Advances in Diabetes & Endocrinology
Download PDF
Case Report
Insulinoma Treated As Epilepsy; a Case of Misdiagnosis
Mian FJ1*, Yousuf Q2 and Khan AA3
1Consultant Physician in Endocrinology and Diabetes, UK
2Specialist Registrar Endocrinology and Diabetes UK
3Consultant Physician in Endocrinology and Diabetes, UK
2Specialist Registrar Endocrinology and Diabetes UK
3Consultant Physician in Endocrinology and Diabetes, UK
*Address for Correspondence Mian FJ, Consultant Physician in Endocrinology and Diabetes, Manor Hospital, Walsall Healthcare NHS Trsut Walsall, West Midlands, WS2 9PS, UK
Submission: 04-March, 2020
Accepted: 10-April, 2020
Published: 13-April, 2020
Copyright: © 2020 Mian FJ, et al. This is an open access article
distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium,
provided the original work is properly cited.
Abstract
Insulinoma is a rare neuroendocrine tumour. It can occur at any
age with nonspecific signs and symptoms. Due to its nonspecific
clinical features, insulinoma may be misdiagnosed with other disorders.
While evaluating a patient presenting with a seizure, toxic and
metabolic causes should always be considered as they are potentially
curable and can be fatal if left untreated. We describe a case of
hypoglycaemia induced seizures treated as epilepsy in a patient with
learning disability.
Keywords
Insulinoma; Epilepsy; Seizure; Learning disability
Case History
A49 year old female with learning disability and 2 years history of
epilepsy was admitted following a siezure like activity and fall during
a routine visit to her GP surgery. She had sustained injury to left
knee and x- ray confirmed a distal femur and a patellar fracture. On
admission her serum glucose was 2.0 mmol, all other routine blood
tests including Full Blood Count, Renal function and Liver function
test were normal. Her regular medications included Lamotrigine
and Mebeverien. On the ward she had recurrent spontaneous
hypoglycaemic episodes requiring regular treatment with glucose
infusions. The lowest capillary blood glucose recorded was 1.9
mmol. Due to the learning disability, patient could not describe
any symptoms usually experienced at the onset of hypoglycaemia.
Glycated Haemoglobin (HbA1c) was 19.0 mmol (Ref range: 20-
41 mmol) indicating lower than normal average glucose. Random
cortisol and thyroid function test were normal. She underwent left
knee exploration and repair of medial retinaculum of quadriceps
tendon and excision of patellar fragment. While awaiting results of
further investigations, Diazoxide was initiated with good effect. The
results of the specific biochemical investigations for her recurrent
hypoglycaemia were consistent with endogenous hyperinsulinaemic
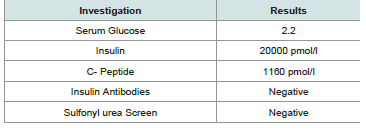
hypoglycaemia (Table 1).
A CT scan abdomen and pelvis was normal. Endoscopic
ultrasound was performed that showed a small 13 x 9 mm well defined
lesion with good doppler signals indicating high vascularity raising
the possibility of a Neuroendocrine tumour (Image 1). Biopsies
taken from the pancreatic lesion (Image 2) showed clusters of ovoid
epithelial cells with bland nuclei resembling those of neuroendocrine
cells that stained with synaptophysin and chromogranin in addition
to BerEP4 consistent with a well-differentiated neuroendocrine
tumour.
Patient was referred and discussed with the regional Neuroendocrine centre. The pancreatic lesion was surgically excised with
enucleation of the mass resulting in complete resolution of the
symptoms.
Discussion
Spontaneous hypoglycaemia can be difficult to diagnose in patients with a learning disability as patient may be unable to
describe hypoglycaemia related symptoms. Hypoglycaemia can
exhibit various neurogenic and neuroglycopenic symptoms. These
can mimic neuropsychiatric symptoms including unconsciousness,
confusion, seizure, personality change and bizarre behaviour in most
patients [1,2]. Over half of patients with these symptoms are initially
misdiagnosed with neuropsychiatric disorders such as epilepsy [1,3].
The siezures can be tonic-clonic, complex partial or absence seizures
[4-6]. However, correct diagnosis of epilepsy is also challenging in
clinical settings and can lead to inappropriate treatment with antiepileptic medication. In the setting of an Insulinoma, these symptoms
become typically evident after fasting and are often precipitated by
physical exercises. However, the median duration of symptoms
before diagnosis remains variable and can reach 12-18 months on
average or even years in rare cases [7].
