Advances in Diabetes & Endocrinology
Download PDF
Cardiovascular: Pulse 102 bpm, blood pressure 220/120 mmHg. Neurological: Altered mental status with confusion; focal neurological deficits were present.
Abdomen: Generalized tenderness with a rigid abdomen on palpation.
Capillary blood glucose measurement read “too high” on the glucometer; therefore, a serum glucose test was ordered for accurate assessment.
ondansetron (Emeset) 4 mg IV and pantoprazole (Pantop) 40 mg IV were given. Levetiracetam (Levera) IV was administered for seizure control and prophylaxis.
Case Report
Management of Hyperglycemic Hyperosmolar State with Hypertension
Sibi Das*
Department of Medicine, NC Medical College, Israna, Panipat, Haryana, India.
*Address for Correspondence:Dr. Sibi Das, Department of Medicine, NC Medical College,
Israna, Panipat, Haryana, India. E-mail Id: sdsilvanose@gmail.com
Submission:27 January, 2026
Accepted:24 February, 2026
Published: 26 February, 2026
Copyright: © 2026 Das S This is an open access article
distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction
in any medium, provided the original work is properly cited.
Abstract
Management of Hyperosmolar Hyperglycemic State (HHS) with coexisting
hypertension is a medical emergency requiring urgent, multidisciplinary care
and immediate hospitalization. Treatment focuses on aggressive intravenous fluid
resuscitation, careful electrolyte replacement—particularly potassium—and lowdose
insulin therapy to gradually reduce hyperglycemia while minimizing the risk
of cerebral edema. Concurrently, underlying precipitating factors, most commonly
infection, must be identified and treated. Preventive measures against complications
such as thromboembolism, including prophylactic heparin, are also essential in the
absence of hemorrhagic stroke.
Introduction
Hyperosmolar Hyperglycemic State (HHS), previously known as
Hyperosmolar Non-Ketotic Syndrome/Coma (HONK), is a severe,
life-threatening complication of diabetes characterized by extreme
hyperglycemia, profound dehydration, and marked hyperosmolarity.
Blood glucose levels typically exceed 600 mg/dL, with effective serum
osmolality greater than 320 mOsm/L. Severe hyperglycemia causes
osmotic diuresis, leading to frequent urination, excessive fluid loss,
intense thirst, and ultimately severe dehydration. This dehydration
results in a high concentration of solutes in the blood, producing
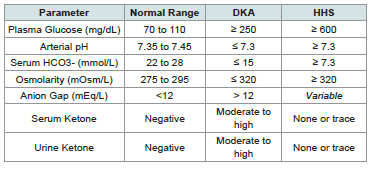
hyperosmolarity. Unlike Diabetic Ketoacidosis (DKA), HHS is
associated with minimal or no ketone production, and the anion gap
is usually normal due to the absence of significant ketoacidosis.
The key differential diagnosis between DKA and HHS are summarized in [Table 1].
Serum osmolality is calculated to estimate blood solute concentration using sodium, glucose, and blood urea nitrogen (BUN). The commonly used formula is Osmolality = 2[Na+] + (Glucose ÷ 18) + (BUN ÷ 2.8), where sodium is measured in mEq/L and glucose and BUN are measured in mg/dL.
The anion gap (AG) is used to assess metabolic acidosis by calculating the difference between measured cations and measured anions in the blood, reflecting the presence of unmeasured anions such as albumin, phosphate, and organic acids. The standard formula
The key differential diagnosis between DKA and HHS are summarized in [Table 1].
Serum osmolality is calculated to estimate blood solute concentration using sodium, glucose, and blood urea nitrogen (BUN). The commonly used formula is Osmolality = 2[Na+] + (Glucose ÷ 18) + (BUN ÷ 2.8), where sodium is measured in mEq/L and glucose and BUN are measured in mg/dL.
The anion gap (AG) is used to assess metabolic acidosis by calculating the difference between measured cations and measured anions in the blood, reflecting the presence of unmeasured anions such as albumin, phosphate, and organic acids. The standard formula
is: AG = (Na+ + K+) − (Cl+ + HCO₃-), with values expressed in mEq/L.
A normal anion gap typically ranges from 8–16 mEq/L (or 6–12
mEq/L if potassium is excluded).
Case Presentation
A 73-year-old female patient was admitted to the emergency
department of N.C. Medical College and Hospital, Israna, District
Panipat, Haryana, with a reduced level of consciousness and focal
neurological deficits. Prior to admission, she experienced shortness
of breath, vomiting, and a seizure episode. She had a known history of
diabetes mellitus and hypertension for the past 13 years.
