Advances in Diabetes & Endocrinology
Download PDF
Research article
New Reference Values for Thyrotropin Hormone in Diabetic Patients
Nada AM1* and Shaheen D2
- 1Department of Internal Medicine, Mansoura University, Egypt
- 2Department of Biochemistry, Mansoura University, Egypt
*Address for correspondence: Mohamed Nada, Faculty of Medicine, Assistant Professor of Internal Medicine and Endocrinology, Department of Internal Medicine, Unit of Endocrinology & Diabetes, Mansoura University, Tell no: +20 102 741 3981, Egypt, E-mail: aml_nadanoha@yahoo.com
Citation: Nada M, Shaheen D. New Reference Values for Thyrotropin Hormone in Diabetic Patients. Adv Diabetes Endocrinol 2018;3(1): 9.
Advances inDiabetes & Endocrinology | ISSN: 2475-5591 | Volume: 3, Issue: 1
Submission: 28 November, 2018| Accepted: 28 December, 2018 | Published: 31 December, 2018
Copyright: © 2018 Nada M. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Objective: The present study aimed to find out how prevalent the thyroid disorders and the association of thyroid function with cardiovascular risk factors in type 2 diabetic patients..
Material and methods: This is a retrospective study which included 186 type 2 diabetic patients. Thyroid Function Tests (TFT), Fasting Plasma Glucose (FPG), Glycosylated Haemoglobin (HbA1c), Complete Blood Count (CBC), serum lipids and clinical data including Body Mass Index (BMI), blood pressure, and pulse rate and medications were traced.
Results: 9.7% of the study sample (186 patients) were on thyroxine therapy indicating previous diagnosis of clinical or subclinical hypothyroidism. Of the remaining number of patients (159), 15.1% had TSH value higher than the upper limit of reference range, 0.6% of patients showed suppressed TSH level. THS showed direct and significant association with Body Mass Index (BMI), Red cell Distribution Width (RDW) and Mean Platelet Volume (MPV). No significant association was found between thyroid function and glycemic control. At TSH value > 2.5 miU/ml, patients showed significantly higher BMI, serum triglycerides and white blood cell (WBC) count, all of which are cardiovascular risks.
Conclusion: Abnormal thyroid function is common in diabetic patients. TSH is directly and significantly associated with atherosclerotic and cardiovascular risk factors. We recommend a TSH value of 2.5 uIU/ml as an upper limit of normal thyrotropin in diabetic patients, as above this level cardiovascular risks are significantly higher.
Keywords
Diabetes; RDW; MPV; HbA1c; MPV; Triglycerides; TSH
Introduction
Prevalence of thyroid diseases in the general population rangesfrom 6.6% to 13.4%. This prevalence is increasing, particularly in women. Both diabetes mellitus and thyroid dysfunction are related to each other through complex biochemical, genetic, and hormonal malfunctions [1-5].
Diabetes is believed to affect the hypothalamo-pituitary thyroid axis as well as the peripheral conversion of tetraiodo to triiodothyronine. Thyroid hormones; namely Triiodothyronine (FT3) and Tetraiodothyronine (FT4) are considered as insulin antagonists which potentiate insulin action indirectly. Low thyroid hormones in iabetic patients may be explained by the decreased production of thyrotropin releasing hormone [6,7].
According to Lang, muscles support the bone structure, particularly during the process of growth and development [7]. The close relationship between muscle and bone and this relationship are observed through the mechanostat theory, evaluated firstly to determine gender differences by Frost [8,9]. During the attainment of aging, peak bone and muscle strength, both men and women begin to lose both bone and muscle tissues and possibilities of muscles and bone damage widely known as sarcopenia [3-7].
Prevalence of abnormal thyroid function in T2DM is comparableto that in T1DM as per available reports although the genetic links are less clear. Despite the evident association between diabetes and hyroid dysfunction, there is no special recommendation for thyroid screening as part of diabetes health care. Guidelines are vague with a lot of variation ranging from ignoring the thyroid function test to yearly testing [8,9].
Recently, thyroid diseases including subclinical hypothyroidism, MPV and RDW showed strong relationship to cardiovascular and atherosclerotic heart diseases and are associated with higher morbidity and mortality [10-12].
Materials and Methods
This is a retrospective cross-sectional study conducted at king Fahd Hospital, a tertiary care hospital, in Asir province, Saudi Arabia. A sample of 270 Saudi type 2 diabetic patients who had attended the endocrinology and diabetes clinics between July 2016 and July 2017 were randomly selected for the study. Their medical records were reviewed. The final number of patients after applying the inclusion and exclusion criteria was 186. Inclusion criteria were type 2 diabetes, of both sexes, whose age was above 18 years. The exclusion criteria included anemia, pregnancy, postpartum period and type 1 DM. Individuals with previous history of thyroid disease, co-existing hepatobiliary disease, heart failure, history of myocardial infarction or on systemic drug therapy such as, glucocorticoids or oral contraceptives were not included in the study. Patients were also excluded if they had recent acute condition such as acute respiratory or gastrointestinal infection within one month before data collection. Clinical data such as age, sex, weight, height, BMI and blood pressure were collected from medical records. BMI was calculated as per the equation: BMI = weight (kg)/ height (m2). The Modification of Diet in Renal Disease (MDRD) study formula was used for calculation of eGFR [13].
Glucose oxidase method (Spinreact, Girona, Spain) was used to measure fasting plasma glucose. Total Hb was measured colorimetrically. HbA1c was determined immunoturbidimetrically and was expressed as a percentage of Hb by a conversion equation to match a HPLC reference method; HbA1c (%) = HbA1c/Hb × 175.8 + 1.73 [14].