An insulinoma is a rare pancreatic endocrine tumour that is
typically sporadic, solitary, and usually less than 2 cm in diameter.
It is reported in 1-4 people per one million person years1. Because
of the nonspecific symptoms, insulinoma may be misdiagnosed with
other disorders. It can be seen at any age and occurs slightly more
frequently in women than in men [8,9]. The clinical clues suggesting
insulinoma are based on the clinician`s recognition of the presence
of hypoglycaemic symptoms included in Whipple’s triad [10,11].
This triad includes symptoms of hypoglycaemia induced by fasting or
exercise, plasma glucose less than 2.5 mmol and relief of symptoms
following the administration of glucose. A previous diagnosis of
epilepsy and/or a drug history of anti-epileptic drugs can obscure
the clinical relationship between patient symptoms and possible
hypoglycaemia.
Most insulinomas are benign and are associated with MEN 1 in
5% of patients. It is estimated that 21% of patients with MEN1 develop
insulinomas [12,13]. The incidence is 3-10 cases per million people
per year. The insulinomas occurring in this autosomal dominant
syndrome have a higher risk of relapse [14].
The supervised 72 h fasting test remains the gold standard for
biochemical diagnosis with measurement of plasma glucose, insulin,
C-peptide, and proinsulin during the onset of hypoglycaemic
symptoms. Various preoperative procedures can be used to localize the
tumour in order to plan therapeutic strategy. The reported sensitivity
of conventional CT and MRI for detection of pancreatic insulinoma
ranges respectively from 33-64% and 40-90 % respectively. However,
the advent of helical CT scan has enabled detection of approximately
94% of insulinomas [15,16].
Endoscopic Ultrasound (EUS) is now largely considered as the
best investigation for preoperative localization of insulinoma with a
sensitivity of up to 94 %. It can detect even small tumours of up to 5
mm, and reveal important relation to the bile duct and adjacent blood
vessels. In addition EUS allows performing fine-needle aspiration cytology of suspicious lesions and preoperative marking of tumours
to facilitate surgical excision particularly with laparoscopic approach.
However, EUS findings largely depend on clinical experience [8,15].
Medical management of insulinoma, used to treat and prevent
hypoglycaemia, is generally restricted to un-resectable metastatic
tumours, unsuccessful operation with persistent symptoms,
inoperable patients, and patients waiting or refusing surgery [3,15].
Moreover, other recent techniques for the management of insulinoma
have been reported I Including injection of octreotide; EUS guided
alcohol ablation, radiofrequency ablation [17], or embolization of an
insulinoma.
Surgical excicion is the treatment of choice for most Insulinomas.
Tumour enucleation is the procedure of choice especially in case of
small and solitary nodule that is not encroaching on the pancreatic
or bile ducts [18]. More recently robotic enucleation of intrapancreatic Insulinoma has also been reported [11]. Pancreatic
resection is indicated for lesions invading or in close proximity to the
pancreatic duct or major vessels or suspicious for malignancy [19].
Resection options include distal pancreatectomy (with or without
splenectomy), Whipple procedure (pancreaticoduodenectomy), or
median pancreatectomy, depending on the site of insulinoma.
Insulinomas are typically reddish-brown, firm, and encapsulated
with a clear plane of dissection between the tumour and surrounding
soft pancreatic parenchyma [20].
Histologically, insulinomas are epithelial neoplasms associated
with strong and diffuse immunohistochemically expression of
neuroendocrine markers such as synaptophysin and chromogranin.
Mitotic rate (number of mitoses per 10 HPF) and proliferation index
(Ki-67 labelling index) are particularly helpful to separate welldifferentiated from poorly differentiated tumours [21,22].
Conclusion
Careful assessment is required to exclude hypoglycaemia as the
cause of siezures in all patients in general and particularly in patients
with learning disability as it is easily treatable and the underlying
cause such as Insulinoma potentially curable.
Recommendation
We recommend exclusion of
References
Citation
Mian FJ, Yousuf Q, Khan AA. Insulinoma Treated As Epilepsy; a Case of Misdiagnosis. Adv Diabetes Endocrinol 2020;5(1): 3.