Clinical Examination:
On arrival, the patient’s vital signs were notable for severe
hypertension and signs of poor perfusion. Blood pressure was 220/120
mmHg, heart rate 95 beats/min, respiratory rate 24–28 breaths/min
with SpO₂ 93% on room air, and Glasgow Coma Scale was 8/15.Physical examination findings::
Respiratory: Shortness of breath with oxygen saturation of 93%
on room air.Cardiovascular: Pulse 102 bpm, blood pressure 220/120 mmHg. Neurological: Altered mental status with confusion; focal neurological deficits were present.
Abdomen: Generalized tenderness with a rigid abdomen on palpation.
Capillary blood glucose measurement read “too high” on the glucometer; therefore, a serum glucose test was ordered for accurate assessment.
Key Management Steps::
Initial management included intravenous hydration with normal
saline at 60 mL/hour and administration of labetalol 20 mg IV to
treat the hypertensive emergency. To control nausea and vomiting,ondansetron (Emeset) 4 mg IV and pantoprazole (Pantop) 40 mg IV were given. Levetiracetam (Levera) IV was administered for seizure control and prophylaxis.
Laboratory investigations revealed that marked hyperglycemia
with a blood glucose level of 658 mg/dL and serum osmolality of
330 mOsm/kg, consistent with hyperosmolarity. Total leukocyte
count was significantly elevated at 24,100/mm³. Serum troponin,
renal function tests, and liver function tests were within normal
limits, while CK-MB showed mild elevation (34.5 mmol/L; normal
<25 mmol/L). Arterial blood pH level was 7.4. Urine analysis
demonstrated trace ketones. Based on these findings, a diagnosis of
hyperosmolar hyperglycemic state (HHS) was confirmed, and the
patient was transferred to the Intensive Care Unit (ICU) for further
management.
In the ICU, aggressive hydration with normal saline was
continued. A low-dose insulin infusion was initiated using 50 units of
rapid-acting insulin in 50 mL normal saline at a rate of 3 units/hour.
Given the markedly elevated leukocyte count and the possibility of
infection precipitating HHS, intravenous ceftriaxone and amikacin
were started empirically.
The first glucose measurement in the ICU after insulin
administration was 396 mg/dL, and levels gradually declined with
ongoing insulin therapy and fluid replacement, falling to below 250
mg/dL on the second day. Initially, blood glucose was monitored
hourly; once levels dropped below 200 mg/dL, monitoring was
extended to every two hours. To prevent a rapid fall in blood glucose
and reduce the risk of cerebral edema, 5% dextrose infusion was
initiated once glucose approached target levels. As insulin therapy
caused intracellular potassium shift, serum potassium dropped to 2.7
mmol/L and was corrected with 40 mmol potassium supplementation
in IV fluids to prevent hypokalemia. Antihypertensive therapy with
tablet telmisartan 40 mg and capsule nifedipine 10 mg was continued.
By the third day, the hyperosmolar hyperglycemic state had
resolved; however, the patient remained confused. The primary
therapeutic goals were to gradually reduce serum osmolality, control
blood pressure, and lower blood glucose by approximately 50 mg/dL
per hour. Multiple attempts to perform an MRI scan were unsuccessful
due to the patient’s non-compliance. Given the hypercoagulable state
associated with hyperosmolarity, low-molecular-weight heparin was
indicated for thromboprophylaxis; however, its administration was
deferred as hemorrhagic stroke could not be excluded in the absence
of MRI imaging.
Discussion
Hyperosmolar Hyperglycemic State (HHS) is a life-threatening
metabolic complication of diabetes mellitus characterized by severe
hyperglycemia, hyperosmolality, and dehydration in the absence
of significant ketoacidosis. The underlying pathophysiology results
from a relative insulin deficiency combined with increased counterregulatory
hormones such as glucagon, cortisol, and catecholamines.
This hormonal imbalance promotes excessive hepatic glucose
production and impaired peripheral glucose utilization, leading to
extreme elevations in blood glucose levels, often exceeding 600 mg/
dL.
