Journal of Veterinary Science & Medicine
Download PDF
Research Article
Gross and HistopathologicalStudy of the Genitalia in Goats:1. Ovaries
Mukhtar Rafiq Hama Karim1 and Faraidoon A.Sattar Muhammad2*
- 1Directorate of Veterinary Hospital in Sulaimani, Sulaimani Province, Kurdistan Region, Iraq
- 2Department of Surgery and Theriogenology, College of Veterinary Medicine, Sulaimani University, Sulaimani, Kurdistan Region, Iraq
*Address for Correspondence: Faraidoon A. Sattar Muhammad, Department of Surgery and Theriogenology, College of Veterinary Medicine, Sulaimani University, Sulaimani, Kurdistan Region, Iraq, E-mail: araidoon.muhamad@univsul.edu.iq
Citation: Karim MR, Faraidoon A, Muhammad S. Gross and Histopathological Study of the Genitalia in Goats: 1. Ovaries. J Veter Sci Med. 2017;5(1): 4.
Copyright: © 2017 Karim MR. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Veterinary Science & Medicine | ISSN: 2325-4645 | Volume: 5, Issue: 1
Submission: 10 January, 2017| Accepted: 02 February, 2017 | Published: 07 February, 2017
Abstract
Among the 340 samples collected from the pregnant and nonpregnant animals, 126(37.058%) were pregnant, 127(37.35%) were found without gross pathological abnormalities, and the remaining 43(21.64%) samples were found with gross and pathological abnormalities of ovaries. The present study is achieved to find out the gross and pathological lesions in ovaries of goats. The ovarian lesions were 72 (21.176%) abnormalities in 340 specimens recorded, the most common lesions were ovaro-bursal adhesion 28 cases (26; 7.647% mild and 2; 0.588% severe), para-ovarian cysts (22; 6.470%), follicular cystic ovary (7; 2.058%). Histopathologically, the follicular cysts characterized by the presence of a large empty cyst in the ovarian stroma surrounded by proliferating theca cells layer, and marked proliferation of granulosa cells layer. Cystic corpus luteum was (5; 1.470%), luteal cystic ovary (4; 1.175%). Microscopically, the cysts were lined by a fibrous layer surrounded by a luteinized theca cells layer. Other rare pathological conditions of the ovaries were an ovarian hematoma 4 (1.176%), ovarian tuberculosis, 1 (0.294%). Histopathologically, ovarian tuberculosis is characterized by typical granulomatous tubercle presence in the ovarian stroma characterized by pinkish caseous necrosis in the center surrounded with thick purple granuloma. Ovarian Cysticercus tenuicollis was 1(0.294%). It can be concluded that ovarian abnormalities constitute a percentage (21.176%), and ovarian tuberculosis was firstly recorded in Iraq and Kurdstan region.
Keywords
Ovaries; Goats genitalia; Abnormalities; Ovarian tuberculosis
Introduction
The Kurdish goat is multipurpose animal provide an enormous range of products including meat, milk, skin for leather making and cashmere and mohair fibers production [1,2]. Reproduction of the goat is the immediate importance to the goat keepers because it has a high fertility rate and produces much youngs in a year [3,4]. Thus, abnormalities of goat reproductive organs have a significant effect on its fertility [5]. Reproduction in goats defined as seasonal, various factors affecting the seasonality include latitude, climate, breed, physiological stage, presence of male, breeding system, andspecifically photo period [6]. In general, the breeding season starts in autumn and ends in winter, with anoestrus in spring/summer [7].
In Kurdistan, very few works have been done on this effect. Although gross lesions of the female genital tracts make the fertility be reduced in the goats to varying degree, and tiny information is known about incidence and nature of the abnormalities in Sulaimani region [4].
The gross and pathological abnormalities of reproductive organs have been surveyed and reviewed by various authors [8,9].
