Journal of Clinical and Investigative Dermatology
Download PDF
Case Report
Rapid Clearance of Erythrodermic Psoriasis with Acitretin in an Unstable Case of Psoriasis under Treatment with Secukinumab
Ntavari N, Roussaki- Schulze AV and Zafi riou E*
Department of Dermatology, University of Thessaly, Greece
*Address for Correspondence: Zafiriou E, Faculty of Medicine, Department of Dermatology, General University Hospital of Larissa Biopolis Larissa 41110, Greece, Fax: 30-2413501337; E-mail: zafevi@hotmail.com
Submission: 31 January, 2021;
Accepted: 2 March, 2021;
Published: 5 March, 2021
Copyright: ©2021 Ntavari N, et al. This is an open access article distributed
under the Creative Commons Attribution License, which permits unrestricted
use, distribution, and reproduction in any medium, provided the original work
is properly cited.
Abstract
Erythrodermic Psoriasis (EP) is a rare, severe and disabling form of psoriasis
in children and adults. Clinical characteristics of this subtype of psoriasis include
a diffuse erythema involving at least 75% of the body surface area, oedema, itch,
scaling, hair loss, onychodystrophy, palmoplantar keratoderma and furthermore
systemic findings. The triggers of erythrodermic psoriasis are an abrupt
extraction of anti-psoriatic drugs such as methotrexate and topical steroids,
systemic illness (e.g. HIV, infections), ultraviolet burns, drug reaction, abuse of
alcohol, and emotional stress. Erythrodermic psoriasis may occur in association
with life-threatening complications such as super infections and sepsis from skin
pathogens such as Staphylococcus aureus and Streptococcus species, hypovolemic
shock and acute kidney injury secondary to skin fluid loss, severe anemia, acute
respiratory distress syndrome, hydroelectrolytic abnormalities and protein loss.
Despite the plethora of treatment options for plaque psoriasis the management
of erythrodermic psoriasis remains a challenge. Here we report a case of complete
and rapid resolution of erythrodermic psoriasis with acitretin, demonstrating its
efficacy for controlling the occurrence of erythrodermic flares, in a patient with
moderate-to-severe plaque psoriasis effectively treated with secukinumab until
then. Even today, in the era of biological agents, acitretin still remain a valuable
treatment option for resistant and difficult to treat erythrodermic psoriasis.
Abbreviations
EP: Erythrodermic Psoriasis; PASI: Psoriasis Area Severity Index; BSA: Body Surface Area; DLQI: Dermatology Life Quality Index; NAPSI: Nail Psoriasis Severity Index
Introduction
Erythrodermic Psoriasis (EP) is a rare, severe and disabling form
of psoriasis in children and adults with an estimated prevalence
among psoriatic patients ranging from 1%-2.25% [1]. Th is subtype
of psoriasis presents with distinct clinical characteristics, which
include a diff use erythema involving at least 75% of the body surface
area. Other authors argue that there must be aff ected at least 90%
of the body surface area [2-4]. EP can manifest with oedema, itch,
scaling, hair loss, onychodystrophy, palmoplantar keratoderma
and furthermore systemic findings [1,4,5,]. The pathogenesis of EP is not well understood; although, numerous studies advocate that
the disease is associated with a predominantly T helper 2 (Th2)
phenotype [6,7]. Th e triggers of erythrodermic psoriasis are an
abrupt extraction of anti-psoriatic drugs such as methotrexate and
topical steroids, systemic illness (e.g. HIV, infections), ultraviolet
burns, drug reaction, abuse of alcohol, and emotional stress [1,2].
EP may occur in association with life-threatening complications
such as superinfections and sepsis from skin pathogens such as
Staphylococcus aureus and Streptococcus species, hypovolemic shock
and acute kidney injury secondary to skin fluid loss, severe anemia,
acute respiratory distress syndrome, hydroelectrolytic abnormalities
and protein loss [2,3,5,8]. Th e management of this condition is based on the use of anti-infl ammatory, immunosuppressive and biologic
agents following the supportive care [1].
Acitretin is a second-generation retinoid used in the treatment
of severe resistant psoriasis. Experts support the use of acitretin
as a first-line therapy in stable cases of erythrodermic psoriasis,
although it typically works slowly [4]. We report a case of complete
and rapid resolution of EP with acitretin, demonstrating its efficacy
for controlling the occurrence of erythrodermic flares, in a patient
with moderate-to-severe plaque psoriasis effectively treated with
secukinumab until then (PASI 100).