The profound hyperglycemia induces osmotic diuresis, causing
massive fluid and electrolyte losses. Progressive dehydration leads
to reduced renal perfusion, further limiting glucose excretion and
worsening hyperosmolality. Unlike diabetic ketoacidosis, some
endogenous insulin activity is preserved in HHS, which prevents
lipolysis and ketone formation; this explains the absence or minimal
presence of ketones in this patient. Neurological manifestations such
as confusion, focal deficits, and coma are primarily due to cellular
dehydration within the central nervous system caused by elevated
serum osmolality.
Management of HHS focuses on four key principles: aggressive fluid resuscitation, gradual correction of hyperglycemia, electrolyte replacement, and identification of precipitating factors. Intravenous isotonic saline is the cornerstone of therapy because restoration of intravascular volume improves renal perfusion and enhances glucose clearance. In this case, normal saline was appropriately initiated to correct dehydration and reduce osmolality.
Insulin therapy is introduced after initial fluid replacement to avoid rapid shifts in osmolality. A low-dose continuous insulin infusion was used to gradually reduce blood glucose by approximately 50 mg/dL per hour, which aligns with recommended guidelines and minimizes the risk of cerebral edema. Once glucose levels approached 250 mg/dL, dextrose infusion was added to prevent an abrupt decline and to allow continued insulin administration for resolution of hyperosmolarity.
Electrolyte monitoring is essential, particularly potassium. Insulin drives potassium into cells, predisposing patients to hypokalemia, as observed in this case. Timely potassium supplementation was necessary to prevent cardiac arrhythmias and neuromuscular complications. Additionally, prophylactic low-molecular-weight heparin was justified because hyperosmolarity and dehydration increase blood viscosity and thrombotic risk.
Infections are common precipitants of HHS due to stress induced hormone release and insulin resistance. The markedly elevated leukocyte count raised suspicion of an underlying infection; therefore, empirical broad-spectrum antibiotics were initiated early, which is consistent with best practice.
Despite metabolic correction, the patient remained confused, reflecting delayed neurological recovery from prolonged hyperosmolality or a possible cerebrovascular event, warranting neuroimaging. Early recognition and adherence to evidence-based management were crucial in reversing the metabolic derangements and preventing further complications.
The cornerstone of Hyperosmolar Hyperglycemic State (HHS) management is the rapid correction of dehydration, gradual reduction of hyperglycemia, restoration of electrolyte balance—particularly potassium—and identification and treatment of precipitating factors such as infection. Effective management requires a structured and closely monitored approach:
Fluid Resuscitation: Immediate initiation of intravenous isotonic saline (0.9% NaCl) is essential to correct severe volume depletion and improve tissue perfusion. Depending on serum sodium levels and hydration status, 0.45% saline may later be considered.
Management of HHS focuses on four key principles: aggressive fluid resuscitation, gradual correction of hyperglycemia, electrolyte replacement, and identification of precipitating factors. Intravenous isotonic saline is the cornerstone of therapy because restoration of intravascular volume improves renal perfusion and enhances glucose clearance. In this case, normal saline was appropriately initiated to correct dehydration and reduce osmolality.
Insulin therapy is introduced after initial fluid replacement to avoid rapid shifts in osmolality. A low-dose continuous insulin infusion was used to gradually reduce blood glucose by approximately 50 mg/dL per hour, which aligns with recommended guidelines and minimizes the risk of cerebral edema. Once glucose levels approached 250 mg/dL, dextrose infusion was added to prevent an abrupt decline and to allow continued insulin administration for resolution of hyperosmolarity.
Electrolyte monitoring is essential, particularly potassium. Insulin drives potassium into cells, predisposing patients to hypokalemia, as observed in this case. Timely potassium supplementation was necessary to prevent cardiac arrhythmias and neuromuscular complications. Additionally, prophylactic low-molecular-weight heparin was justified because hyperosmolarity and dehydration increase blood viscosity and thrombotic risk.
Infections are common precipitants of HHS due to stress induced hormone release and insulin resistance. The markedly elevated leukocyte count raised suspicion of an underlying infection; therefore, empirical broad-spectrum antibiotics were initiated early, which is consistent with best practice.
Despite metabolic correction, the patient remained confused, reflecting delayed neurological recovery from prolonged hyperosmolality or a possible cerebrovascular event, warranting neuroimaging. Early recognition and adherence to evidence-based management were crucial in reversing the metabolic derangements and preventing further complications.
