Journal of Clinical and Investigative Dermatology
Download PDF
Review Article
*Address for Correspondence: Tongyu C. Wikramanayake, Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine, 1600 NW 10th Avenue, RMSB 2023A, Miami, Florida 33136, USA, Tel: (305) 243-8878; Fax: (305) 243-3082; E-mail: tcao@med.miami.edu
Citation:Borda LJ, Wikramanayake TC. Seborrheic Dermatitis and Dandruff: A Comprehensive Review. J Clin Investigat Dermatol. 2015;3(2): 10.
Copyright © 2015 Borda LJ, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reviewed & Approved by: Dr. Craig G Burkhart, Clinical Professor, Department of Medicine, Ohio University, USA.
journal of Clinical & Investigative Dermatology | ISSN: 2373-1044 | Volume: 3, Issue: 2
Submission: 01 December 2015| Accepted: 11 December 2015 | Published: 15 December 2015
In infants, SD may present on the scalp, face, retro-auricular area, body folds, and trunk; rarely it may be generalized. Cradle cap is the most common clinical manifestation. SD in children is usually self-limited [3,15]. On the other hand, in adults, SD is a chronic or relapsing condition, featured by erythematous patches, with flaky, large, oily or dry scales in sebum-rich areas such as face (87.7%), scalp (70.3%), upper trunk (26.8%), lower extremities (2.3%), and upper extremities (1.3%) [5,7,29]. Pruritus is not an obligatory feature, but it is often present, mainly in scalp involvement [2]. The main complication is secondary bacterial infection, which increases the redness and exudate and local irritation [3,15].
Other less common conditions that may resemble SD are pemphigus foliaceous, pityriasis rosea, secondary syphilis, diaper dermatitis and cutaneous Langerhans cell histiocytosis [3,4,7,30], which are summarized in Table 2. The majority of these conditions can be differentiated by clinical presentation and history; although syphilis, pemphigus foliaceous and SLE may require laboratory confirmation.
Seborrheic Dermatitis and Dandruff: A Comprehensive Review
Luis J. Borda and Tongyu C. Wikramanayake*
- 1Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine, 1600 NW 10th Avenue, RMSB 2023A, Miami, Florida 33136, USA
*Address for Correspondence: Tongyu C. Wikramanayake, Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine, 1600 NW 10th Avenue, RMSB 2023A, Miami, Florida 33136, USA, Tel: (305) 243-8878; Fax: (305) 243-3082; E-mail: tcao@med.miami.edu
Citation:Borda LJ, Wikramanayake TC. Seborrheic Dermatitis and Dandruff: A Comprehensive Review. J Clin Investigat Dermatol. 2015;3(2): 10.
Copyright © 2015 Borda LJ, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reviewed & Approved by: Dr. Craig G Burkhart, Clinical Professor, Department of Medicine, Ohio University, USA.
journal of Clinical & Investigative Dermatology | ISSN: 2373-1044 | Volume: 3, Issue: 2
Submission: 01 December 2015| Accepted: 11 December 2015 | Published: 15 December 2015
Abstract
Seborrheic Dermatitis (SD) and dandruff are of a continuous spectrum of the same disease that affects the seborrheic areas of the body. Dandruff is restricted to the scalp, and involves itchy, flaking skin without visible inflammation. SD can affect the scalp as well as other seborrheic areas, and involves itchy and flaking or scaling skin, inflammation and pruritus. Various intrinsic and environmental factors, such as sebaceous secretions, skin surface fungal colonization, individual susceptibility, and interactions between these factors, all contribute to the pathogenesis of SD and dandruff. In this review, we summarize the current knowledge on SD and dandruff, including epidemiology, burden of disease, clinical presentations and diagnosis, treatment, genetic studies in humans and animal models, and predisposing factors. Genetic and biochemical studies and investigations in animal models provide further insight on the pathophysiology and strategies for better treatment.Keywords
Seborrheic dermatitis; Dandruff; Sebaceous gland; Malassezia; Epidermal barrierAbbreviations
AIDS: Acquired Immune-Deficiency Syndrome; FTA-ABS: Fluorescent Treponemal Antibody-Absorption; HAART: Highly Active Antiretroviral Therapy; HIV: Human Immune-Deficiency Virus; ICD: Irritant Contact Dermatitis; QOL: Quality of Life; RPR: Rapid Plasma Regain; SC: Stratum Corneum; SD: Seborrheic Dermatitis; VDRL: Venereal Disease Research LaboratoryIntroduction
Seborrheic Dermatitis (SD) and dandruff are common dermatological problems that affect the seborrheic areas of the body. They are considered the same basic condition sharing many features and responding to similar treatments, differing only in locality and severity. Dandruff is restricted to the scalp, and involves itchy, flaking skin without visible inflammation. SD affects the scalp as well as face, retro-auricular area, and the upper chest, causing flaking, scaling, inflammation and pruritus, and can have marked erythema. Flaking in SD and dandruff is usually white-to-yellowish, and may be oily or dry.It is estimated that SD and dandruff combined affect half of the adult population. Despite such high prevalence, their etiology is not well understood. Various intrinsic and environmental factors, such as sebaceous secretions, skin surface fungal colonization, individual susceptibility, and interactions between these factors, all contribute to the pathogenesis. Genetic, biochemical studies and investigations in animal models further provided insight on the pathophysiology and strategies for better treatment. In this comprehensive review, we summarize the current knowledge on SD and dandruff, and attempt to provide directions for future investigations and treatments.
Epidemiology
SD is a common dermatological disorder in the United States and worldwide [1]. Its incidence peaks during three age periods - in the first three months of life, during puberty, and in adulthood with an apex at 40 to 60 years of age [1-4]. In infants up to three months of age, SD involves the scalp (termed “cradle cap”), the face, and diaper area. Incidence can be up to 42% [4-6]. In adolescents and adults, SD affects the scalp and other seborrheic areas on the face, upperchest, axillae, and inguinal folds [4,7,8]. Incidence is 1-3% of the general adult population [3,9]. Men are affected more frequently than women (3.0% vs. 2.6%) in all age groups, suggesting that SD may be associated with sex hormones such as androgens [1,3,8]. No apparent differences were observed in SD incidence between ethnic groups [3].SD is also associated with neurological disorders and psychiatric diseases, including Parkinson’s disease, neuroleptic induced parkinsonism, tardive dyskinesia, traumatic brain injury, epilepsy, facial nerve palsy, spinal cord injury and mood depression [4,5,16,17], chronic alcoholic pancreatitis, hepatitis C virus [18,19], and in patients with congenital disorders such as Down syndrome [20]. Furthermore, seborrhea-like dermatitis of the face may also develop in patients treated for psoriasis with psoralen and ultraviolet A (PUVA) therapy [21].