Macroscopic abnormalities in pregnant and non-pregnant Iraqi goats were 14.69% [5]. At Sulaimani abattoir, the incidence of gross abnormalities in non-pregnant genitalia was 16% while in the pregnant genitalia macroscopic abnormalities were 9.42% [10]. Alwan and Amin observed that the ovarobursal adhesion is the most common condition, which is about 5.66% of the ovarian lesions in Sulaimani Region. In the Iraqi goats, the most common ovarian abnormality seen in abattoir was a para-ovarian cyst 3.95% [5]. The incidence of a follicular cyst in Pakistan was 0.24% [11], in Uganda 3.8% [12], in Kurdistan the percentage is 4%, recorded by [4]. Incidence of luteal cyst was 0.17% [12], while in Kurdistan was 2% in non-pregnant goats [4].
The present study was conducted to determine the microscopic and macroscopic lesions of ovarian abnormalities in goats slaughtered at the abattoir of Sulaimani province - Kurdistan Region of Iraq.
Materials and Methods
The study was conducted on 340 samples of goats´ genitalia which were collected on three days per week from the Slaughter house that located in “Qaragol” district in Sulaimani province. Samples were collected during the period of the beginning of September 2014 to the June 2015. No information regarding the identity and history of the animals included in this study were obtained. The genitalia were separated immediately after evisceration at the slaughter house. The broad ligaments, the loose connective tissues, and the fat surrounding the vulva and retroperitoneal part of the vagina and other genitalia were removed as far as possible to clear the reproductive organs for a better examination. The flexures of the uterine tubes were straightened out by freeing them from the mesosalpinx. The samples were being placed in plastic bags and transported to the laboratory of Veterinary Teaching Hospital of the College of Veterinary Medicine at the University of Sulaimani.
The ovaries were the first part of the genitalia to be examined. Each ovary of the non-pregnant genitalia was carefully observed. Externally and internally for the presence of different types of gross pathological abnormalities like tumors, cysts (follicular, luteal and para ovarian cyst), inflammatory conditions, congestion, and other visible changes. Ovaries with cysts greater than 1 cm in diameter were considered pathological cyst, based on the appearance of the cysts, were classified into follicular and luteinized cysts. Follicular cysts were tensor than luteal cysts, and thin-walled while luteinized cysts were presented as thick wall cysts. Ovarian bursa examined for adhesions; adhesions ranged from a fine strand of connective tissues between the ovarian bursa and the ovary ovaro- bursal adhesion to severe adhesions, were completely embedded in fibrous tissues. Then the data were recorded in percentage as well as photographed. A tissue sample from the affected ovaries (section of approximately one cubic centimetre) is being fixed in a plastic container that contains 10% buffered formalin for histopathological examination.
Table 1: Gross pathological abnormalities of the ovaries in goats´ genitalia in the Sulaimani abattoir.
Results
The genitalia from 340 goats were examined 126(37.05%) were pregnant. 127(37.35%) genitalia without gross pathological abnormalities in Table 1.
Discussion
The present study describes most of the lesions that occurred in the ovaries of goats slaughtered in Sulaimani slaughterhouse (Table 1).
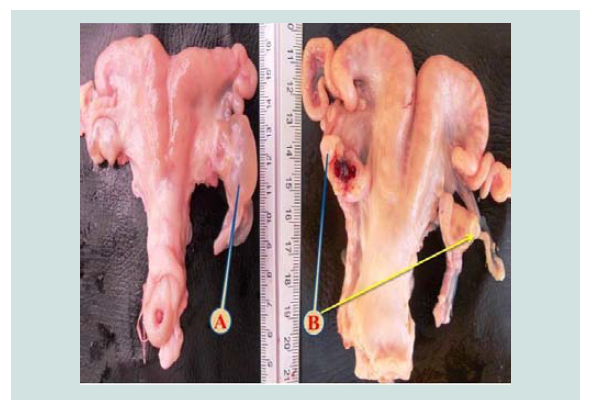
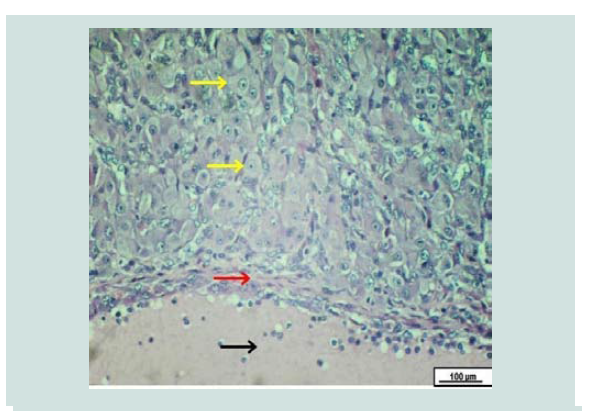
Figure 3: Photomicrograph of the broad ligament. The red arrows reveal an empty para-ovarian cyst embedded within a loose connective tissue of broad ligament. Black arrows show muscle bundle. The cyst surrounded by a fibromuscular thick wall lined by simple epithelium (yellow arrows). H&E. Scale bar: 500 µm.
