to have a positive correlation with the Psoriasis Area
and Severity Index scores (PASI score) [14-16].
At this time, the content of pro-inflammatory cytokine IL-16
increases significantly and regulates the accelerated secretion of SAA.
By drawing ROC curve of SAA and IL-6, we found SAA AUC=0.789,
95% CI, 0.7002–0.8777, while IL-6 AUC=0.762, 95% CI, 0.6721–
0.8527. This showed that SAA and IL-6 have a very good predictive
value in the prediction of LP severity.
We found an increase in serum levels of SAA, IL6 in LP patients.
However, further studies on larger scales in different populations are
still needed to validate our results and to investigate SAA levels in the
skin lesions of different forms of LP.
Conclusion
In conclusion, the levels of SAA and IL-6 are significantly
increased in sever LP and they are positively correlated. They
may participate in the development and progression of LP and can
effectively predict and affect the progress of LP.
References
Citation
Metwalli M, Ibraheem AH, Abu bakr H, Fathia MK. Serum Amyloid A as an Inflammation Marker in Lichen Planus. J Clin Investigat Dermatol.
2021;9(1): 3