Comparing with SD, dandruff is much more common, and affects approximately 50% of the general adult population worldwide. It is also more prevalent in males than females [22,23]. Dandruff starts at puberty, reaches peak incidence and severity at the age of about 20 years, and becomes less prevalent among people over 50 [23]. Incidence varies between different ethnic groups: in a study in the U.S. and China, dandruff prevalence was 81-95% in African Americans, 66-82% in Caucasians, and 30-42% in Chinese [23].
Burden of Disease
It is estimated that at least 50 million Americans suffer from dandruff, who spend $300 million annually on over-the-counter products to treat scalp itching and flaking [22]. Besides physical discomfort such as itching, dandruff is socially embarrassing and negatively impacts patients’ self-esteem [22].While SD is much less prevalent, outpatient office visits alone cost $58 million in the United States in 2004, and $109 million were spent on prescription drugs [24]. Together with over-the-counter products and hospital services, the total direct costs of SD were estimated to be $179 million, plus another $51 million indirect costs in the form of lost work days [24]. In addition, because SD frequently occurs on the face and other visible areas, it has significant negative effects on patients’ quality of life (QOL) in the form of psychological distress or low self esteem; the willingness to pay for relief of the symptoms was $1.2 billion [24]. Furthermore, although the QOL impact in SD patients ranked lower than in patients with atopic or contact dermatitis, it was found to be higher than skin ulcers and solar radiation damage, and women, younger patients, and subjects with higher educational level were more affected [24].
Clinical Presentation and Diagnosis
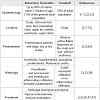
Clinical presentationsThe clinical presentations of SD and dandruff in children and adults are summarized in Table 1. SD often presents as well-delimited erythematous plaques with greasy-looking, yellowish scales of varying extents in regions rich in sebaceous glands, such as the scalp, the retro-auricular area, face (nasolabial folds, upper lip, eyelids and eyebrows), and the upper chest. Distribution of the lesions is generally symmetrical, and SD is neither contagious nor fatal. SD has a seasonal pattern, presenting more frequently during winter, and improving usually during summer [5,25,26]. Additionally, aggravation of SD has been associated with sleep deprivation and stress [7,27,28].
In infants, SD may present on the scalp, face, retro-auricular area, body folds, and trunk; rarely it may be generalized. Cradle cap is the most common clinical manifestation. SD in children is usually self-limited [3,15]. On the other hand, in adults, SD is a chronic or relapsing condition, featured by erythematous patches, with flaky, large, oily or dry scales in sebum-rich areas such as face (87.7%), scalp (70.3%), upper trunk (26.8%), lower extremities (2.3%), and upper extremities (1.3%) [5,7,29]. Pruritus is not an obligatory feature, but it is often present, mainly in scalp involvement [2]. The main complication is secondary bacterial infection, which increases the redness and exudate and local irritation [3,15].
In immune-suppressed patients, SD is often more extensive, intense, and refractory to treatment [3,26,30]. It is considered an early skin presentation of AIDS in both children and adults [14]. SD may also be a cutaneous sign of the immune reconstitution inflammatory syndrome in patients with highly active antiretroviral therapy (HAART) [31]. However, there have also been reports of SD regression with HAART [10].
Differential diagnosis
The main differential diagnosis of SD and dandruff includes psoriasis, atopic dermatitis (mainly in the pediatric form of SD), tinea capitis, rosacea, and systemic lupus erythematous (SLE) [3,7,8] (Table 2). While psoriasis can affect similar locations as SD, typical lesions in psoriasis are thicker and present as plaques sharply limited with silvery white scales [8,32]. Lesions in atopic dermatitis usually do not appear until after 3 months of age, while lesions in SD usually appear earlier and rarely affect extensor areas. Tinea capitis, a highly contagious disease, typically shows scaly patches of scalp hair loss associated with “black dots”, which represent distal ends of broken hairs [33]. Conversely, SD is not associated with hair loss. Rosacea usually targets the malar areas on the face, sparing the nasolabial folds, and do not have scales; on the other hand, facial SD lesions are usually scaly, and affect the nasolabial folds, eyelids, and eyebrows, without associated flushing or telangiectasias [7,8,34]. Finally, skin lesions in SLE often follow a clear photo distribution, such as acute flares of bilateral malar rash, and may be associated with extra-cutaneous abnormalities such as arthritis, mouth ulcers, glomerulonephritis or cardiomyopathy [8,35]; SD does not have a photo distribution pattern, and does not affect organ systems other than the skin.
Other less common conditions that may resemble SD are pemphigus foliaceous, pityriasis rosea, secondary syphilis, diaper dermatitis and cutaneous Langerhans cell histiocytosis [3,4,7,30], which are summarized in Table 2. The majority of these conditions can be differentiated by clinical presentation and history; although syphilis, pemphigus foliaceous and SLE may require laboratory confirmation.
Additionally, some drugs (griseofulvin, ethionamide, buspirone, haloperidol, chlorpromazine, IL-2, interferon-α, methyldopa, psoralens) and nutritional deficiencies (pyridoxine, zinc, niacin and riboflavin) may induce an SD-like dermatitis, although the mechanism remains unknown [36,37]. These conditions can coexist with SD as well, making the diagnosis more challenging.
Pathology
Diagnosis of SD is typically made by history and physical examination. In rare cases, a skin biopsy is needed for differential diagnosis. Histologically, the development of SD can be divided into two stages. In the acute and sub-acute stages, SD shows superficial perivascular and perifollicular inflammatory infiltrates, composed mainly of lymphocytes and histiocytes in association with spongiosis and psoriasiform hyperplasia, and can be coupled with parakeratosis around follicular opening (“shoulder parakeratosis”). Neutrophils can also be found in the scale crust at the margins of follicular ostia. On the other hand, in chronic lesions, marked psoriasiform hyperplasia and parakeratosis can be present with dilation of the venules of surface plexus which resembles psoriasis [3,4,38]. However, in psoriasis parakeratosis is often associated with thinning or loss of the granular layer due to accelerated keratinocyte differentiation.Dandruff shows many common features as SD in histology, such as epidermal hyperplasia, parakeratosis, and Malassezia yeasts surrounding the parakeratotic cells [23]. Whereas inflammatory cells such as lymphocytes and NK cells may be present in great numbers in SD, dandruff shows subtle neutrophil infiltration or no infiltration. These findings support the notion that dandruff and SD are of a continuous spectrum of the same disease entity with different severity and location [39].
