Journal of Clinical and Investigative Dermatology
Download PDF
Case Report
Primary Classic Kaposi’s Sarcoma Of Lymph Nodes In Ultrasound Resembling Lymphatic Metastases Of A Malignant Tumor And Successful First-Line Therapy With Ipilimumab
Kraehnke J*, Bogumil A, Grabbe S, Loquai C, Weidenthaler-Barth B and Tuettenberg A
Department of Dermatology, University Medical Center Mainz, Germany
*Address for Correspondence: Kraehnke J, Department of Dermatology, University Hospital Mainz, Langenbeckstrasse 1, 55131 Mainz, Germany, Tel: + 49 61 31 17 0; Fax: + 49 61 31 34 99; E-mail: juliane.kraehnke@unimedizin-mainz.de
Submission: 10 November, 2019
Accepted: 09 December, 2019
Published: 14 December 2019
Copyright: © 2019 Kraehnke J, et al. This is an open access article
distributed under the Creative Commons Attribution License, which permits
unrestricted use, distribution, and reproduction in any medium, provided the
original work is properly cited.
Abstract
We report on a 71-year-old patient with a classic, primary Kaposi’s sarcoma of
the lymph nodes. He presented with an inguinal lymphadenopathy. An ultrasound
examination showed suspicious lesions (oval lymph nodes with a single, round,
homogeneous hypoechoic structure with peripheral and diffuse vascularization),
yet typical sonomorphological criteria for lymph node metastases of a solid
malignant tumor such as melanoma (round lymph nodes with a round hypoechoic
area that often contains hyperechoic structures due to necrosis and a peripheral
vascularization) were not fully met. Nevertheless, due to the suspicious findings,
inguinal lymph nodes were extirpated. Histologically, there were formations of
a malignant tumor that led to the diagnosis of a nodal Kaposi’s sarcoma. Skin
examinations, HIV testing and imaging procedures presented no pathological
findings. A therapy with ipilimumab (3mg/kg every three weeks for four times)
was initiated, which resulted in durable tumor control.
Sonomorphological patterns for Kaposi’s sarcoma of the lymph nodes are
not yet defined. We report on this case to describe the specific sonomorphological
structures in our patient as a first step towards establishing sonomorphological
criteria for nodal Kaposi’s sarcoma. Furthermore, this is the first case to report on
a successful first-line therapy with the immune checkpoint inhibitor ipilimumab in
classical Kaposi’s sarcoma.
Abbreviations
KS: Kaposi’s Sarcoma
Introduction
Kaposi‘s Sarcoma (KS) is a rare, malignant, angioproliferative,
multifocal systemic disease. Angioproliferative tumors can occur
in the skin, mucous membranes, lymph nodes and other organs.
It has been classified into four sub-types: classic (typically presents
in middle or old age in Mediterranean or eastern European men),
endemic (in indigenous Africans), iatrogenic (associated with
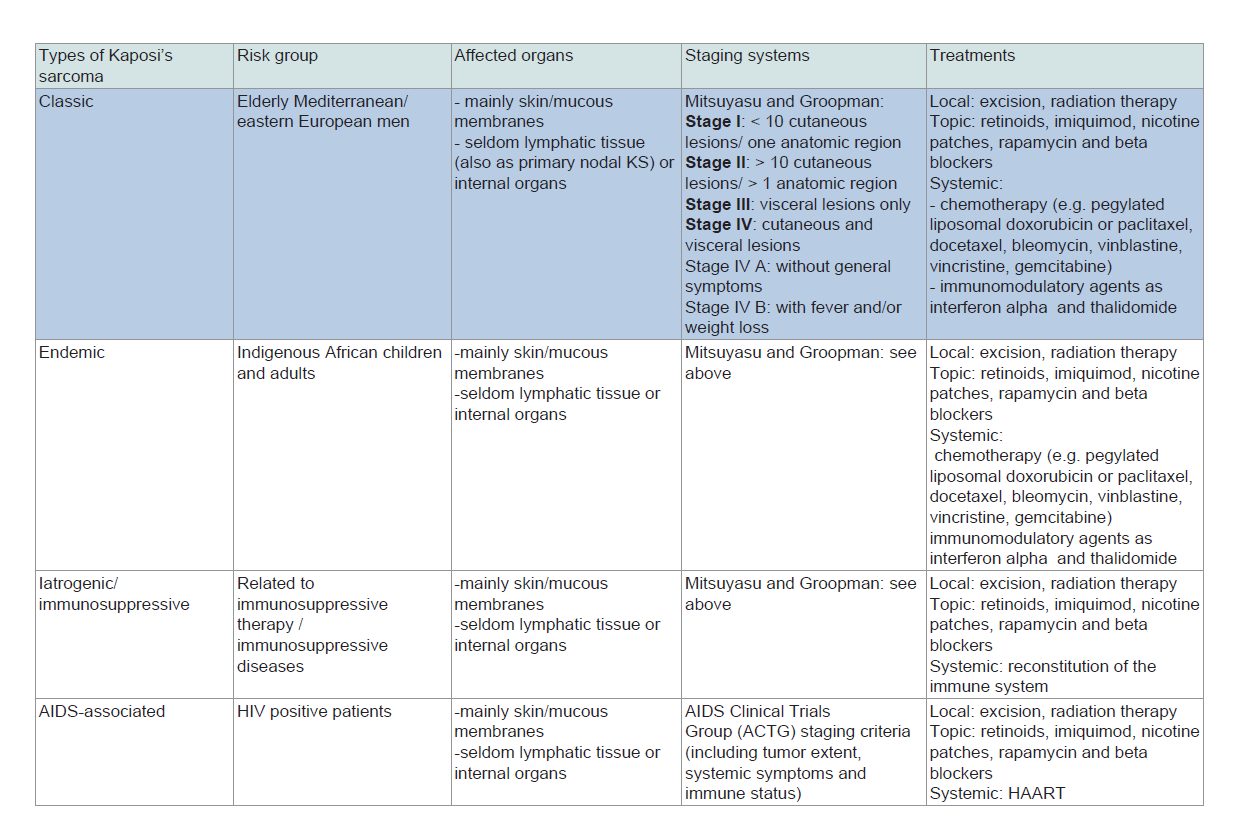
immunosuppressive therapy) and AIDS-associated KS (Table 1). In