Total cholesterol, HDL-cholesterol, and TG were measured by BioMerieux Laboratory, Marcy l′ France; LDL-cholesterol was calculated by Friedewald′s formula; LDL-C = TC - HDL-C - TG/5 [15].
Urea and creatinine were performed respectively with an enzymatic kinetic UV assay and a kinetic colorimetric assay based on the Jaffe method on Cobas c701 (Roche Diagnostics, Mannheim) according to the manufacturer′s instructions. TSH, FT3, FT4 were measured by Hemiluminescence Immunoassay (CLIA) [Immunospec Corporation, Canoga Park, CA, USA] [16].
Our laboratory reference rangesa
RBC 4.5-6.3 109/L, WBC 4-11 109/L, MCH 26-36 pg, MCHC 32- 36 gm/dL, HCT 38-52%, Hb 14-18 gm/dL, Platelets 140-440 109/L, RDW 11-14%, MPV 7-13 fL. TSH: 0.35-4.9 uIU/ml, FT4: 7.5-21.1 pmol/l, FT3: 3.8-7.8 pmol/l, TC: 0-5.2 mmol/l, LDL-C: 3-5.2 mmol/l, HDL-C: 1.04-1.55 mmol/l, TG: 0.34-1.95 mmol/l, creatinine 80-115 μmol/l, Blood Urea Nitrogen (BUN): mmol/l. Anemia was defined by hemoglobin level <13 g/dl in men and <12 g/dl in women [17].
Thyroid hypofunction was considered when TSH was > 4.94 uIU/ ml and hyperfunction when TSH was < 0.3 uIU/ml. SPSS software (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.) was used for data entry and analysis. Qualitative data were expressed as count and percent. Quantitative data were initially tested for normality using Kolmogorov-Smirnov and Shapiro-Wilk′s test with data being normally distributed if p>0.050. Quantitative data were expressed as mean± Standard Deviation (SD) if normally distributed or median and Interquartile Range (IQR) if not. Qualitative data for two groups (2×2 table) were compared using Chi-Square test (or Fisher′s exact test). Quantitative data between two groups were compared using Independent-Samples t-test if data were normally distributed in both groups. Mann-Whitney U test was used if data are not normally distributed. Pearson correlation coefficient, denoted as r, and it is the coefficient that measures the strength and direction of a linear relationship between two continuous variables. Its value can range from -1 for a perfect negative linear relationship to +1 for a perfect positive linear relationship. A value of 0 (zero) indicates no relationship between two variables. The Spearman’s rank-order correlation calculates a coefficient, rs, which is a measure of the strength and direction of the association/relationship between two continuous or ordinal variables. The diagnostic performance of a test, or the accuracy of a test to discriminate diseased cases from nondiseased cases is evaluated using Receiver Operating Characteristic (ROC) curve analysis (Metz, 1978; Zweig & Campbell, 1993). For any of the used tests, p value ≤0.05 indicated statistical significance. Appropriate charts were used to graphically present the results whenever needed including scatterplot [18].
Results
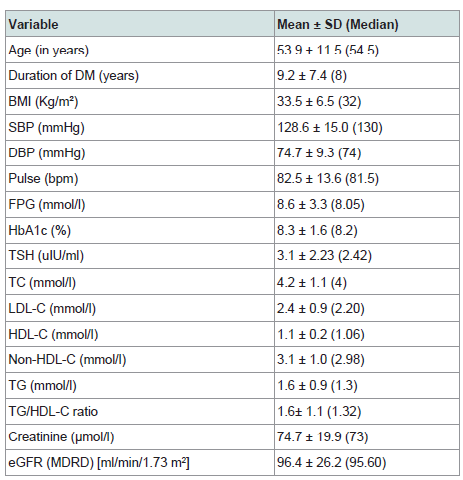
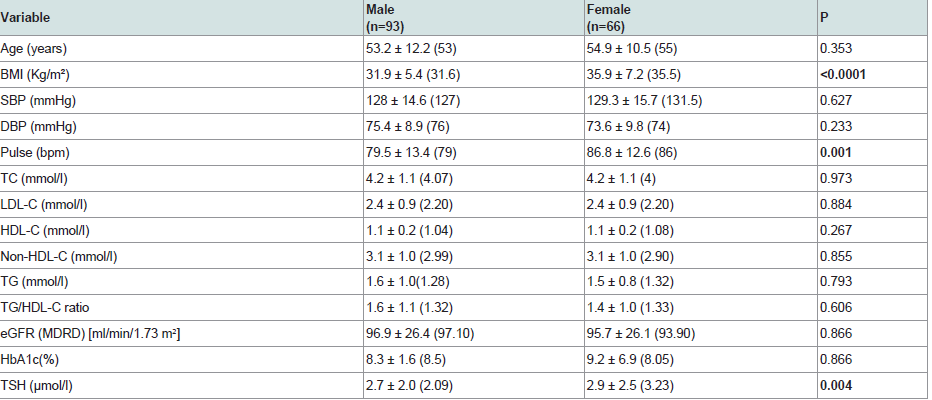
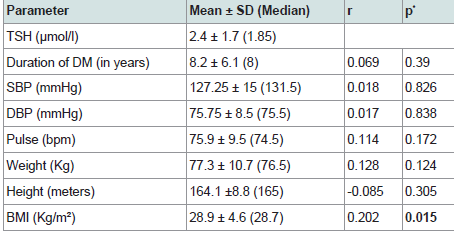
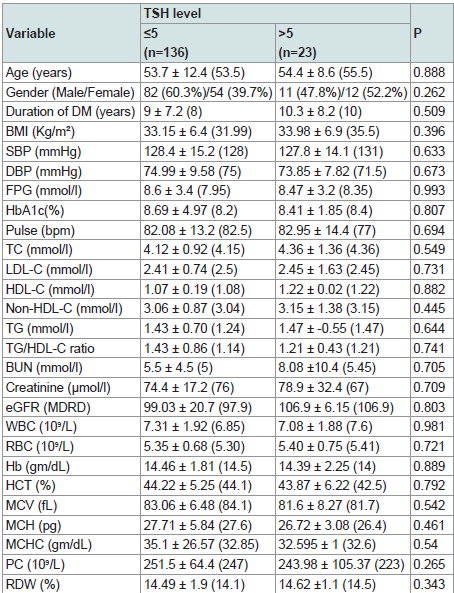
186 patients were valid for screening as they had TSH level as the screening test for thyroid function. Out of 186 patients, 18 patients (9.7% of the study sample) were on thyroxine replacement therapy indicating initial diagnosis of clinical or subclinical hypothyroidism. Of the 186 patients, 27 patients were excluded because of insufficient other data required for further statistical analysis. So, our statistical analysis was applied to 159 patients who were of a mean age 53.9±11.5 years, diabetes duration 9.2±7.4 years, HbA1c 8.6±4.7% and TSH level 3.05±2.23 uIU/ml. Descriptive analysis is shown in (Table 1). Comparison of various parameters in males and females showed higher TSH, BMI and heart rate in females (p = 0.004, < 0.0001, 0.001 respectively) while, serum creatinine and BUN were significantly higher in males (p < 0.0001, 0.011 respectively). No significant gender difference was noticed in the other study variables (Table 2).