The total percentage of ovarian abnormalities 72(21.17%) from 43 genitalia, this result was higher than that recorded by other workers [11,13-15] with the incidence of 6.07%, 5.21%, 11.8%, and 10.4%, respectively. The significant differences in the incidence of obvious abnormalities found in the various areas, and might be due to differences in breed, species, and age [5]. While a high number of genital lesions in goats, especially in Iraqi Kurdistan region, probably is due to many factors that include: poor housing, inadequate knowledge of the owner&acus, bad managements, insufficient nutrition level, and increased contamination [4,10].
Figure 5: Photomicrograph of an ovary demonstrates excessive fluid filled follicular cyst within the ovarian stroma; The cyst is surrounded by proliferated theca layer cells (yellow arrow). Marked proliferation of granulosa cells within the ovarian cyst (red arrow). H&E. Scale bar: 200 µm.
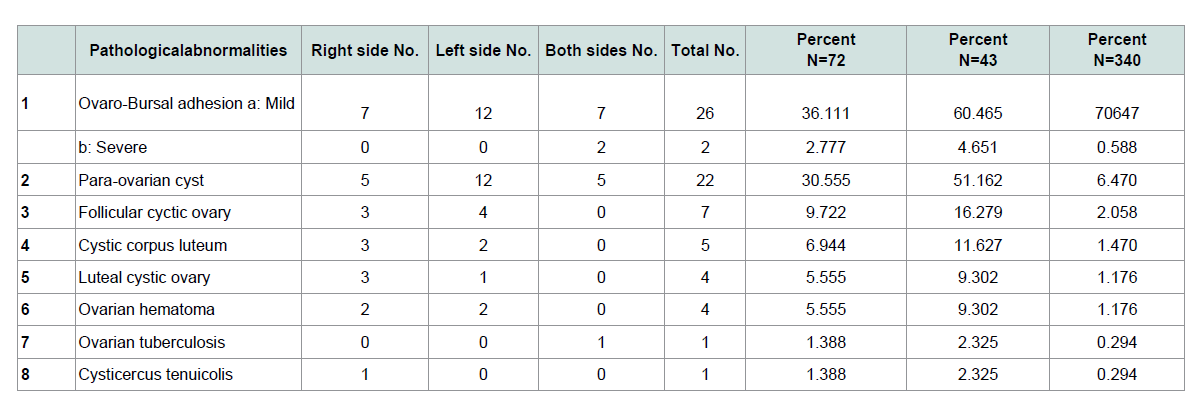
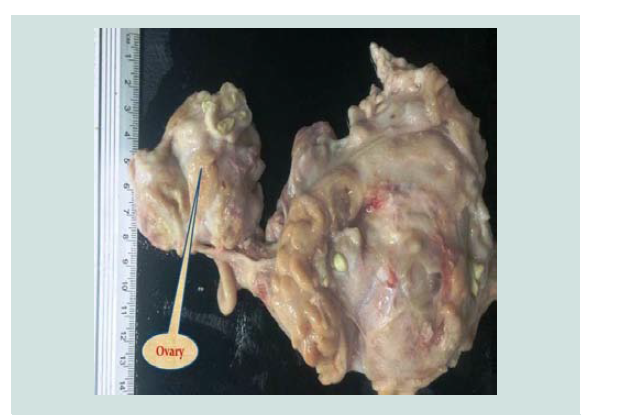
The total incidence of ovaro-bursal adhesion Figure 1 was 8.235% of which mild ovaro-bursal adhesion was 7.647%. Severe ovaro-bursal adhesion was 0.588% that found in non-pregnant goats (Table 1), both cases of severe ovaro-bursal adhesion were bilateral and completely enveloped the ovary. This result is higher than that reported by [5,11,16] which were 2.99%, 2.47% and 1.96%, respectively. Ovaro-bursal adhesion may have resulted from ovulatory processes [17]. Ovulation has been regarded as a substantial hazard that involves slight hemorrhage [18] and inflammatory process [19].