The cornerstone of Hyperosmolar Hyperglycemic State (HHS) management is the rapid correction of dehydration, gradual reduction of hyperglycemia, restoration of electrolyte balance—particularly potassium—and identification and treatment of precipitating factors such as infection. Effective management requires a structured and closely monitored approach:
Fluid Resuscitation: Immediate initiation of intravenous isotonic saline (0.9% NaCl) is essential to correct severe volume depletion and improve tissue perfusion. Depending on serum sodium levels and hydration status, 0.45% saline may later be considered.
Electrolyte Correction: Potassium levels must be monitored
closely, as insulin therapy and osmotic diuresis can precipitate
hypokalemia. Potassium should be added to intravenous fluids when
serum levels fall below the normal range (3.5–5.5 mmol/L).
Low-Dose Insulin Therapy: After initial fluid replacement, a
continuous intravenous insulin infusion (approximately 0.05 U/kg/
hour) is recommended to gradually reduce blood glucose, targeting
a decline of about 50 mg/dL per hour. Glucose levels are initially
maintained around 300 mg/dL to avoid rapid osmotic shifts and
cerebral edema; dextrose-containing fluids are introduced once
glucose falls below approximately 250 mg/dL.
Close Monitoring: Hourly blood glucose measurements, frequent electrolyte assessment, and continuous monitoring of vital signs are required, ideally in an intensive care setting.
Identification and Treatment of Precipitating Factors: Infections are the most common triggers of HHS and should be actively investigated and treated with appropriate antibiotics when present. Thromboprophylaxis: Due to increased blood viscosity and hypercoagulability associated with hyperosmolarity, low-molecularweight heparin is recommended unless contraindicated.
Although hyperglycemic crises are relatively common in individuals with diabetes, the occurrence of acute strokes in the immediate context of HHS and severe hypertension is uncommon. This case illustrates the complex interplay between type 2 diabetes mellitus, uncontrolled hypertension, seizures, and subsequent cerebrovascular insults. Early recognition of HHS and prompt initiation of evidence-based therapy were critical in stabilizing the patient. Management with isotonic saline, insulin infusion, potassium supplementation, and empirical antibiotics led to gradual improvement in metabolic parameters and mental status within 48 hours.
The underlying pathophysiology in this patient was likely multifactorial. An acute infection probably precipitated severe hyperglycemia, leading to osmotic diuresis, dehydration, and marked hyperosmolarity. These changes, compounded by uncontrolled hypertension, may have contributed to cerebral hypoperfusion and increased risk of stroke.
Close Monitoring: Hourly blood glucose measurements, frequent electrolyte assessment, and continuous monitoring of vital signs are required, ideally in an intensive care setting.
Identification and Treatment of Precipitating Factors: Infections are the most common triggers of HHS and should be actively investigated and treated with appropriate antibiotics when present. Thromboprophylaxis: Due to increased blood viscosity and hypercoagulability associated with hyperosmolarity, low-molecularweight heparin is recommended unless contraindicated.
Although hyperglycemic crises are relatively common in individuals with diabetes, the occurrence of acute strokes in the immediate context of HHS and severe hypertension is uncommon. This case illustrates the complex interplay between type 2 diabetes mellitus, uncontrolled hypertension, seizures, and subsequent cerebrovascular insults. Early recognition of HHS and prompt initiation of evidence-based therapy were critical in stabilizing the patient. Management with isotonic saline, insulin infusion, potassium supplementation, and empirical antibiotics led to gradual improvement in metabolic parameters and mental status within 48 hours.
The underlying pathophysiology in this patient was likely multifactorial. An acute infection probably precipitated severe hyperglycemia, leading to osmotic diuresis, dehydration, and marked hyperosmolarity. These changes, compounded by uncontrolled hypertension, may have contributed to cerebral hypoperfusion and increased risk of stroke.
Conclusion
HHS is a life-threatening complication of diabetes mellitus,
and when accompanied by severe hypertension, it may significantly
increase the risk of acute stroke. This case emphasizes the importance
of proactive metabolic surveillance and strict glycemic and blood
pressure control in patients with type 2 diabetes. Successful
management of HHS with coexisting hypertension and neurological
complications requires urgent, multidisciplinary intervention to
restore metabolic stability and prevent long-term morbidity and
mortality.
Ethics Approval:
This case report is based on a retrospective analysis of anonymized
patient data and does not constitute human subject’s research.
The report was prepared with the approval of the hospital ethics
committee.Conflicts of interest: The author declares no conflict of interest.
References
Citation
Das S. Management of Hyperglycemic Hyperosmolar State with Hypertension. Adv Diabetes Endocrinol 2026;9(1): 1.