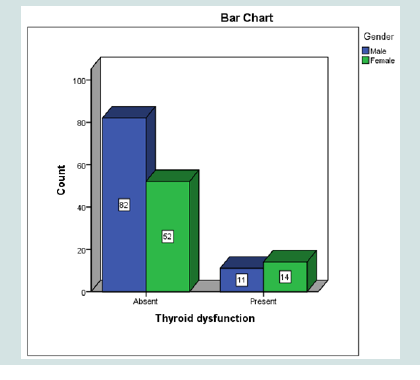
Thyroid dysfunction was found in 15.7 % of patients. High TSH above the reference range was found in 24 patients (15.1%) indicating clinical or subclinical hypothyroidism and suppressed in 1 patient (0.6%) indicating thyroiditis or subclinical hyperthyroidism requiring further investigations. Prevalence was comparable among males and females (p= 0.119) (Figure 1).
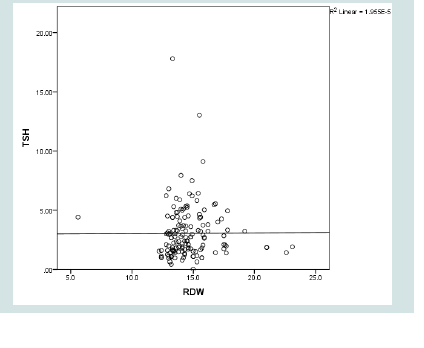
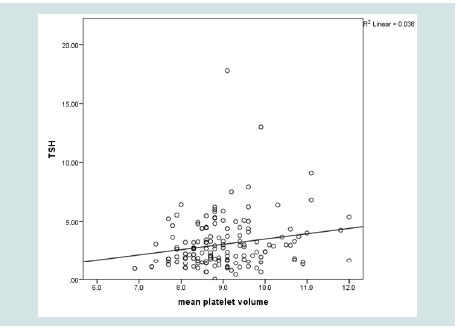
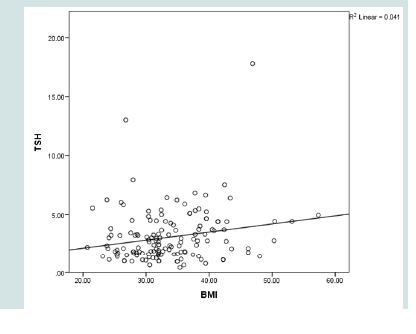
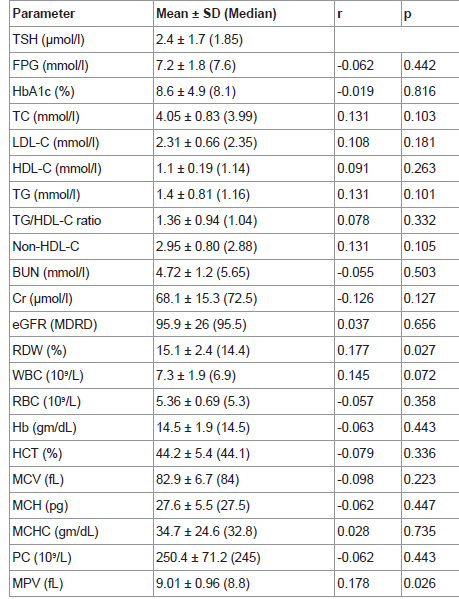
TSH significantly and directly correlated to RDW, MPV and BMI (p= 0.027, 0.026, 0.015 respectively). Neither FT4 nor FT3 showed such significant correlations. No significant association was found between TSH and duration of diabetes, FPG, HbA1c or lipid profile (Table 3 and 4 & Figure 2-4).
At TSH value of 5 uIU/ml as a cut off for hypothyroidism, no significant difference between patients on either side of this level as regard to any of the measured clinical or laboratory variables (Table 5).