Case report
A 34-year-old man with generalized erythema and extensive
scaling presented to our dermatology department. The patient had
a 20-year history of moderate-to-severe plaque psoriasis vulgaris
(baseline PASI score 29.0), without concomitant arthritis, and was
successfully treated with secukinumab in the last year (PASI score
0). He did not have any other new drug exposures and did not use
topical medications other than moisturizers, in the last 3 months before and during the episode. Upon evaluation, he was tachycardic and febrile (38 ºC). On physical examination, the involved Body
Surface Area (BSA) was approximately 90% with a Psoriasis Area and
Severity Index (PASI) score of 40.7 accompanied by psoriatic nail
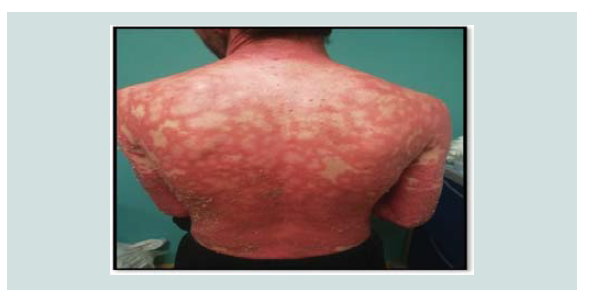
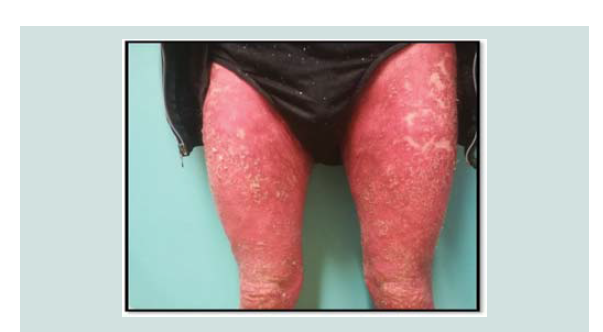
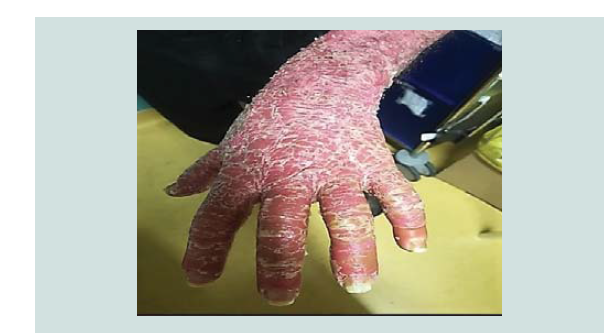
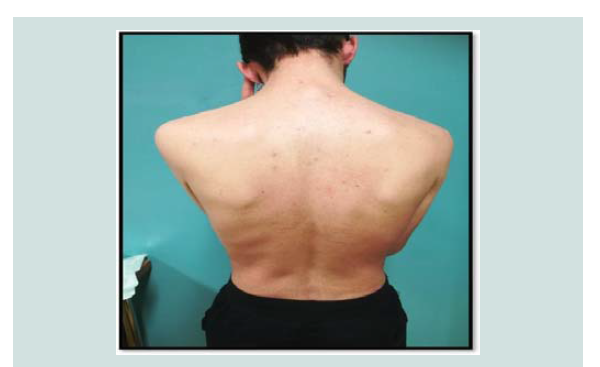
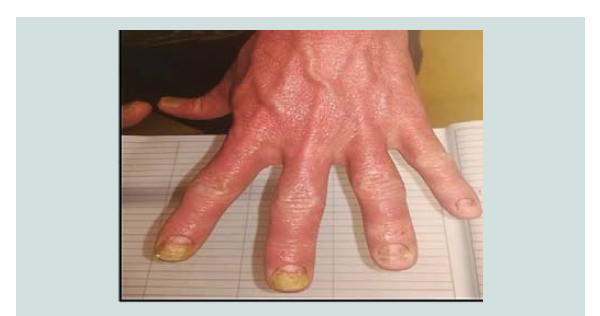
disorders especially onycholysis on his finger and toenails (NAPSI score 30.0) (Figure 1-3). Mucosal involvement and Nikolsky sign were absent. Th is condition had an important impact on his quality of life (DLQI score was 20.0). Cardiovascular, respiratory, neurological, lymphatic and musculoskeletal investigations were normal. Our clinical diff erential diagnosis included drug-induced erythroderma, erythrodermic psoriasis and cutaneous T-cell lymphoma; less
likely was any other cause of erythroderma. Τhere was no evidence of atopic dermatitis from the history and clinical examination. Laboratory evaluation was signifi cant for leukocytosis (12,850/μL)
with neutrophilia (9,690/μL), elevated C-reactive protein (8.3 mg/dL)
and erythrocyte sedimentation rate (25 mm/h). Eosinophils (1.4%),
serum calcium (6.9 mg/dL) and total serum IgE levels (15 UI/ml)
were normal. Urine culture revealed a clear finding of urinary tract
infection with Pseudomonas aeruginosa. The initial management of
our patient’s conditions included a discontinuation of secukinumab,
fluid resuscitation and antibiotic treatment for the urinary tract
infection. Punch biopsy demonstrated acanthosis, mild spongiosis
and hyperkeratosis. Perivascular infi ltrates in the derma consisted of
lymphocytes, macrophages and fibroblasts. Clinical, laboratory, and
histological findings were suggestive of the diagnosis erythrodermic
psoriasis. Administration of systematic treatment with acitretin 25
mg per day was decided. At 2 weeks, a clinical improvement was observed. Erythema, scaling and itching showed an excellent and rapid response, signifi cantly reducing the mean PASI score from
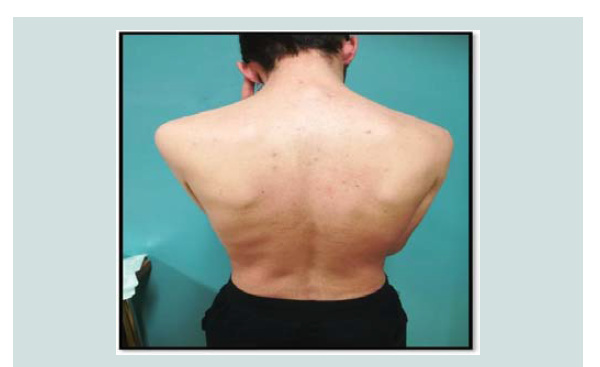
baseline to 16.1. Within 5 weeks of acitretin therapy, complete
clearance of all the skin was achieved (Figure 4 and 5), with a PASI
score of 0 (PASI 100). In addition, an important amelioration on his
impact quality of life (DLQI score: 2.0) was notable. Nail dystrophy
and onycholysis delayed recovery and fully recovered 12 weeks aft er
the start of acitretin (Figure 6). The patient remains clear of psoriasis
lesions with ongoing acitretin therapy aft er 16 weeks, with a tapering
of acitretin while transitioning to the precedent biologic agent.