about 90% of cases it is associated to human herpes virus-8 (HHV-
8), also known as KS-Associated Herpes Virus (KSHV) [1-3]. In this paper we will focus on classic KS since our patient suffered from a rare subtype of it: a primary nodal classic KS.
Usually, the suspected diagnosis is based upon the appearance
of skin lesions that vary from reddish blue to dark brown macules,
plaques or nodules. KS can be confirmed by histological examination
of the affected tissue, which typically shows evidence of angiogenesis
with thin-walled cleft-shaped vascular spaces, inflammation
(lymphocytes, plasma cells and macrophages) and spindle cell
proliferation [4-6]. In nodular lesions, tumors are more solid and
there are large fascicles of spindle-shaped endothelial cells with fewer
and more compact vascular slits. The mononuclear cellular infiltrate
decreases [4-6]. The histological diagnosis of nodal involvement is
complicated. At early stages there are similarities to reactive processes.
Only later in the course of the disease do spindle-shaped cells become
prominent [7]. Cells of the vascular structures and spindle cells stain
for endothelial markers such as CD31, CD34 and ERG as well as for
D2-40/Podoplanin. Immunohistochemical staining can be done to
detect the presence of HHV-8-LNA (latent nuclear antigen) within
the tumor cells or polymerase chain reaction can be performed
to detect HHV-8-DNA [6-8]. Kaposi sarcoma is the only vascular
tumor with positivity for HHV-8.
An important aspect during initial examination is the palpation
of lymph nodes to check for nodal involvement. Additionally, an
ultrasound of lymph nodes should be done to rule out nodal spreading.
To our best knowledge, there are no published sonomorphological
criteria of affected lymph nodes to date.
Further screening for distant organ involvement is dependent
on the subtype. In the case of asymptomatic patients with classic
KS, a radiographic evaluation is not normally needed due to the low
frequency of radiographically evident metastatic disease [9]. Patients
with gastrointestinal symptoms (e.g. bleeding, diarrhea, proteinlosing
enteropathy, perforation) should undergo an endoscopy to
check for visceral mucosal involvement [9-11]. A staging system
for classic KS, proposed by Mitsuyasu and Groopman (1998), is
commonly used. The disease is grouped into stage I-IV based upon
disease distribution and clinical symptoms (Table 1). The course of the disease is dependent on the subtype. Classic
Kaposi’s sarcoma mostly has a chronic, indolent course and rarely
impacts survival. Sometimes, however, it has an acute onset with
rapid progression or a previously indolent disease can undergo
sudden worsening, causing significant morbidity and mortality [12-14]. In 5-20% of cases there is a mucosal and/or organic involvement
[15].
Table 1: Four variants of Kaposi’s sarcoma. We focus on classic Kaposi’s sarcoma (primarily nodal) in this paper.