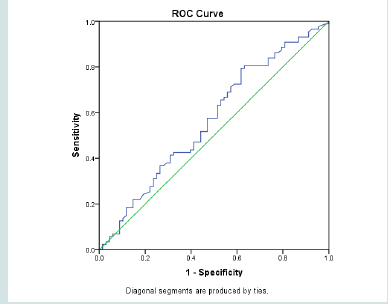
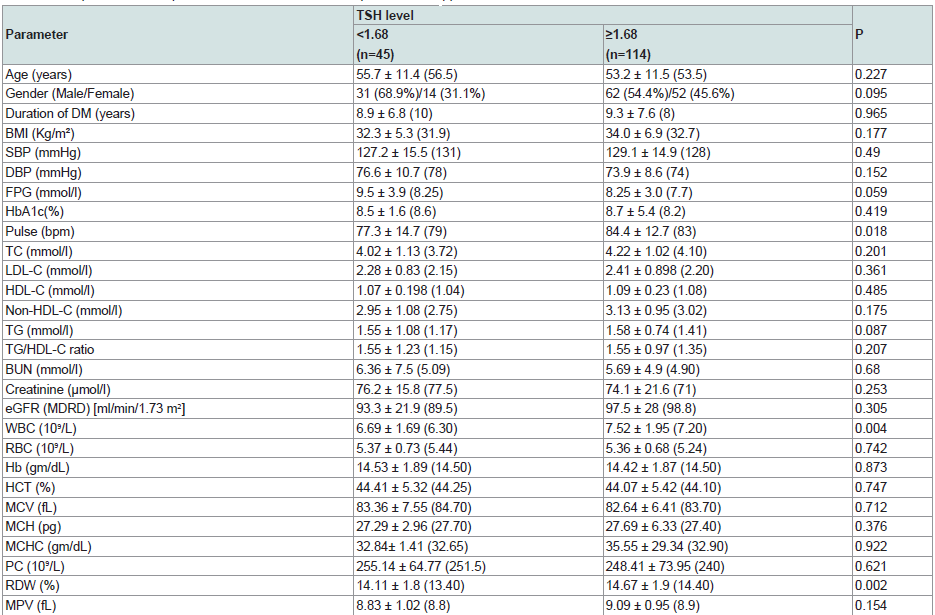
Because of the direct linear relationship between TSH level and RDW, using the highest point of the reference range of RDW, defined a cut off value of TSH of 1.68 uIU/ml with sensitivity of 79% but with low specificity 38% (Figure 5).
Comparing various clinical and laboratory variables on both sides of TSH 1.68 uIU/ml did showed significant difference in WBC and pulse rate (p= 0.004, 0.018 respectively), being higher in the higher TSH group (Table 6).
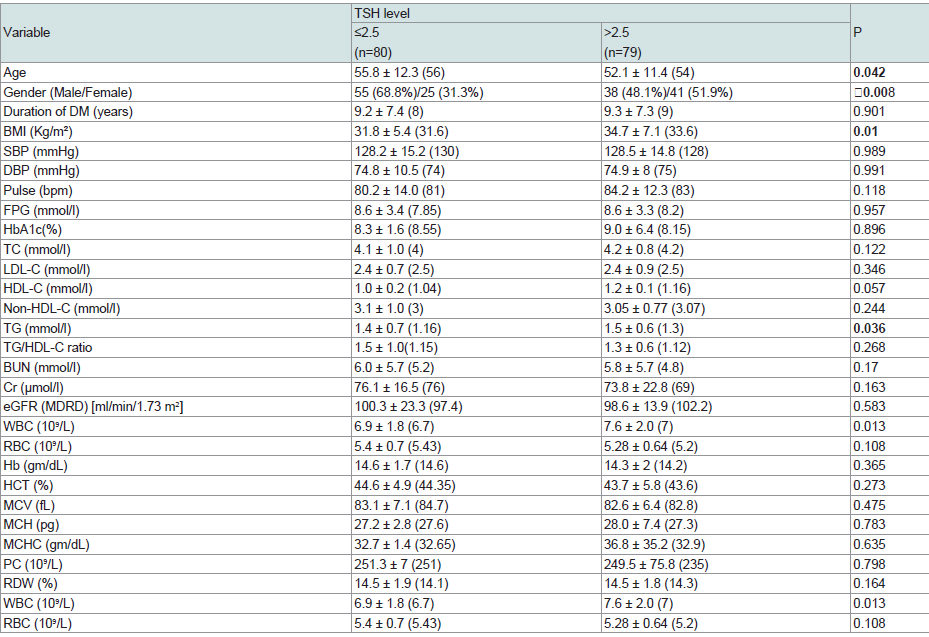
Patients with TSH> 2.5 uIU/ml were significantly younger in age (p= 0.042) and more females than males (p= 0.008). BMI, triglycerides and WBC were significantly higher in the higher TSH group (p= 0.01, 0.036, 0.013 respectively) (Table 7).
Discussion
Similar to our finding of higher BMI in diabetic Saudi females than males (<0.0001), Kamath showed that a larger percentage of type 2 diabetic Indian females had significantly higher central and general obesity in comparison to males (p= <0.001) [19].
The observed lower serum creatinine in female patients can be explained by lower muscle mass than males while their higher TSH in our study is reported by many other authors [2,20-22].
Noteworthy is our observation of sex difference in heart rate being higher in females. This was also mentioned in other reports [23,24].
Third generation TSH level is of above 98% sensitivity and 92% specificity in detection of both hypo and hyperfunction. So, in our study TSH was the main hormone available as a screen for thyroid function [3,25].
Many studies showed higher prevalence of thyroid dysfunctionamong diabetic population in comparison with non-diabetic subjects. One of meta-analyses reported a frequency of 11%. Perros reported a prevalence of 13.4%, being 31.4% in females as compared to 6.9% in males [26-28].
In other population studies, thyroid disorders affected 16% of diabetic patients meanwhile Celani reported a prevalence of 9.7% [29,30].