Treatment
Treatment of SD and dandruff focuses on clearing signs of the disease; ameliorating associated symptoms, especially pruritus; and maintaining remission with long-term therapy. Because the main underlying pathogenic mechanisms involve Malassezia proliferation and local skin irritation and inflammation, the most common treatment is topical antifungal and anti-inflammatory agents (Table 3). Other widely used therapies are coal tar, lithium gluconate/ succinate and phototherapy (Table 3). New therapies have also emerged including immune modulators such as topical calcineurin inhibitors, and metronidazole, but their efficacy remains controversial [5]. Alternative therapies have been reported as well, such as tea tree oil [40,41]. Some factors to be considered before selecting a treatment include efficacy, side effects, ease of use/compliance, and age of the patient [5]. Systemic therapy is needed only in widespread lesions and in cases that do not respond to topical treatment [3, 26].Pathophysiology
Despite the high prevalence, the pathogenesis of SD and dandruff is not well understood. However, studies have identified several predisposing factors, including fungal colonization, sebaceous gland activity, as well as several factors that confer individual susceptibility [2].Fungal colonization
Several lines of evidence suggest a pathogenic role for yeasts of the genus Malassezia in SD and dandruff [42-46]. Malassezia are lipophilic yeasts that are found mainly on seborrheic regions of the body [5,7,47]. Studies have detected Malassezia on the scalp of dandruff patients [45,48], and higher numbers of Malassezia (M. globosa and M. restricta) correlate with SD appearance/severity [4,49,50]. Additionally, among the multiple chemical entities that are effective in treating SD and dandruff, such as azoles, hydroxypyridones, allylamines, selenium and zinc, the sole common mechanism of action is antifungal activity [47-49]. Furthermore, Malassezia was shown to have lipase activity, which hydrolyzes human sebum triglycerides and releases unsaturated fatty acids such as oleic and arachidonic acid [51,52]. These metabolites cause aberrant keratinocytes differentiation, resulting in stratum corneum abnormalities such as parakeratosis, intracellular lipid droplets, and irregular corneocyte envelope [53]. Such changes lead to disrupted epidermal barrier function and trigger inflammatory response, with or without visible local inflammation. In addition, these metabolites induce keratinocytes to produce pro-inflammatory cytokines such as IL-1α, IL-6, IL-8 and TNF-α, thus prolonging the inflammatory response [39,54]. Furthermore, arachidonic acid can be a source of prostaglandins, which are pro-inflammatory mediators that can cause inflammation via neutrophil recruitment and vasodilation [38]. Interestingly, Malassezia infection has also been reported in goats, dogs and monkeys with seborrhea (dry or greasy) and dermatitis [55-59].
While these observations support a pathogenic role for Malassezia in SD and dandruff, there is also strong evidence suggesting that individual predispositions and host interactions with Malassezia, rather than the mere presence of Malassezia, contribute to SD and dandruff pathogenesis. For example, Malassezia was detected on normal skin of majority of healthy adults, making it a commensal organism [2,5,26]. Moreover, while topical application of oleic acid did not induce visible changes in non-dandruff subjects, it caused skin flaking on the non-lesional scalp of dandruff patients [48]. These observations are suggestive of intrinsic epidermal barrier defects in the pathogenesis of SD and dandruff [48].
Sebaceous gland activity
Sebaceous glands (SGs) are distributed over the entire skin surface in humans, except on the palms and soles. Secretion of sebum is highest on the scalp, face and chest [44]. Sebum production is under hormonal control, and SGs are activated at birth under the influence of maternal androgens via androgen receptors in sebocytes [60]. SGs are activated again at puberty under the control of circulating androgens [38,61], resulting in increased sebum secretion during adolescence, which is kept stable between 20 and 30 years of age and is then reduced [62]. During the period of active sebum secretion, the secretion rate is higher in males and stays high longer, between 30 and 60 years of age; in females, the rate drops fast after menopause [44]. Thus, SD and dandruff have a strong time correlation with SG activity, with cradle cap after birth, increased incidence throughout the teens, between third and sixth decades and then decreasing [3,4,9]. However, SD patients may have normal sebum production, and individuals with excessive sebum production sometimes don’t develop SD [38,63]. These findings suggest that while SG activity strongly correlates with SD and dandruff, sebum production by itself is not a decisive cause.
In addition to the level of sebum production, abnormalities of lipid composition may also play a role in SD development, likely through a favorable milieu for Malassezia growth [64]. In patients with SD, triglycerides and squalene were reduced, but free fatty acids and cholesterol were considerably elevated [38,44]. The elevated levels of free fatty acids and cholesterol may be the result of triglyceride degradation by Malassezia’s lipase, and these metabolites promote Malassezia growth and lead to recruitment of inflammatory infiltrates in the skin [64].
Individual susceptibility
Besides sebaceous activity and Malassezia colonization, other factors also contribute to the pathogenesis of SD. Epidermal barrier integrity, host immune response, neurogenic factors and emotional stress, and nutritional factors have all been shown to play a role in individual susceptibility
Epidermal barrier integrity:
The stratum corneum (SC), the anucleated outer layers of the epidermis, functions as a barrier against water loss and entry of microorganisms and harmful agents from the environment [65]. The SC consists of several layers of terminally differentiated keratinocytes, the “corneocytes”, encased in lipid lamellae, held together by specialized intercellular cell adhesion structures called corneodesmosomes [66]. Any changes in the lamellar lipid composition, corneocyte size or shape, corneodesmosome number and SC thickness, could lead to alterations in the epidermal permeability barrier (EPB) function [66].
Normally, sebum may influence intercellular lipid organization to aid desquamation [66,67]. In SD and dandruff, however, altered corneodesmosomal hydrolysis may disrupt lipid organization and disturb the desquamation process, leading to aberrant barrier function [53,68]. In support of this notion, barrier structural abnormalities have been detected in dandruff scalp by electron microscopy that included intercellular Malassezia yeasts, changes in corneocyte shape and corneodesmosomes, and disrupted lipid lamellar structure [23,53,66]. Consistent with the structural findings, dandruff patients have been found to be more reactive (higher itch perception or flaking) than controls to topical applications of histamine or oleic acid to the scalp [48,69.70]. These observations indicate that disrupted EPB function can contribute to the aggravation of dandruff. Recent genetic studies in humans and animals suggest that disrupted barrier function may even directly cause SD-like conditions [71]. Biochemical analysis further demonstrated that dandruff skin displayed altered protein profiles as well as those of SC ceramides and free fatty acids, in the absence of apparent inflammation [72]. These studies underscore the importance of barrier restoration and maintenance in the management of SD and dandruff.
Immune response:Both the incidence and severity of SD are associated with immune-suppression, particularly in HIV/AIDS patients. Because no clear differences were found in Malassezia levels between individuals with and without SD in this population, it is likely that an immune or inflammatory reaction could be the predisposition [5,9]. Indeed, one study found elevated levels of human leukocyte antigens HLA-AW30, HLA-AW31, HLA-A32, HLA-B12 and HLA-B18 in SD [3,73,74]. Additionally, increased levels of total serum IgA and IgG antibodies have been detected in SD patients [75]. However, no increase in the titers of antibodies against Malassezia was detected, suggesting that the elevated immunoglobulin production occurs rather as a response to yeast etabolites [26,75,76]. The strong inflammatory reaction provoked by these metabolites includes infiltration of Natural Killer (NK) cells and macrophages, with concurrent local activation of complement and an increased local production of inflammatory cytokines, such as IL-1α, IL-1β, IL-6 and TNF-α in affected skin areas [54]. The lack of increase in anti-Malassezia antibodies also indicates a change in cellular immune response instead of humoral response [76,77]. The specific role of lymphocyte activity remains controversial [76-79].