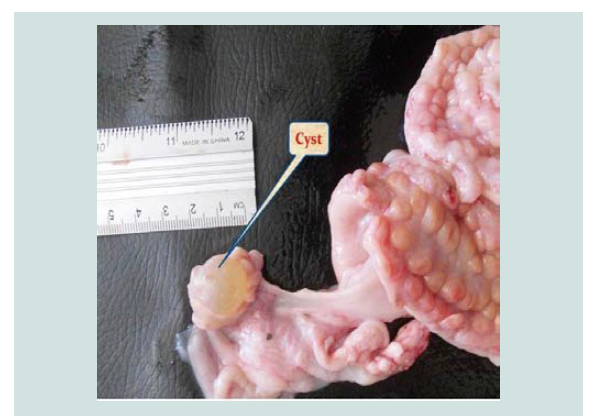
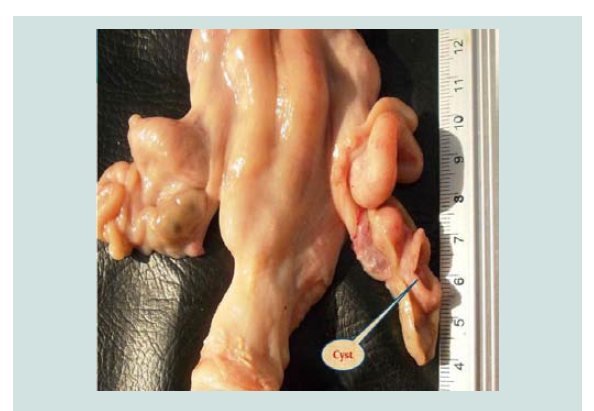
Para-ovarian cysts Figure 2 were observed in 22(6.470%) cases in non-pregnant goats (Table 1). These findings higher than the results of [5] who recorded 3.95% in Iraq [20], recorded 3.2% in Sri Lanka while [11] registered 1.36% in Pakistan. Para-ovarian cysts derived from the mesonephros [21]. All the cases detected were not capable of interfering with the infertility of the goats unless they cause pressure on the uterine tubes. Histologically Figure 3 the lesion was composed of the cysts that embedded in loose connective tissues of broad ligaments and the cyst surrounded by a thick fibromuscular tissue.