In Saudi Arabia, Akbar reported a prevalence of 16 % while in another retrospective study, Al-Geffari found thyroid disorders in 28.5%, of the study group, with 25.3% as hypothyroidism and 3.2% as hyperthyroidism. Al-Geffari′s report is one of the highest prevalence reports of thyroid dysfunction in T2DM [7,31,32].
In our study of Saudi population, the prevalence of thyroid dysfunction was 15.7%, 23.8% in females, 13 % in males without gender preference (p-value= 0.119).
Al-Geffari explained his observation by the high prevalence of Latent Autoimmune Diabetes of Adult (LADA) in Saudi type 2 diabetic patients reaching 26%. This was evident in his study population with high prevalence of autoimmune thyroid diseases. He also mentioned several risk factors for thyroid dysfunction of which are family history of thyroid disease, female gender, and diabetes duration more than 10 years. In our study, gender and duration of diabetes and its control did not correlate to thyroid dysfunction. Female gender was a risk factor for developing thyroid dysfunction in several studies while age, duration and control of diabetes were not in others. In Afkhami- Ardekani′s report, HbA1C was significantly higher patients who had both diabetes and abnormal thyroid function [28-34].
Being a retrospective study, we could not trace the presence of goitre or family history of thyroid dysfunction and no data were available about thyroid auto antibodies. Hyperthyroidism is a less common thyroid dysfunction in both general and diabetic patients. This is very clear in our study where we found only one case of subclinical thyrotoxicosis that may indicate the presence of thyroiditis or subclinical hyperthyroidism [29,35].
Although thyroid hormones affect insulin sensitivity, absence of an association between thyroid disease and glycemic control may be explained by the other various factors that can play a role in the control of diabetes including patient lifestyle, compliance to treatment and other used medications.
In a Jordanian study, prevalence was 12.5 %. In other gulf countries like Oman, overall prevalence was 12.5 % and was higher in females (p= 0.01) [31,36].
In concordance with several reports our study showed a strong direct association between TSH level and BMI (p=0.015) [19,37,38].
Similar to Khurana, we did not find an association between TSH and serum lipids. However, most of our patients were on hypolipidemic agents mainly statins that may altered their lipid profile [39].
Presence of an association between thyrotropin and both RDW and MPV in the current study is in agreement with both Geetha and Yu′s reports. Kim and Lippi also demonstrated the strong positive association between thyrotropin and MPV. In Lippi′s study, high TSH even with normal FT4 was an independent predictor of high MPV. Both RDW and MPV are believed to be risk factors for cardiovascular diseases. So the association between TSH and both risk factors in diabetic patients indicates the significance of screening and defining an upper limit of TSH reference in this special category of people who are at higher risk for cardiovascular disease due to diabetes [35,40-44].
Since the 2002 Consensus Conference on subclinical thyroid disease studies showed that even slight elevation of TSH level (3-10 mIU/liter) can increase the risk of atherosclerosis in some individuals who are susceptible, like diabetic patients [45-48].
In 2002, the American Thyroid Association and American Association of Clinical Endocrinologists recommended that each laboratory should define its TSH reference values. In iodine sufficient areas, TSH range of 0.45-4.12 mIU/L should be considered if the laboratory reference is not available patients having TSH level higher than 10 mIU/L are recommended for thyroxine replacement to avoid heart failure and cardiovascular mortality. When TSH is in the range of upper normal level to 10 mIU/L, the need for thyroxine intake should be individualized according to several factors such as the presence of symptoms of hypothyroidism, autoantibodies, heart failure, atherosclerosis or risk factors for these diseases [49].
Several reports confirmed the improvement of markers of atherosclerosis such as serum lipids and carotid intima media thickness with thyroxine treatment even with very mild elevation of Several reports confirmed the improvement of markers of atherosclerosis such as serum lipids and carotid intima media thickness with thyroxine treatment even with very mild elevation of TSH [50]. Patients at high risk for ASCVD with TSH levels of 6.1- 10 mIU/L or higher than 10 mIU/L and their age was less than 65 years who did not receive thyroid hormone treatment had higher allcause mortality when compared with those who started their thyroid replacement therapy [51].
In a UK study, individuals with TSH between 5.01 and 10 mIU/L and normal FT4 in the age group of 40 to 70 years and older were followed for about 8 years. The hazard ratio for ischemic heart disease events significantly decreased in patients who received thyroxine treatment when compared to those who did not take the medicine (0.61, CI 0.49-0.92). This reduction was not observed in patients older than 70 years [52].
The American Association of Clinical Endocrinologists recommended a TSH upper limit of 3.0 mIU/liter as this level was associated with lowest prevalence of thyroid autoimmunity in NHANES III study [53].
In our study considering TSH upper limit of 5 µiU/ml could not detect a significant difference in CVD risks between the groups of patients having a value below and above this limit. On the other hand, a TSH level of 1.68 µiu/ml as an upper limit, was defined retrospectively, as the figure at which there is increase in RDW above the upper normal value (14%). This showed significant differences in RDW and WBC count. At a value of 2.5 µiU/ml as an upper limit of reference range, people with TSH higher than this level showed higher BMI, Triglycerides and WBC in comparison to those having lower TSH values [54-56].
BMI is well known to be associated with cardiovascular and coronary heart diseases and their associated risk factors such as hypertension and hyperlipidemia [57,58].
Hypertriglyceridemia is one of the criteria of the metabolic syndrome and is strongly associated with atherosclerosis, hypercoagulable and proinflammatory states [59].
Breakdown of triglycerides leads to release of Free Fatty Acids (FFA) into the circulation. Long term exposure of beta-cell of the pancreas to FFA and deposition of triglycerides in the pancreatic islets leads to suppression of insulin secretion [60].