Since there is no treatment to cure classic KS, the treatment
generally aims to achieve symptom palliation, improving function
and delaying or preventing progression [9]. Local therapy includes
excision even though local recurrences are common [15]. KS
is radiosensitive, so radiation therapy of cutaneous tumors is a
good option. Topical therapy with retinoids, imiquimod, nicotine
patches, rapamycin and beta blockers is only marginally efficient
[9]. Systemic treatments such as chemotherapy are used in cases
of rapid progression or multilocular disease. Standard treatment
includes pegylated liposomal doxorubicin or paclitaxel, docetaxel,
bleomycin, vinblastine, vincristine, gemcitabine, and etoposide
as second line [9,16,17]. However, chemotherapy is mostly
palliative. Immunomodulatory agents as interferon alpha and
immunomodulatory imide drugs (as thalidomide) have been used
in single cases with variable efficacy [16,18]. Recently published case
reports indicate that a therapy with anti-PD-1 agents might be a new
and successful therapy option [17,19,20].
Case Report
We report on a 71-year-old patient with a classic, primary
Kaposi’s sarcoma of the lymph nodes. Due to palpable inguinal
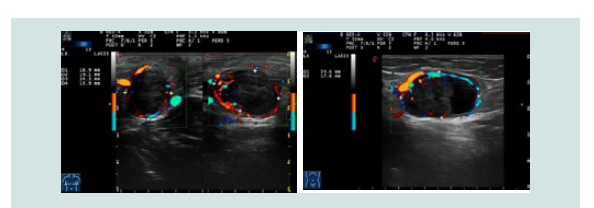
lymphadenopathy the patient presented at the clinic. On ultrasound,
lymph nodes presented suspicious being hyperechoic, oval shaped
with a single, round, homogeneous hypoechoic structure. The
hypoechoic structure showed a strong peripheral and diffuse
vascular pattern (Figure 1). These sonographic findings, albeit not
entirely typical, resembled those of lymph node metastases which
regularly present as hyperechoic, round shaped lymph nodes with
a round, nonhomogeneous hypoechoic area due to necrotic parts
of the metastases and a mainly peripheral vascularization (Figure 2). However, due to missing hyperechoic parts within the round,
hypoechoic structure and a strong diffuse vascularization, criteria
for lymph node metastases were only partially met. Furthermore, the
bilateral appearance of suspicious lymph nodes at the same time was
not impossible but uncommon for metastases. The presenting lymph
nodes seemed to be a mixture of metastases (due to the hypoechoic
structure with peripheral perfusion) and reactive lymph nodes (due
to the additional, strong diffuse perfusion). Due to these ultrasound
findings, imaging was extended. A CT showed soft-tissue masses,
unknown pulmonary lesions and suspect inguinal, iliac and axillary
lymph nodes. A bilateral extirpation of inguinal lymph nodes was
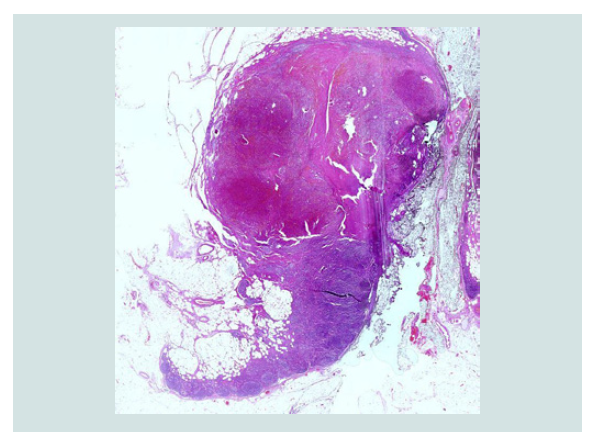
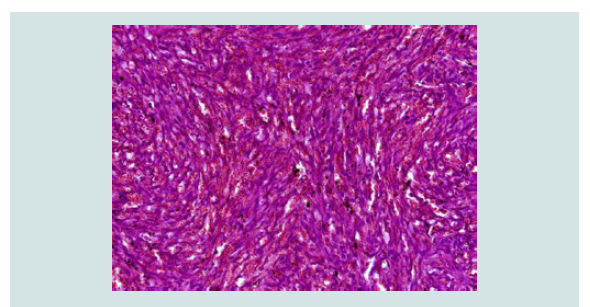
done. Histologically, there were formations of a malignant tumor
with siderosis and a mesenchymal proliferation of atypical spindleshaped
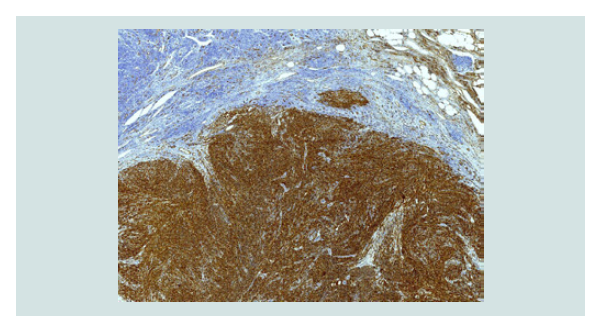
endothelial cells (Figure 3 and 4). The vascular markers CD34,
CD31, D2-40/Podoplanin as well as HHV8 showed a positive result
in the immunohistochemical staining (Figure 5 and 6). Histologically,
the diagnosis of a Kaposi’s sarcoma was rendered. Skin examinations,
HIV testing and blood tests that were done to rule out other organ
manifestations and immunodeficiency were without pathological
findings. An esophagogastroduodenoscopy was performed due to
intermitting sickness. A gastritis was diagnosed but there was no
sign of mucosal tumor lesions. The patient also had not received
any immunosuppressive therapy. Therefore he was diagnosed with a
classic primary Kaposi’s sarcoma of the lymph nodes, an exceptionally