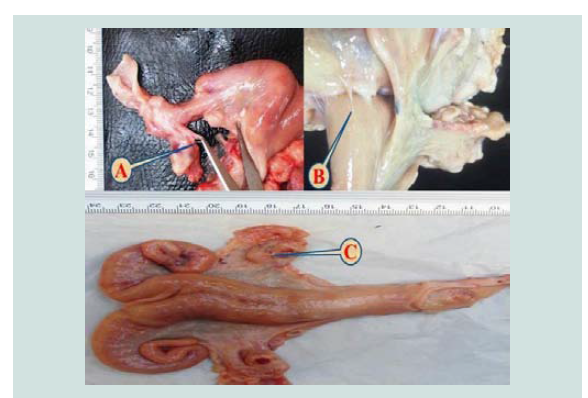
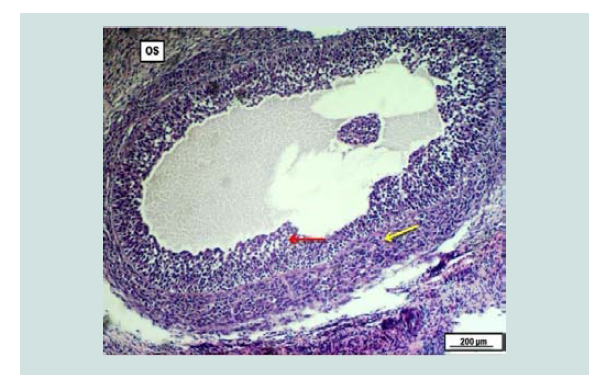
The incidence of cystic ovaries (follicular and luteal cystic) might be attributed to insufficient luteinizing hormone before or at the time of ovulation, age of animals and nutrient deficiency [22]. The effect of uterine infection on ovarian function could be due to the role of endotoxins that are produced by gram-negative bacteria that prevents preovulatory LH surge and ovulation causing persistent ovarian cysts [23]. Follicular cystic ovary Figure 4 had a prevalence of (2.058%); all of the cases were unilateral. This result was higher than that reported by other workers [11-13] which were 0.24%, 1.88% and 0.08%, respectively. While lower than the results recorded by Amin, Alwan and Kumar, they recorded 4%, and 7%, subsequently [4,10,15]. Histologically, cystic Graffian follicles characterized by a large fluid-filled cyst in the ovarian stroma and surrounded by a thick layer of the theca cell layer with the proliferation of granulosa cell layer in the lumen of the cyst Figure 5. These findings have been described by [12,13].
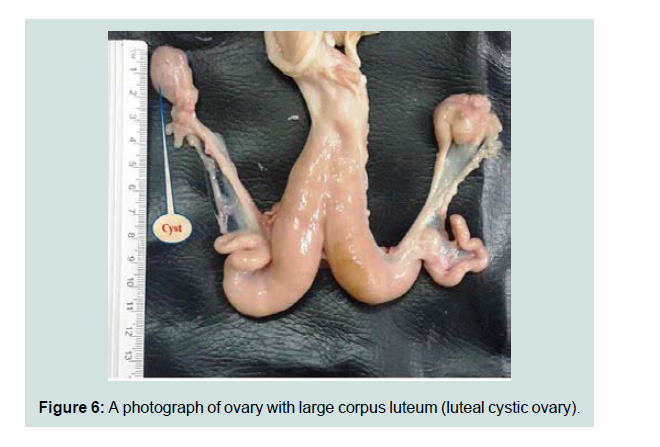
The total incidence of the luteal cystic ovary was 1.175%. All the cases were unilateral having thick walled and the size of the cyst about 2.2 cm in diameter Figure 6. Histopathologically, the cyst was contains excessive pinkish fluid in the ovarian stroma and surrounded by a layer of luteinized cells (Figure 7). These lesions were similar to that observed by other workers [11,24]. This observation was higher than the results reported by Al-Rawi which was 0.66% and according to Francis who recorded 1.4% [12,25].
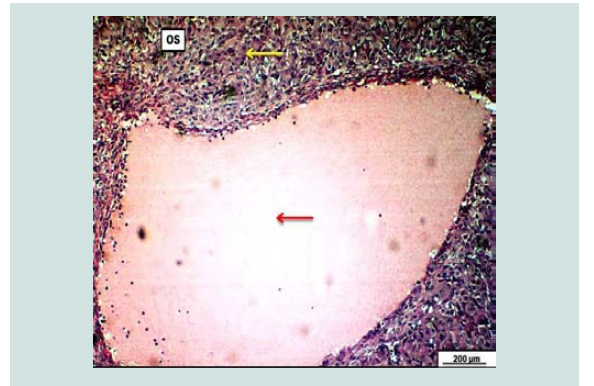
Figure 7: Photomicrograph of an ovary demonstrates significant fluid filled luteal cyst within the ovarian stroma; The cyst is surrounded by massively proliferated luteal cells (yellow arrow). The cyst filled with pinkish fluid (redarrow). H&E. Scale bar: 200 µm. A photograph of ovary with large corpus luteum (luteal cystic ovary).