Atherosclerosis is associated with an underlying inflammatory process. WBC is considered as one of the inflammatory markers that can predict the future development of a cardiovascular insult. Higher WBC is associated with higher body weight, systolic blood pressure, and insulin level; all of which are cardiovascular risks. Several studies found that higher WBC is an independent predictor of coronary heart disease in diabetic patients in our study, higher WBC was found in association with TSH value higher than 2.5 uIU/ml indicating a higher risk of cardiovascular disease [61,62].
Lower reference values of thyrotropin such as 3 or 2.5 uIU/ml were previously suggested, however, this suggestion was criticized. Lowering the upper limit of TSH reference was favored by the observation of the presence of thyroid autoantibodies in subjects with TSH level between 3 to 5 uIU/ml, and those were at the highest risk to develop clinical thyroid dysfunction [57,58,63,64].
Lowering TSH value to a level less than 3 uIU/ml through titration of thyroxine dosage, was associated with a significant improvement in lipid profile, symptomatology and cardiovascular health [2,65-68].
In NHANES III, mean serum TSH was 1.5 uIU/ml in healthy persons who did not have goiter, thyroid autoantibodies or family history of thyroid abnormality and highest value was 2.5 uIU/mlin. Similarly, African-Americans, known with the lowest susceptibility to Hashimoto thyroiditis; had a mean TSH of 1.18 uIU/ml indicating the vulnerability of this figure as a mean for healthy people [69,70].
In our study, TSH, at different cut off values, is associated withdifferent risks for atherosclerosis and CVD. So, it is reasonable to recommend a TSH of 2.5 μiu/ml as an upper limit for normal thyroid function in type 2 diabetic population and advice a TSH level ≤ 1.68 uIU/ml as a target for diabetic patients on thyroxine replacement. We agree with other authors (2,70) in that decreasing the upper limit of reference range of TSH to 2.5 uIU/ml will help early detection of patients at risk of thyroid hypofunction with follow up and early management to avoid any complication.
Conclusion
Our study indicates that a routine screening for thyroid dysfunction is justified in T2DM as it is associated with many CVDS risks. We recommend a TSH of > 2.5 uIU/ml as an upper limit to diagnose thyroid hypofunction in T2DM. We also advise that diabetic patients who are receiving thyroid replacement therapy to attain a TSH level ≤ 1.68 uIU/ml.
References
- Silva RC (2005) Importância da avaliação da função tireoidiana em pacientes com diabetes mellitus. Arq Bras Endocrinol Metabol 49: 180-182.
- Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, et al. (2002) Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87: 489-499.
- Lippi G, Montagnana M, Salvagno GL, Guidi GC (2009) Should women with abnormal serum thyroid stimulating hormone undergo screening for Anaemia. Arch Pathol Lab Med 133: 1-4..
- Brenta G, Danzi S, Klein I (2007) Potential therapeutic applications of thyroid hormone analogs. Nat Clin Pract Endocrinol Metab 3: 632-640.
- Goglia F, Moreno M, Lanni A (1999) Action of thyroid hormones at the cellular level: the mitochondrial target. FEBS Lett 452: 115-120.
- Suzuki Y, Nanno M, Gemma R, Tanaka I, Taminato T, et al. (1994) The mechanism of thyroid hormone abnormalities in patients with diabetes mellitus. Nihon Naibunpi Gakkai Zasshi 70: 465-470.
- Perros P, McCrimmon RJ, Shaw G, Frier BM (1995) Frequency of thyroid dysfunction in diabetic patients: value of annual screening. Diabet Med 12: 622-627.
- Johnson JL (2006) Diabetes control in thyroid disease. Diabetes Spectr 19:148-153.
- Michael L (2004) Screening for thyroid dysfunction: US preventive services task force recommendation statement. Ann Intern Med 140: 125-127.
- Razvi S, Ingoe L, Keeka G, Oates C, McMillan C, et al. (2007) The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J Clin Endocrinol Metab 92: 1715-1723.
- van Kimmenade RR, Mohammed AA, Uthamalingam S, van der Meer P, Felker GM, et al. (2010) Red blood cell distribution width and 1-year mortality in acute heart failure. Eur J Heart Fail 12: 129-136.
- Kodiatte TA, Manikyam UK, Rao SB, Jagadish TM, Reddy M, et al. (2012) Mean platelet volume in type 2 Diabetes mellitus. J Lab Physicians 4: 5-9.
- Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, et al. (2006) Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med 145: 247-254.
- European Association for the Study of Diabetes (EASD) News Section 5/2004. Report of the ADA/EASD/IDF workin group of the HbA1c assay. London, UK. January 20.
- Friedewald WT, Lvey RI, Fredrickson DS (1972) Estimation of the concentration of low density lipoprotein cholesterol in plasma without the use of the preparative ultracentrifuge. Clin Chem 18: 499-502.
- Chen CK, Tsai KS (1996) Clinical application of chemiluminescent immunoassay for thyroid stimulating hormone, free-T4 and intact-parathyroid hormone. J Formos Med Assoc 95: 197-202.
- McDonnell CH, Jeffrey RB Jr, Bjorkengren AG, Li KC (1992) Intraarticular sonography for imaging the knee menisci: evaluation in cadaveric specimens. Am J Roentgenol 159: 573-574.Nutritional anaemias. Report of a WHO scientific group (1968) World Health Organ Tech Rep Ser 40: 1-40.
- Metz CE (1978) Basic principles of ROC analysis. Semin Nucl Med 8: 283-298.
- Kamath A, Shivaprakash G, Adhikari P (2011) Body mass index and Waist circumference in Type 2 Diabetes mellitus patients attending a diabetes clinic. Int J Biol Med Res 2: 636-638.