Genetic factors: The genetic components of SD and dandruff had been under-appreciated until recently, when studies in animal models and humans identified inherited dominant and recessive forms of SD and dandruff. In the autosomal recessive “inherited seborrheic dermatitis” (seb) mice, a spontaneous mutation in the outbred Him:OF1 mice caused seborrhea, rough coat, alopecia, growth retardation, and sometimes abnormal pigmentation in homozygous mutants [80]. Histological examination revealed enlarged sebaceous glands, hyperkeratosis, parakeratosis, acanthosis and inflammatory infiltrates in the epidermis and dermis. Neither yeasts nor dermatophytes were detected. These mice were the first animal model of SD to show a clear mode of inheritance, though the underlying mutation remains unidentified [80,81].
Consistent with a role for altered immunity in the pathogenesis of SD, transgenic mice carrying the 2C T cell receptor (TCR) transgene in the DBA/2 background developed extremely inflammatory phenotype in seborrheic areas, such as the ears, around the eyes, and muzzle area [82]. Additionally, positive fungal staining by PAS was consistently detected in lesional skin but not readily apparent in non-lesional skin from diseased mice or from DBA/2 control mice. Furthermore, antifungal treatment reversed clinical and pathology presentations, and reduced PAS staining [82]. These observations support the notion that immune compromise and fungal infection play active roles in SD.
Another spontaneous mutant mouse strain that shows SD-like phenotype is the rough coat (rc) mice, which showed sebaceous hypertrophy and greasy hair coat, alopecia, and growth retardation [83]. The rc is transmitted in an autosomal recessive mode. We have since identified the cause of the rc phenotype to be a missense mutation in the Mpzl3 gene, which is expressed in the superficial layers of the epidermis [84,85]. Our Mpzl3knockout mice recapitulated the rc phenotype, and mice with white hair coat developed more severe and persistent inflammatory skin phenotype and dandruff in the seborrheic areas [85]. We have shown that the early-onset inflammatory skin phenotype was not caused by immune defects [85]. However, skin abnormalities in Mpzl3 knockout mice and perturbed epidermal differentiation in organotypic human skin models with MPZL3 knockdown indicate that MPZL3 is a key regulator of epidermal differentiation [85,86]. Interestingly, a frame-shift mutation in ZNF750, a transcription factor controlling epidermal differentiation and an upstream regulator of MPZL3, caused autosomal dominant seborrhea-like dermatitis in patients [71,86]. These studies in humans and animal models underscore the consequence of abnormal epidermal differentiation in the pathogenesis of SD and dandruff, and have provided the genetic basis for some of the predisposing factors discussed above. These animal models will be important tools to dissect the underlying pathways that will identify novel targets for better treatment of these disorders.
Neurogenic factors and emotional stress: The high incidence of SD in patients with Parkinson’s disease [17,87,88] and neurolepticinduced Parkinsonism [89,90] has long been observed, especially in those with severe seborrhea, which provides favorable conditions for Malasseziaproliferation. Bilateral seborrhea has been observed in patients with unilateral Parkinsonism, suggesting that these sebum changes were likely regulated neuro-endocrinologically rather than purely neurologically [5,26,91]. Consistent with this notion, α-melanocyte stimulating hormone (α-MSH) levels were elevated in Parkinson patients, possibly due to inadequate dopaminergic input. Moreover, treatment with L-dopa reduced α-MSH, and reestablished the synthesis of MSH-inhibiting factor, reducing sebum secretion [26,92].
Additionally, there is evidence for a link between neurological damage (e.g. traumatic brain, spinal cord injury) and SD [93]. Facial immobility of Parkinsonian patients (mask-like face) and immobility due to facial paralysis can induce elevated sebum accumulation and lead to SD, but only on the affected side [26,43,94]. Because poor hygiene has been implicated in SD, these observations suggest that sustained reservoirs of residual sebum associated with immobility may influence the manifestation of the disease [3,22,26,88]. SD is also more commonly seen in depressive disorders and emotional stress [5,16].
Other factors: In the past, nutrition has been studied as a possible contributing factor for SD. Zinc deficiency in patients with acrodermatitis enteropatica, riboflavin, pyridoxine and niacin deficiency can manifest seborrheicdermatitis-like rash [26,36]. Other medical conditions, such as familial amyloidotic polyneuropathy and Down syndrome, have also been associated with SD [95,96].
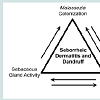
In summary, multiple predisposing factors have been identified in the pathogenesis of SD and dandruff (Figure 1). The presence and abundance of Malassezia yeast, host epidermal conditions and sebaceous secretion, combined with various other factors, and interactions between these factors, determine an individual’s susceptibility to SD and dandruff. In a likely scenario, there may be aberrant epidermal barrier function due to genetic predisposition, and excessive or altered sebum composition would exacerbateEPB disruption and provides a favorable milieu for Malassezia colonization. Disrupted EPB function facilitates entry of Malassezia and its metabolites, and irritates the epidermis and elicits host’s immune response. The host inflammatory response further disturbs epidermal differentiation and barrier formation, and pruritus and subsequent scratching would damage the barrier even further, leading to cycles of immune stimulation, abnormal epidermal differentiation,and barrier disruption.
Conclusions
SD and dandruff are of a continuous spectrum of the same disease that affects the seborrheic areas of the body (Table 4). They share many common features and respond to similar treatments. Various intrinsic and environmental factors, such as Malassezia yeast, host epidermal conditions, sebaceous secretion, immune response, and the interactions between these factors, may all contribute to the pathogenesis. Effective management of SD and dandruff requires clearing of symptoms with antifungal and anti-inflammatory treatment, ameliorating associated symptoms such as pruritus, and general scalp and skin health to help maintain remission. Studies in humans and animal models to investigate the genetic and biochemical pathways will help identify new targets for the development of more efficacious treatment with less side effects, and better management of these conditions.Acknowledgements
This work was supported by AR059907 from NIH/NIAMS, (T.C.W.), the Brian V. Jegasothy M.D. Basic Science Research Award (T.C.W.) and a Dermatology Gift Fund from the Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine (T.C.W.).References
- Gupta AK, Madzia SE, Batra R (2004) Etiology and management of Seborrheic dermatitis. Dermatology 208: 89-93.
- Del Rosso JQ (2011) Adult seborrheic dermatitis: a status report on practical topical management. J Clin Aesthet Dermatol 4: 32-38.
- Sampaio AL, Mameri AC, Vargas TJ, Ramos-e-Silva M, Nunes AP, et al. (2011) Seborrheic dermatitis. An Bras Dermatol 86: 1061-1071.