Figure 1: Erythroderma affecting the whole body. Generalized erythema and extensive scaling, before treatment with acitretin.
Figure 2: Erythroderma affecting the whole body. Generalized erythema and extensive scaling, before treatment with acitretin.
Discussion
Erythrodermic psoriasis is a dermatologic emergency, may be
associated with serious morbidity and mortality [1,9]. Unfortunately, the management of EP is difficult due to limited available data leading
to the choice of appropriate treatment. The initial approach to the
management of EP should include fluid, nutritional and electrolyte
replacement; impaired thermo regulation; keep the skin moisture
with wet dressings, oatmeal baths, emollients and low-potency topical
steroids; treatment of secondary infections [10]. It is important to
mention that the decision about the optimal treatment regimen is
difficult and should be dictated by complete evaluation of the severity
of disease and each individual patient’s underlying comorbidities.
According to a consensus guideline of the National Psoriasis
Foundation Medical Board, regarding the appropriate management
of EP, conventional systemic therapies, such as cyclosporine,
methotrexate, and acitretin, were used as first-line therapy in
unstable cases (cyclosporine) and in more stable cases (methotrexate/acitretin) [10-12]. A meta-analysis of 12 patients receiving 25-35 mg/day of acitretin proves the efficacy of this systemic agent in EP. In 83.3% of the cases achieved an important improvement of EP [13]. Mycophenolate mofetil is another immune suppressant that was used successfully in two patients with severe EP [14].
Figure 4: Clearance of erythrodermic psoriasis 5 weeks after onset of treatment with acitretin 25 mg/d.
Figure 5: Clearance of erythrodermic psoriasis 5 weeks after onset of treatment with acitretin 25 mg/d.
Figure 6: Complete clearance of skin psoriasis but delayed recovery of nail dystrophy and onycholysis 5 weeks after onset of acitretin 25 mg/d.
Th e TNF-α inhibitors agents reviewed in EP, with satisfactory
results, include etanercept, adalimumab, infl iximab, and golimumab [10,15,16]. According to a multicentre national retrospective study of 28 patients, in the French Psoriasis Group network, representing 42 flares of erythrodermic psoriasis used successfully anti-TNFa
alongside ustekinumab (anti-IL12/23) and efalizumab (Mab IgG1)
[16]. Review of the literature suggests that infliximab and ustekinumab may be decided on as first-line therapy, as a result of the rapidity of
clearance and the excellent safety profile [3,17]. Ixekizumab, an anti-IL-17A monoclonal antibody, confirmed a high level of efficiency
in management of EP in accordance with an open-label study in a small number of patients in Japan [18]. Another anti-IL-17 agent, secukinumab may therefore also be an eff ective and safe option in the treatment of this severe disease [19,20]. A case series and a case report
have been published regarding the use of alefacept and panitumumab (anti-EGFR antibody) in EP, respectively. Both, exhibited significant therapeutic eff ects on skin symptoms in the erythrodermic psoriasis patients [21,22].
Conclusion
Erythrodermic psoriasis is a severe variant of psoriasis and the
choice of appropriate treatment remains a challenge. Over the years,
biologic agents have revolutionized the management of erythrodermic
psoriasis, giving us alternative therapeutic options. On the other
hand, however, traditional systemic therapies, with their oral route of
administration and low cost, form an important ally in the treatment
of this rare and potentially lethal form of psoriasis. Even today, in
the era of biological agents, acitretin still remain a valuable treatment
option for resistant and difficult to treat erythrodermic psoriasis.
References
Citation
Ntavari N, Roussaki- Schulze AV and Zafiriou E. Rapid Clearance of Erythrodermic Psoriasis with Acitretin in an Unstable Case of Psoriasis under Treatment with Secukinumab. J Clin Investigat Dermatol. 2021;9(1): 3