- Perrone RD, Madias NE, Levey AS (1992) Serum creatinine as an index of renal function: new insights into old concepts. Clin Chem 38: 1933-1953.
- Andersen S, Bruun NH, Pedersen KM, Laurberg P (2003) Biologic variation is important for interpretation of thyroid function tests. Thyroid 13: 1069-1078.
- Gonzalez-Sagrado M, Martin-Gil FJ (2004) Population-specific reference values for thyroid hormones on the Abbott ARCHITECT i2000 analyzer. Clin Chem Lab Med 42: 540-542.
- Greet LR, Youri ET, Jean M K, Van Daele CM, De Buyzere ML, et al. (2013) Thyroid hormone levels within reference range are associated with heart rate, cardiac structure, and function in middle-aged men and women. Thyroid 23: 947-954.
- Silva de Paula R, Antelmi I, Vincenzi MA, André CD, Artes R, et al. (2005) Influence of age, gender, and serum triglycerides on heart rate in a cohort of asymptomatic individuals without heart disease. Int J Cardiol 105: 152-158.
- Helfand M, Crapo LM (1990) Testing for suspected thyroiddisease. In: Sox HC, ed. Common Diagnostic Tests. Philadelphia: American College of Physicians.
- Feely J, Isles TE (1979) Screening for thyroid dysfunction in diabetics. Br Med J 1: 1678.
- Kadiyala R, Peter R, Okosieme OE (2010) Thyroid dysfunction in patients with diabetes: clinical implications and screening strategies. Int J Clin Pract 64: 1130-1139.
- Akbar DH, Ahmed MM, Al-Mughales J (2006) Thyroid dysfunction and thyroid autoimmunity in Saudi type 2 diabetics. Acta Diabetol 43: 14-18.
- Celani MF, Bonati ME, Stucci N (1994) Prevalence of abnormal thyrotropin concentrations measured by a sensitive assay in patients with type 2 diabetes mellitus. Diabetes Res 27: 15-25.
- Al-Geffari M, Ahmad NA, Al-Sharqawi AH, Youssef AM, AlNaqeb D, et al. (2013) Risk factors for Thyroid Dysfunction among Type 2 Diabetic patients in a highly diabetes mellitus prevalent society. Int J Endocrinol. PP: 6.
- Radaideh AR, Nusier MK, Amari FL, Bateiha AE, El-Khateeb MS, et al. (2004) Thyroid dysfunction in patients with type 2 diabetes mellitus in Jordan. Saudi Med J 25: 1046-1050.
- Díez JJ, Sánchez P, Iglesias P (2011) Prevalence of thyroid dysfunction in patients with type 2 diabetes. Exp Clin Endocrinol Diabetes 119: 201-207.
- Díez JJ, Iglesias PJ (2014) Serum cholesterol and triglyceride concentrations in diabetic patients with subclinical hypothyroidism. Endocrinol Nutr 61: 419-425.
- Afkhami-Ardekani M, Rashidi M, Shojaoddiny A (2010) Effect of Thyroid Dysfunction on Metabolic Response in Type 2 Diabetic Patients. Iran J Diabetes Obesity 2: 20-26.
- Al-Sumry S, Al-Ghelani T, Al-Badi H, Al-Azri M, Elshafie K (2015) Thyroid Diseases in Omani Type 2 Diabetics: A Retrospective Cross-Sectional Study. Adv Endocrinol. pp: 6.
- Stanická S, Vondra K, Pelikánová T, Vlcek P, Hill M, et al. (2005) Insulin sensitivity and counter-regulatory hormones in hypothyroidism and during thyroid hormone replacement therapy. Clin Chem Lab Med 43: 715-720.
- Papazafiropoulou A, Sotiropoulos A, Kokolaki A, Kardara M, Stamataki P, et al. (2010) Prevalence of thyroid dysfunction among Greek type 2 diabetic patients attending an outpatient clinic. J Clin Med Res 2: 75-78.
- Proces S, Delgrange E, Vander Borght TV, Jamart J, Donckier JE (2001) Minor alterations in thyroid function tests associated with diabetes mellitus and obesity in outpatients without known thyroid illness. Acta Clin Belg 56: 86-90.
- Khurana A, Dhoat P, Jain G (2016) Prevalence of thyroid disorders in patients of type 2 diabetes mellitus. JIACM 17: 12-15.
- Wang C (2013) The relationship between type 2 diabetes mellitus and related thyroid diseases. J Diabetes Res..
- Geetha JP, Srikrishna R (2012) Role of red blood cell distribution width (rdw) in thyroid dysfunction. Int J Biol Med Res 3: 1476-1478.
- Yu HM, Park KS, Lee JM (2014) The value of red blood cell distribution width in subclinical hypothyroidism. Arq Bras Endocrinol Metabol 58: 30-36.
- Kim JH, Park JH, Kim SY, Bae HY (2013) The mean platelet volume is positively correlated with serum thyrotropin concentrations in a population of healthy subjects and subjects with unsuspected subclinical hypothyroidism. Thyroid 23: 31-37..
- Lippi G, Danese E, Montagnana M, Nouvenne A, Meschi T, et al. (2015)Mean platelet volume is significantly associated with serum levels of thyroid stimulating hormone in a cohort of older euthyroid subjects. Endocr Res 40: 227-230.
- Lippi G, Filippozzi L, Montagnana M, Salvagno GL, Franchini M, et al. (2009) Clinical usefulness of measuring red blood cell distribution width on admission in patients with acute coronary syndromes. Clin Chem Lab Med 47: 353-357.
- Markovic D, Carevic V, Bonacin D, Sekulić BP, Sapunar A, et al. (2013) Correlation between mean platelet volume and total risk of cardiovascular disease. Signa vitae 8: 49-55.