- Schwartz RA, Janusz CA, Janniger CK (2006) Seborrheic dermatitis: an overview. Am Fam Physician 74: 125-130.
- Dessinioti C, Katsambas A (2013) Seborrheic dermatitis: etiology, risk factors, and treatments: facts and controversies. Clin Dermatol 31: 343-351.
- Foley P, Zuo Y, Plunkett A, Merlin K, Marks R (2003) The frequency of common skin conditions in preschool-aged children in Australia: seborrheic dermatitis and pityriasis capitis (cradle cap). Arch Dermatol 139: 318-322.
- Clark GW, Pope SM, Jaboori KA (2015) Diagnosis and treatment of seborrheic dermatitis. Am Fam Physician 91: 185-190.
- Naldi L, Rebora A (2009) Clinical practice. Seborrheic dermatitis. N Engl J Med 360: 387-396.
- Gupta AK, Bluhm R, Cooper EA, Summerbell RC, Batra R (2003) Seborrheic dermatitis. Dermatol Clin 21: 401-412.
- Dunic I, Vesic S, Jevtovic DJ (2004) Oral candidiasis and seborrheic dermatitis in HIV-infected patients on highly active antiretroviral therapy. HIV Med 5: 50-54.
- Lally A, Casabonne D, Newton R, Wojnarowska F (2010) Seborrheic dermatitis among Oxford renal transplant recipients. J Eur Acad Dermatol Venereol 24: 561-564.
- Ozcan D, Seckin D, Ada S, Haberal M (2013) Mucocutaneous disorders in renal transplant recipients receiving sirolimus-based immunosuppressive therapy: a prospective, case-control study. Clin Transplant 27: 742-748.
- Okada K, Endo Y, Fujisawa A, Tanioka M, Kabashima K, et al. (2014) Refractory seborrheic dermatitis of the head in a patient with malignant lymphoma. Case Rep Dermatol 6: 279-282.
- Nnoruka EN, Chukwuka JC, Anisuiba B (2007) Correlation of mucocutaneous manifestations of HIV/AIDS infection with CD4 counts and disease progression. Int J Dermatol 46 Suppl 2: 14-18.
- Ramos-E-Silva M, Sampaio AL, Carneiro S (2014) Red face revisited: Endogenous dermatitis in the form of atopic dermatitis and seborrheic dermatitis. Clin Dermatol 32: 109-115.
- Maietta G, Fornaro P, Rongioletti F, Rebora A (1990) Patients with mood depression have a high prevalence of seborrhoeic dermatitis. Acta Derm Venereol 70: 432-434.
- Mastrolonardo M, Diaferio A, Logroscino G (2003) Seborrheic dermatitis, increased sebum excretion, and Parkinson's disease: a survey of (im)possible links. Med Hypotheses 60: 907-911.
- Barba A, Piubello W, Vantini I, Caliari S, Cocchetto R, et al. (1982) Skin lesions in chronic alcoholic pancreatitis. Dermatologica 164: 322-326.
- Cribier B, Samain F, Vetter D, Heid E, Grosshans E (1998) Systematic cutaneous examination in hepatitis C virus infected patients. Acta Derm Venereol 78: 355-357.
- Bilgili SG, Akdeniz N, Karadag AS, Akbayram S, Calka O, et al. (2011) Mucocutaneous disorders in children with down syndrome: case-controlled study. Genet Couns 22: 385-392.
- Tegner E (1983) Seborrhoeic dermatitis of the face induced by PUVA treatment. Acta Derm Venereol 63: 335-339.
- Manuel F, Ranganathan S (2011) A new postulate on two stages of dandruff: a clinical perspective. Int J Trichology 3: 3-6.
- Schwartz JR, Cardin CW, Dawson TL (2010) Seborrheic dermatitis and dandruff. In: Baran R, Maibach HI, (eds). Textbook of Cosmetic dermatology, London: Martin Dunitz, Ltd; pp. 230-241.
- Bickers DR, Lim HW, Margolis D, Weinstock MA, Goodman C, et al. (2006) The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol 55: 490-500.
- Berg M (1989) Epidemiological studies of the influence of sunlight on the skin. Photodermatol 6: 80-84.
- Bukvic Mokos Z, Kralj M, Basta-Juzbasic A, Lakos Jukic I (2012) Seborrheic dermatitis: an update. Acta Dermatovenerol Croat 20: 98-104.
- Misery L, Touboul S, Vincot C, Dutray S, Rolland-Jacob G, et al. (2007) Stress and seborrheic dermatitis. Ann Dermatol Venereol 134: 833-837.
- Szepietowski JC, Reich A, Wesolowska-Szepietowska E, Baran E, National Quality of Life in Dermatology Group (2009) Quality of life in patients suffering from seborrheic dermatitis: influence of age, gender and education level. Mycoses 52: 357-363.
- Peyri J, Lleonart M, Grupo español del Estudio SEBDERM (2007) Clinical and therapeutic profile and quality of life of patients with seborrheic dermatitis. Actas Dermosifiliogr 98: 476-482.
- Mathes BM, Douglass MC (1985) Seborrheic dermatitis in patients with acquired immunodeficiency syndrome. J Am Acad Dermatol 13: 947-951.
- Osei-Sekyere B, Karstaedt AS (2010) Immune reconstitution inflammatory syndrome involving the skin. Clin Exp Dermatol 35: 477-481.
- Boehncke WH (2015) Etiology and pathogenesis of psoriasis. Rheum Dis Clin North Am 41: 665-675.
- Meadows-Oliver M (2009) Tinea capitis: diagnostic criteria and treatment options. Pediatr Nurs 35: 53-57.
- Tuzun Y, Wolf R, Kutlubay Z, Karakus O, Engin B (2014) Rosacea and rhinophyma. Clin Dermatol 32: 35-46.
- Lisnevskaia L, Murphy G, Isenberg D (2014) Systemic lupus erythematosus. Lancet 384: 1878-1888.
- Valia RG (2006) Etiopathogenesis of seborrheic dermatitis. Indian J Dermatol Venereol Leprol 72: 253-255.
- Yamamoto T, Tsuboi R (2008) Interleukin-2-induced seborrhoeic dermatitis-like eruption. J Eur Acad Dermatol Venereol 22: 244-245.
- Reider N, Fritsch PO (2012) Other eczematous eruptions. In: Bolognia JL, Jorizzo JL, Schaffer JV (eds). Dermatology: Elsevier Health Sciences UK. pp. 219-221.
- Schwartz JR, Messenger AG, Tosti A, Todd G, Hordinsky M, et al. (2013) A comprehensive pathophysiology of dandruff and seborrheic dermatitis - towards a more precise definition of scalp health. Acta Derm Venereol 93: 131-137.
- Pazyar N, Yaghoobi R, Bagherani N, Kazerouni A (2013) A review of applications of tea tree oil in dermatology. Int J Dermatol 52: 784-790.