rare sub-type of classic KS since in most cases the diagnosis is
rendered due to skin lesions (i.e. not primarily nodal). A therapy with
ipilimumab (3mg/kg body weight, 4 cycles, every 3 weeks) was given
from June to August 2014. It was well tolerated except for a transient
hypophysitis as an immune-related adverse event. The last staging
was done in 2019 and showed ongoing regressive lymph nodes and
no other pathological findings. The soft tissue masses that were
apparent upon primary staging had also fully resolved after therapy
with ipilimumab. Thus no further therapy was initiated up to now.
Discussion
The main and so far undescribed aspects of this case report are
sonomorphological criteria in KS of the lymph nodes and the therapy
with the immune checkpoint inhibitor ipilimumab in classic KS.
The use of ultrasound as a non-invasive imaging procedure
is performed in several skin diseases [21]. Nevertheless, there is
little data available regarding the ultrasound of KS lesions. Only
cutaneous lesions have been characterized a few times. Herein, the
authors describe partly contradictory sonomorphological patterns.
In 1993, Bogner et al. reported on skin lesions as hypoechoic,
with a homogeneous structure and well-defined outlines whereas
Cammarota described solid [22], nonhomogenous nodules with
poorly defined outlines [23]. In 2015, Carrascosa et al. described
details on the sonomorphological structure and vascularization of
cutaneous tumor lesions [24]. The pattern revealed by a B-mode
ultrasound was solid and hypoechoic and lesions were located in the dermis. Nodules were more homogeneous, with regular, well-defined
outlines, whereas plaques were less homogeneous with irregular
edges and less well-defined outlines [24]. The color doppler showed
intralesional vascularization that was prominent at the lower pole in
nodules and little or absent vascularization in plaques [24].
To our best knowledge, there are no published sonomorphological
criteria of affected lymph nodes in KS to date. As described, lymph
nodes in our patient presented hyperechoic, oval shaped with
a round, homogeneous hypoechoic structure with a peripheral
and diffuse vascular pattern in ultrasound (Figure 1). Therefore,
sonomorphological criteria overlap with those of e.g. melanoma
metastases. Nevertheless it was not possible to determine the tumor
entity by ultrasound only especially since there are no described
specific sonomorphological patterns of nodal involvement in KS.
Thus, it would be of great interest to include ultrasound sonography
in today’s routine staging for KS patients (i) in order to define in
more detail lymph node involvement in KS and (ii) to establish
sonomorphological criteria. This is desirable not only to improve
nodal diagnostics in patients with known KS but also to evaluate
therapeutic responses. Nevertheless, a histological examination of the
suspect tissue is important. This case demonstrates the importance of
combining ultrasound and histology to differentiate between various
malignancies. As described above, there is no curative systemic treatment of
advanced classic KS to date. Systemic chemotherapy with liposomal
doxorubicin, which is used in a progressive disease, achieves mostly
transient responses. A literature review of systemic treatment was
done by Régnier-Rosencher et al. in 2013, who described response
rates of various treatments from 43-100% but found that evidence
for efficacy of any particular therapy was of low quality and a specific
therapeutic strategy could not be recommended [16].