- Gharib H, Tuttle RM, Baskin HJ, Fish LH, et al. (2005) Subclinical thyroid dysfunction: a joint statement on management from the American Association of Clinical Endocrinologists, the American Thyroid Association, and the Endocrine Society. J Clin Endocrinol Metab 90: 586-587.
- Wartofsky L, Dickey RA (2005) The evidence for a narrower thyrotropin reference range is compelling. J Clin Endocrinol Metab 90: 5483-5488.
- Razvi S, Ingoe L, Keeka G, Oates C, McMillan C, et al. (2007) The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J Clin Endocrinol Metab 92: 1715-1723.(
- Kvetny J, Heldgaard PE, Bladbjerg EM, Gram J (2004) Subclinical hypothyroidism is associated with a low-grade inflammation, increased triglyceride levels and predicts cardiovascular disease in males below 50 years. Clin Endocrinol (Oxf) 61: 232-238.
- Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, et al. (2012) ATA/ AACE Guidelines: Clinical Practice Guidelines for Hypothyroidism in Adults: Cosponsored by the American Association Of Clinical Endocrinologists and the American Thyroid Association.. Endocr Pract. pp: 1-45.
- Dardano A, Monzani F (2007) Thyroid function and carotid artery intimamedia thickness. Circ J 71: 993.
- McQuade C, Skugor M, Brennan DM, Hoar B, Stevenson C, et al. ( 2011) Hypothyroidism and moderate subclinical hypothyroidism are associated with increased all-cause mortality independent of coronary heart disease risk factors: a PreCIS database study. Thyroid 21: 837-843.
- Baskin HJ, Cobin RH, Duick DS, Gharib H, Guttler RB, et al. (2002) American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocr Pract 8: 457-469.
- Canaris GJ, Manowitz NR, Mayor G, Ridgway EC (2000) The Colorado thyroid disease prevalence study. Arch Intern Med 160: 526-534.
- Spencer CA, Hollowell JG, Kazarosyan M, Braverman LE (2007) National Health and Nutrition Examination Survey III thyroid-stimulating hormone (TSH)-thyroperoxidase antibody relationships demonstrate that TSH upper reference limits may be skewed by occult thyroid dysfunction. J Clin Endocrinol Metab 92: 4236-4240.
- Wartofsky L, Dickey RA (2005) The evidence for a narrower thyrotropin reference range is compelling. J Clin Endocrinol Metab 90: 5483-5488.
- Body mass index and cardiovascular disease risk factors in seven Asian and five Latin American centers: data from the International Clinical Epidemiology Network (INCLEN). Obes Res 4: 221-228.
- Novo S, Peritore A, Trovato RL, Guarneri FP, Di Lisi D, et al. (2013)Preclinical atherosclerosis and metabolic syndrome increase cardio- and cerebrovascular events rate: a 20-year follow up. Cardiovasc Diabetol 12: 155.Ganguly A (2015) Obtaining normal flexion and extension of knee joints on supine, prone and standing positions in osteoarthritis by topical phytotherapeutic treatment irrespective of age and sex. Int J Phytomed 7: 290-301.
- Haber EP, Procópio J, Carvalho CR, Carpinelli AR, Newsholme P, et al. (2006) New insights into fatty acid modulation of pancreatic beta-cell function. Int Rev Cytol 248: 1-41.
- Lee CD, Folsom AR, Nieto FJ, Chambless LE, Shahar E, et al. (2001) White blood cell count and incidence of coronary heart disease and ischemic stroke and mortality from cardiovascular disease in African-American and white men and women: autherosclerosi risk in communities study. Am J Epidemiol 154: 758-764.
- Saito I, Folsom AR, Brancati FL, Duncan BB, Chambless LE, et al. (2000) Nontraditional risk factors for coronary heart disease incidence among persons with diabetes: the Atherosclerosis Risk in Communities (ARIC) study. Ann Intern Med 133: 81-91.
- Surks MI, Hollowell JG (2007) Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab 92: 4575-4582.
- Vanderpump MP, Tunbridge WM, French JM, Appleton D, Bates D, et al. (1995) The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf) 43: 55-68.
- Baloch Z, Carayon P, Conte-Devolx B, Demers LM, Feldt-Rasmussen U, et al. (2003) Guidelines Committee, National Academy of Clinical Biochemistry. Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid 13: 3-126.
- Michalopoulou G, Alevizaki M, Piperingos G, Mitsibounas D, Mantzos E, et al. (1998) High serum cholesterol levels in persons with ‘high-normal’ TSH levels: should one extend the definition of subclinical hypothyroidism. Eur J Endocrinol 138: 141-145.
- Monzani F, Di Bello V, Caraccio N, Bertini A, Giorgi D, et al. (2001) Effect of levothyroxine on cardiac function and structure in subclinical hypothyroidism: a double blind, placebo-controlled study. J Clin Endocrinol Metab 86: 1110-1115.
- Lekakis J, Papamichael C, Alevizaki M, Piperingos G, Marafelia P, et al. (1997) Flow-mediated, endothelium-dependent vasodilatation is impaired in subjects with hypothyroidism, borderline hypothyroidism, and high-normal serum thyrotropin values. Thyroid 7: 411-414.
- Bakker SJ, ter Maaten JC, Popp-Snijders C, Slaets JP, Heine RJ (2001) The relationship between thyrotropin and low density lipoprotein cholesterol is modified by insulin sensitivity in healthy euthyroid subjects. J Clin Endocrinol Metab 86: 1206-1211.
- Metz CE (1978) Basic principles of ROC analysis. Semin Nucl Med 8: 283-298.