- Satchell AC, Saurajen A, Bell C, Barnetson RS (2002) Treatment of dandruff with 5% tea tree oil shampoo. J Am Acad Dermatol 47: 852-855.
- Gaitanis G, Magiatis P, Hantschke M, Bassukas ID, Velegraki A (2012) The Malassezia genus in skin and systemic diseases. Clin Microbiol Rev 25: 106-141.
- Hay RJ (2011) Malassezia, dandruff and seborrhoeic dermatitis: an overview. Br J Dermatol 165 Suppl 2: 2-8.
- Ro BI, Dawson TL (2005) The role of sebaceous gland activity and scalp microfloral metabolism in the etiology of seborrheic dermatitis and dandruff. J Investig Dermatol Symp Proc 10: 194-197.
- Rudramurthy SM, Honnavar P, Dogra S, Yegneswaran PP, Handa S, et al. (2014) Association of Malassezia species with dandruff. Indian J Med Res 139: 431-437.
- Shuster S (1984) The aetiology of dandruff and the mode of action of therapeutic agents. Br J Dermatol 111: 235-242.
- Berk T, Scheinfeld N (2010) Seborrheic dermatitis. P T 35: 348-352.
- DeAngelis YM, Gemmer CM, Kaczvinsky JR, Kenneally DC, Schwartz JR, et al. (2005) Three etiologic facets of dandruff and seborrheic dermatitis: Malassezia fungi, sebaceous lipids, and individual sensitivity. J Investig Dermatol Symp Proc 10: 295-297.
- Heng MC, Henderson CL, Barker DC, Haberfelde G (1990) Correlation of Pityosporum ovale density with clinical severity of seborrheic dermatitis as assessed by a simplified technique. J Am Acad Dermatol 23: 82-86.
- McGinley KJ, Leyden JJ, Marples RR, Kligman AM (1975) Quantitative microbiology of the scalp in non-dandruff, dandruff, and seborrheic dermatitis. J Invest Dermatol 64: 401-405.
- DeAngelis YM, Saunders CW, Johnstone KR, Reeder NL, Coleman CG, et al. (2007) Isolation and expression of a Malassezia globosa lipase gene, LIP1. J Invest Dermatol 127: 2138-2146.
- Plotkin LI, Squiquera L, Mathov I, Galimberti R, Leoni J (1996) Characterization of the lipase activity of Malassezia furfur. J Med Vet Mycol 34: 43-48.
- Warner RR, Schwartz JR, Boissy Y, Dawson TL Jr. (2001) Dandruff has an altered stratum corneum ultrastructure that is improved with zinc pyrithione shampoo. J Am Acad Dermatol 45: 897-903.
- Faergemann J, Bergbrant IM, Dohse M, Scott A, Westgate G (2001) Seborrhoeic dermatitis and Pityrosporum (Malassezia) folliculitis: characterization of inflammatory cells and mediators in the skin by immunohistochemistry. Br J Dermatol 144: 549-556.
- Breen PT (1971) Canine seborrheic dermatitis. Vet Med Small Anim Clin 66: 655-656.
- Eguchi-Coe Y, Valentine BA, Gorman E, Villarroel A (2011) Putative Malassezia dermatitis in six goats. Vet Dermatol 22: 497-501.
- Newcomer CE, Fox JG, Taylor RM, Smith DE (1984) Seborrheic dermatitis in a rhesus monkey (Macaca mulatta). Lab Anim Sci 34: 185-187.
- Pin D (2004) Seborrhoeic dermatitis in a goat due to Malassezia pachydermatis. Vet Dermatol 15:53-56.
- Uzal FA, Paulson D, Eigenheer AL, Walker RL (2007) Malassezia slooffiae-associated dermatitis in a goat. Vet Dermatol 18: 348-352.
- Zouboulis CC, Akamatsu H, Stephanek K, Orfanos CE (1994) Androgens affect the activity of human sebocytes in culture in a manner dependent on the localization of the sebaceous glands and their effect is antagonized by spironolactone. Skin Pharmacol 7: 33-40.
- Niemann C, Horsley V (2012) Development and homeostasis of the sebaceous gland. Semin Cell Dev Biol 23: 928-936.
- Strauss JS, Downing DT, Ebling FJ (1983) Sebaceous glands. In: Goldsmith LA (ed). Biochemistry and physiology of skin, New York, Oxford University Press, pp. 569-595.
- Burton JL, Pye RJ (1983) Seborrhoea is not a feature of seborrhoeic dermatitis. Br Med J (Clin Res Ed) 286: 1169-1170.
- Ostlere LS, Taylor CR, Harris DW, Rustin MH, Wright S, et al. (1996) Skin surface lipids in HIV-positive patients with and without seborrheic dermatitis. Int J Dermatol 35: 276-279.
- Harding CR (2004) The stratum corneum: structure and function in health and disease. Dermatol Ther 17 Suppl 1: 6-15.
- Turner GA, Hoptroff M, Harding CR (2012) Stratum corneum dysfunction in dandruff. Int J Cosmet Sci 34: 298-306.
- Sheu HM, Chao SC, Wong TW, Yu-Yun Lee J, Tsai JC (1999) Human skin surface lipid film: an ultrastructural study and interaction with corneocytes and intercellular lipid lamellae of the stratum corneum. Br J Dermatol 140: 385-391.
- Simon M, Tazi-Ahnini R, Jonca N, Caubet C, Cork MJ, et al. (2008) Alterations in the desquamation-related proteolytic cleavage of corneodesmosin and other corneodesmosomal proteins in psoriatic lesional epidermis. Br J Dermatol 159: 77-85.
- Harding CR, Moore AE, Rogers JS, Meldrum H, Scott AE, et al. (2002) Dandruff: a condition characterized by decreased levels of intercellular lipids in scalp stratum corneum and impaired barrier function. Arch Dermatol Res 294: 221-230.
- Rukwied R, Zeck S, Schmelz M, McGlone F (2002) Sensitivity of human scalp skin to pruritic stimuli investigated by intradermal microdialysis in vivo. J Am Acad Dermatol 47: 245-250.
- Birnbaum RY, Zvulunov A, Hallel-Halevy D, Cagnano E, Finer G, et al. (2006) Seborrhea-like dermatitis with psoriasiform elements caused by a mutation in ZNF750, encoding a putative C2H2 zinc finger protein. Nat Genet 38: 749-751.
- Kerr K, Darcy T, Henry J, Mizoguchi H, Schwartz JR, et al. (2011) Epidermal changes associated with symptomatic resolution of dandruff: biomarkers of scalp health. Int J Dermatol 50: 102-113.
- Sampaio AL, Nunes AP (2011) Study of frequency of human leukocyte antigen (HLA) in seborrheic dermatitis patients in a miscegenated population. School of Medicine, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil.
- Tsuji K, Nose Y, Ito M, Ozala A, Matsuo I (1976) HLA antigens and susceptibility to psoriasis vulgaris in a non-Caucasian population. Tissue Antigens 8: 29-33.