As described above, there is no curative systemic treatment of
advanced classic KS to date. Systemic chemotherapy with liposomal
doxorubicin, which is used in a progressive disease, achieves mostly
transient responses. A literature review of systemic treatment was
done by Régnier-Rosencher et al. in 2013, who described response
rates of various treatments from 43-100% but found that evidence
for efficacy of any particular therapy was of low quality and a specific
therapeutic strategy could not be recommended [16].
Immune checkpoint inhibition has been proven to be effective in
numerous malignancies, including virally mediated tumors such as
merkel-cell carcinoma (MCpV-associated) or Hodgkin‘s lymphoma
(EBV-associated) [25,26]. Oncovirus associated tumors, including
KS, seem to have a virally mediated increased expression of PD-L1
[25,26]. Contrary to these findings, higher-than-anticipated response
rates were also described in virus associated tumors with low PD-L1 expression, suggesting that also the presentation of viral antigens on
tumors may lead to an increased response rate to immunotherapy
[26].
Our patient is the first published case of a classic, primary nodal
Kaposi’s sarcoma that was treated with immunotherapy. The abovementioned
case reports describe patients either suffering from classic,
endemic or HIV-associated KS and receiving immunotherapy with
the anti-PD-1/PD-L1-agents pembrolizumab or nivolumab. Here, we
report on treatment with the CTLA-4-antibody ipilimumab that led
to a stable disease (regressive soft tissue masses and axillary, iliac and
inguinal lymph nodes). After a follow-up period of five years, we have
no sign of disease progression in our patient. Therefore, we conclude
that the therapy with ipilimumab was successful and led to a longterm
stable disease. Especially since we would have expected a disease
progression in this case of classic nodal KS (i.e. that is not limited to
the skin) without immunotherapy. The result supports the suggestion
that immunotherapy might be a promising therapy option in KS and
shows that it might even lead to a long-term stable diseases or even
complete response [17,19,25,26,31]. Furthermore, the use of immunotherapy
as first-line therapy has not been reported before. Since usually used
systemic therapies mostly show only a transient success, this would
be a new, life-saving option for patients with advanced KS. KS in
general often affects immunosuppressive patients. Chemotherapies
often additionally lead to myelosuppression as a side effect. Studies
with immune checkpoint inhibitors on a greater number of patients
are necessary to prove efficacy. The National Cancer Institute is
conducting a prospective study of combined ipililmumab and
nivolumab in patients with HIV associated cancers, including
Kaposi’s sarcoma (Clinical-Trials.gov, Identifier: NCT02408861);
final data will presumably be available at the end of 2020. The use of
immunotherapy as first-line therapy is desirable in order to replace
marginally effective chemotherapies.
Figure 3: HE staining at 40x magnification: Mesenchymal proliferation in the upper half of the lymph node.
Figure 4: HE staining at 400x magnification: Mesenchymal proliferation of atypical spindle-shaped endothelial cells forming vascular spaces; extravascular erythrocytes.
Conclusion
Sonomorphological criteria for KS of the lymph nodes are not
yet known. In our patient affected lymph nodes showed a round,
homogeneous hypoechoic structure with a peripheral and diffuse
vascular pattern in ultrasound leading to further diagnostics. Since
nodal involvement is rare, it is desirable to publish cases like this
one in order to develop a more profound and specific knowledge of
sonomorphological criteria for KS of the lymph nodes and thereby
improve and complement diagnostics as well as therapy evaluation.
Recently published data report on successful treatment with
pembrolizumab and nivolumab in single cases of endemic and HIV associated
KS when used as second- or third-line therapy [17-19].
Considering the knowledge of immunomodulation of KS [28-30], the
positive response rate to immune checkpoint blockage in other virus
associated immunomodulated tumors such as merkel-cell carcinoma
or Hodgkin’s lymphoma and the positive response to immune
checkpoint inhibitors in recently documented cases [17,19,25,26,31],
it is very likely that ipilimumab has resulted in an ongoing stable
disease in our patient. This case shows that immunotherapy seems
to be a promising option in patients with classic, primary nodal KS.
It supports the positive data about the efficacy of immunotherapy in
KS due to a first-time reported follow-up of five years. Furthermore
it indicates that anti-CTLA-4 therapy is a possible alternative if anti-PD-1 therapy is not applicable.
References
Citation
Kraehnke J, Bogumil A, Grabbe S, Loquai C, Weidenthaler-Barth B, et al. Primary Classic Kaposi’s Sarcoma Of Lymph Nodes In Ultrasound Resembling Lymphatic Metastases Of A Malignant Tumor And Successful First-Line Therapy With Ipilimumab. J Clin Investigat Dermatol. 2019;7(2): 5