Figure 9: Photomicrograph of an ovary shows proliferated luteinized cells (yellow arrow), a thin layer of fibrous connective tissues between luteal cells and the lumen of the cystic cavity (red arrow), the black arrow shows the lumen of the cyst. H&E. Scale bar: 100 µm.
Cystic corpora lutea were observed in 5(1.470%) cases from the total numbers of genital tracts. All of the cases were unilateral. This observation is higher than the results that reported by Ogunbodede et al. who reported 0.77%, and lower than those reported by Sattar and Khan that reported 2.08% and Mohammad that registered 5.44% in cows in Sulaimani [11,26,27] (Figure 8). Microscopically, the cysts have central cavities that surrounded by luteinized cells, and a thin layer of fibrous connective tissues between the luteal cells and the lumen of the cyst (Figure 9) that is in agreement with [13,28].
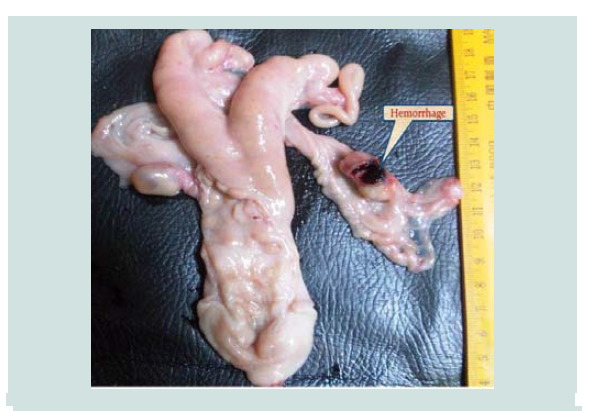
Hematoma (Figure 10) of the ovary occurs at regular ovulation and in an anovulatory follicle. Four cases of ovarian hematoma were observed in the present study and the percentage was (1.175%). One case (0.5%) of ovarian hematoma was recorded by Fathalla et al. in cows [23]. An ovarian hematoma occurs as results of blood losses due to rupture of the follicles and may resolve spontaneously with no effect on the fertility (Smith) [29]. Histologically, it characterized by the presence of massive and extensive hemorrhagic blood clots within the ovarian stroma that surrounded by infiltrating inflammatory cells (Figure 11). This finding were agrees with that reported by Fathalla et al. [23].
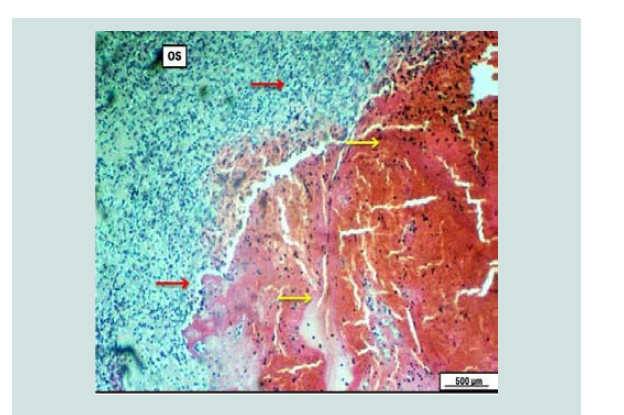
Figure 11: Photomicrograph of the ovary reveals massive and extensive clotted hemorrhagic blood mass embedded within the ovarian stroma (yellow arrows). The hemorrhagic mass is surrounded by diffuse inflammatory exudate (red arrows). H&E. Scale bar: 500 µm.