- Bergbrant IM, Johansson S, Robbins D, Scheynius A, Faergemann J, et al. (1991) An immunological study in patients with seborrhoeic dermatitis. Clin Exp Dermatol 16: 331-338.
- Parry ME, Sharpe GR (1998) Seborrhoeic dermatitis is not caused by an altered immune response to Malassezia yeast. Br J Dermatol 139: 254-263.
- Ashbee HR, Ingham E, Holland KT, Cunliffe WJ (1994) Cell-mediated immune responses to Malassezia furfur serovars A, B and C in patients with pityriasis versicolor, seborrheic dermatitis and controls. Exp Dermatol 3: 106-112.
- Bergbrant IM, Andersson B, Faergemann J (1999) Cell-mediated immunity to Malassezia furfur in patients with seborrhoeic dermatitis and pityriasis versicolor. Clin Exp Dermatol 24: 402-406.
- Neuber K, Kroger S, Gruseck E, Abeck D, Ring J (1996) Effects of Pityrosporum ovale on proliferation, immunoglobulin (IgA, G, M) synthesis and cytokine (IL-2, IL-10, IFN gamma) production of peripheral blood mononuclear cells from patients with seborrhoeic dermatitis. Arch Dermatol Res 288: 532-536.
- Hoger H, Gialamas J, Adamiker D (1987) Inherited seborrheic dermatitis--a new mutant in mice. Lab Anim 21: 299-305.
- Hoger H, Gialamas J, Adamiker D (1994) Reduced tumour incidence in mice with inherited seborrhoeic dermatitis. Lab Anim 28: 340-346.
- Oble DA, Collett E, Hsieh M, Ambjorn M, Law J, et al. (2005) A novel T cell receptor transgenic animal model of seborrheic dermatitis-like skin disease. J Invest Dermatol 124: 151-159.
- Hayashi K, Cao T, Passmore H, Jourdan-Le Saux C, Fogelgren B, et al. (2004) Progressive hair loss and myocardial degeneration in rough coat mice: reduced lysyl oxidase-like (LOXL) in the skin and heart. J Invest Dermatol 123: 864-871.
- Cao T, Racz P, Szauter KM, Groma G, Nakamatsu GY, et al. (2007) Mutation in Mpzl3, a novel [corrected] gene encoding a predicted [corrected] adhesion protein, in the rough coat (rc) mice with severe skin and hair abnormalities. J Invest Dermatol 127: 1375-1386.
- Leiva AG, Chen AL, Devarajan P, Chen Z, Damanpour S, et al. (2014) Loss of Mpzl3 function causes various skin abnormalities and greatly reduced adipose depots. J Invest Dermatol 134: 1817-1827.
- Bhaduri A, Ungewickell A, Boxer LD, Lopez-Pajares V, Zarnegar BJ, et al. (2015) Network analysis identifies mitochondrial regulation of epidermal differentiation by MPZL3 and FDXR. Dev Cell 35: 444-457.
- Burton JL, Cartlidge M, Cartlidge NE, Shuster S (1973) Sebum excretion in parkinsonism. Br J Dermatol 88: 263-266.
- Cowley NC, Farr PM, Shuster S (1990) The permissive effect of sebum in seborrhoeic dermatitis: an explanation of the rash in neurological disorders. Br J Dermatol 122: 71-76.
- Binder RL, Jonelis FJ (1983) Seborrheic dermatitis in neuroleptic-induced parkinsonism. Arch Dermatol 119: 473-475.
- Binder RL, Jonelis FJ (1984) Seborrheic dermatitis: a newly reported side effect of neuroleptics. J Clin Psychiatry 45: 125-126.
- Burton JL, Shuster S (1970) Effect of L-dopa on seborrhoea of parkinsonism. Lancet 2: 19-20.
- Burton JL, Cartlidge M, Shuster S (1973) Effect of L-dopa on the seborrhoea of Parkinsonism. Br J Dermatol 88: 475-479.
- Wilson CL, Walshe M (1988) Incidence of seborrheic dermatitis in spinal injury patients. Br J Dermatol (suppl 33) 119: 48.
- Bettley FR, Marten RH (1956) Unilateral seborrheic dermatitis following a nerve lesion. AMA Arch Derm 73: 110-115.
- Ercis M, Balci S, Atakan N (1996) Dermatological manifestations of 71 Down syndrome children admitted to a clinical genetics unit. Clin Genet 50: 317-320.
- Rocha N, Velho G, Horta M, Martins A, Massa A (2005) Cutaneous manifestations of familial amyloidotic polyneuropathy. J Eur Acad Dermatol Venereol 19: 605-607.
- Hald M, Arendrup MC, Svejgaard EL, Lindskov R, Foged EK, et al. (2015) Evidence-based Danish guidelines for the treatment of Malassezia-related skin diseases. Acta Derm Venereol 95: 12-19.
- Koc E, Arca E, Kose O, Akar A (2009) An open, randomized, prospective, comparative study of topical pimecrolimus 1% cream and topical ketoconazole 2% cream in the treatment of seborrheic dermatitis. J Dermatolog Treat 20: 4-9.
- Okokon EO, Verbeek JH, Ruotsalainen JH, Ojo OA, Bakhoya VN (2015) Topical antifungals for seborrhoeic dermatitis. Cochrane Database Syst Rev 5: CD008138.
- Peter RU, Richarz-Barthauer U (1995) Successful treatment and prophylaxis of scalp seborrhoeic dermatitis and dandruff with 2% ketoconazole shampoo: results of a multicentre, double-blind, placebo-controlled trial. Br J Dermatol 132: 441-445.
- Pierard-Franchimont C, Goffin V, Decroix J, Pierard GE (2002) A multicenter randomized trial of ketoconazole 2% and zinc pyrithione 1% shampoos in severe dandruff and seborrheic dermatitis. Skin Pharmacol Appl Skin Physiol 15: 434-441.
- Segal R, David M, Ingber A, Lurie R, Sandbank M (1992) Treatment with bifonazole shampoo for seborrhea and seborrheic dermatitis: a randomized, double-blind study. Acta Derm Venereol 72: 454-455.
- Faergemann J (1986) Seborrhoeic dermatitis and Pityrosporum orbiculare: treatment of seborrhoeic dermatitis of the scalp with miconazole-hydrocortisone (Daktacort), miconazole and hydrocortisone. Br J Dermatol 114: 695-700.
- Dupuy P, Maurette C, Amoric JC, Chosidow O, Study Investigator Group (2001) Randomized, placebo-controlled, double-blind study on clinical efficacy of ciclopiroxolamine 1% cream in facial seborrhoeic dermatitis. Br J Dermatol 144: 1033-1037.
- Ratnavel RC, Squire RA, Boorman GC (2007) Clinical efficacies of shampoos containing ciclopirox olamine (1.5%) and ketoconazole (2.0%) in the treatment of seborrhoeic dermatitis. J Dermatolog Treat 18: 88-96.
- Danby FW, Maddin WS, Margesson LJ, Rosenthal D (1993) A randomized, double-blind, placebo-controlled trial of ketoconazole 2% shampoo versus selenium sulfide 2.5% shampoo in the treatment of moderate to severe dandruff. J Am Acad Dermatol 29: 1008-1012.
- Gilbertson K, Jarrett R, Bayliss SJ, Berk DR (2012) Scalp discoloration from selenium sulfide shampoo: a case series and review of the literature. Pediatr Dermatol 29: 84-88.
- Reeder NL, Xu J, Youngquist RS, Schwartz JR, Rust RC, et al. (2011) The antifungal mechanism of action of zinc pyrithione. Br J Dermatol 165 Suppl 2: 9-12.
- Shin H, Kwon OS, Won CH, Kim BJ, Lee YW, et al. (2009) Clinical efficacies of topical agents for the treatment of seborrheic dermatitis of the scalp: a comparative study. J Dermatol 36: 131-137.
- Ortonne JP, Lacour JP, Vitetta A, Le Fichoux Y (1992) Comparative study of ketoconazole 2% foaming gel and betamethasone dipropionate 0.05% lotion in the treatment of seborrhoeic dermatitis in adults. Dermatology 184: 275-280.
- Elewski B (2009) An investigator-blind, randomized, 4-week, parallel-group, multicenter pilot study to compare the safety and efficacy of a nonsteroidal cream (Promiseb Topical Cream) and desonide cream 0.05% in the twice-daily treatment of mild to moderate seborrheic dermatitis of the face. Clin Dermatol 27 (6 Suppl): S48-S53.
- Kircik LH (2009) Treatment of scalp and facial seborrheic dermatitis with desonide hydrogel 0.05%. J Clin Aesthet Dermatol 2: 32-36.
- Pierard-Franchimont C, Pierard GE (2002) A double-blind placebo-controlled study of ketoconazole + desonide gel combination in the treatment of facial seborrheic dermatitis. Dermatology 204: 344-347.
- Kircik L (2010) The evolving role of therapeutic shampoos for targeting symptoms of inflammatory scalp disorders. J Drugs Dermatol 9: 41-48.
- 115. Cook BA, Warshaw EM (2009) Role of topical calcineurin inhibitors in the treatment of seborrheic dermatitis: a review of pathophysiology, safety, and efficacy. Am J Clin Dermatol 10: 103-118.
- Kim BS, Kim SH, Kim MB, Oh CK, Jang HS, et al. (2007) Treatment of facial seborrheic dermatitis with pimecrolimus cream 1%: an open-label clinical study in Korean patients. J Korean Med Sci 22: 868-872.
- Ozden MG, Tekin NS, Ilter N, Ankarali H (2010) Topical pimecrolimus 1% cream for resistant seborrheic dermatitis of the face: an open-label study. Am J Clin Dermatol 11: 51-54.
- Thaci D, Salgo R (2010) Malignancy concerns of topical calcineurin inhibitors for atopic dermatitis: facts and controversies. Clin Dermatol 28: 52-56.
- Kim HO, Yang YS, Ko HC, Kim GM, Cho SH, et al. (2015) Maintenance therapy of facial seborrheic dermatitis with 0.1% tacrolimus ointment. Ann Dermatol 27: 523-530.
- Papp KA, Papp A, Dahmer B, Clark CS (2012) Single-blind, randomized controlled trial evaluating the treatment of facial seborrheic dermatitis with hydrocortisone 1% ointment compared with tacrolimus 0.1% ointment in adults. J Am Acad Dermatol 67: e11-e15.
- Davies DB, Boorman GC, Shuttleworth D (1999) Comparative efficacy of shampoos containing coal tar (4.0% w/w; Tarmed™), coal tar (4.0% w/w) plus ciclopirox olamine (1.0% w/w; Tarmed™ AF) and ketoconazole (2.0% w/w; Nizoral™) for the treatment of dandruff/seborrhoeic dermatitis. J Dermatolog Treat 10: 177-183.
- Ballanger F, Tenaud I, Volteau C, Khammari A, Dreno B (2008) Anti-inflammatory effects of lithium gluconate on keratinocytes: a possible explanation for efficiency in seborrhoeic dermatitis. Arch Dermatol Res 300: 215-223.
- Dreno B, Moyse D (2002) Lithium gluconate in the treatment of seborrhoeic dermatitis: a multicenter, randomised, double-blind study versus placebo. Eur J Dermatol 12: 549-552.
- Stefanaki I, Katsambas A (2010) Therapeutic update on seborrheic dermatitis. Skin Therapy Lett 15: 1-4.
- Koca R, Altinyazar HC, Esturk E (2003) Is topical metronidazole effective in seborrheic dermatitis? A double-blind study. Int J Dermatol 42: 632-635.
- Seckin D, Gurbuz O, Akin O (2007) Metronidazole 0.75% gel vs. ketoconazole 2% cream in the treatment of facial seborrheic dermatitis: a randomized, double-blind study. J Eur Acad Dermatol Venereol 21: 345-350.
- Lee E, Koo J, Berger T (2005) UVB phototherapy and skin cancer risk: a review of the literature. Int J Dermatol 44: 355-360.
- Pirkhammer D, Seeber A, Honigsmann H, Tanew A (2000) Narrow-band ultraviolet B (ATL-01) phototherapy is an effective and safe treatment option for patients with severe seborrhoeic dermatitis. Br J Dermatol 143: 964-968.
- Reich A, Medrek K (2013) Effects of narrow band UVB (311 nm) irradiation on epidermal cells. Int J Mol Sci 14: 8456-8466.
- Das J, Majumdar M, Chakraborty U, Majumdar V, Mazumdar G, et al. (2011) Oral itraconazole for the treatment of severe seborrhoeic dermatitis. Indian J Dermatol 56: 515-516.
- Kose O, Erbil H, Gur AR (2005) Oral itraconazole for the treatment of seborrhoeic dermatitis: an open, noncomparative trial. J Eur Acad Dermatol Venereol 19: 172-175.
- Gupta AK, Richardson M, Paquet M (2014) Systematic review of oral treatments for seborrheic dermatitis. J Eur Acad Dermatol Venereol 28: 16-26.
- Scaparro E, Quadri G, Virno G, Orifici C, Milani M (2001) Evaluation of the efficacy and tolerability of oral terbinafine (Daskil) in patients with seborrhoeic dermatitis. A multicentre, randomized, investigator-blinded, placebo-controlled trial. Br J Dermatol 144: 854-857.
- Vena GA, Micali G, Santoianni P, Cassano N, Peruzzi E (2005) Oral terbinafine in the treatment of multi-site seborrhoic dermatitis: a multicenter, double-blind placebo-controlled study. Int J Immunopathol Pharmacol 18: 745-753.