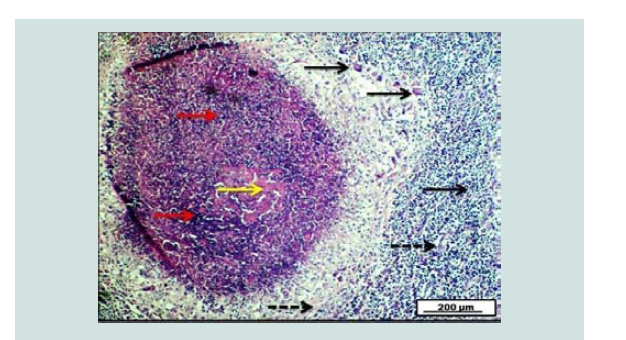
Figure 13: Photomicrograph of ovary demonstrates the presence of typical granulomatous tubercle that is characterized by the presence of pinkish center of caseous necrosis (yellow arrow) interlaced with thick purple granuloma (red arrows). The granulomatous lesion is surrounded by many epithelioid, and langhan’s giant cells (black arrows), together with the proliferation of fibrous connective tissue within the lesion (dashed black arrows). H&E. Scale bar: 200 µm.
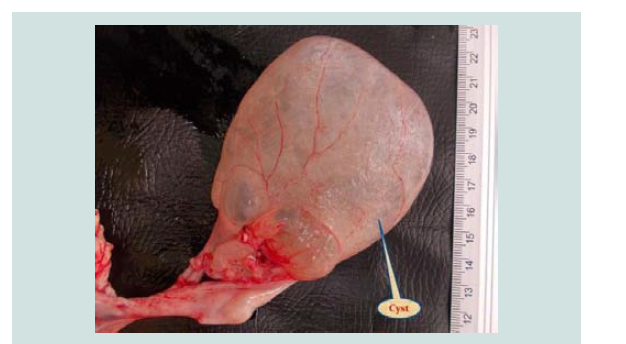
Figure 14: A photograph of ovary shows large bladder like cyst (filled with serofibrinous fluid and floated to the ovary).
Tuberculosis in goats caused by the Mycobacterium bovis, goats are more susceptible than sheep for the disease [30]. The ovarian tuberculosis was reported in the present study and had a prevalence of (0.294%). In Jordan, this disease also recorded in the ovaries by Fathalla et al. with the incidence of 0.5% in cows [23]. The incidence of bovine tuberculosis in Sulaimani region was 5.1% and 8.9% as reported by and Ali [31,32]. Grossly, both ovaries dotted by some tubercles. The size of each tubercle varies from 1-3 cm (Figure 12). Microscopically, the typical granulomatous tubercle characterized by the presence of typical granulomatous tubercle that is described by the presence of the pinkish center of caseous necrosis interlaced with thick purple granuloma. The granulomatous lesion is surrounded by many epithelioid and Langhans giant cells, together with the proliferation of fibrous connective tissues within the lesion (Figure 13). These lesions agree with that observed by Schmitt et al. in the deer [33]. Tuberculosis disease is of public health and of economic importance throughout the world [34]. The ovarian tuberculosis in goats is the pathological abnormality that has not been reported by Iraqi researchers previously [35].
Ovarian Cysticercus tenuicolis cyst was also observed in one (0.294%) case and was attached to the serosa of the right ovary (Figure 14).
References
- Gall C (1996) Goat breeds of the world. Backhuys Publishers, Cornell University.
- Simith MC, Sherman DM (2009) Goat medicine (2ndedn). Wiley-Blackwell
- Jansen C, Burg, KV (2004) Goat keeping in the tropics (4thedn). Agromisa Foundation, Wageningen, Netherlands.
- Amin FA (2009) Biometrical, histological and hormonal study of pregnant and non-pregnant genitalia of sheep and goat in Sulaimani region. University of Sulaimani, Iraq Sulaimani.
- Al-Baggal HA, Al-Dahash SY, Alwan AF (1993) Macroscopic study of the female genital system in Iraqi goats. Small Ruminant Res 9: 341-346.
- Fatet A, Pellicer-Rubio MT, Leboeuf B (2011) Reproductive cycle of goats. Anim Reprod Sci 124: 211-219.
- Brunet AG, Santiago-Moreno J, Toledano-Díaz A, Lopez-Sebastian A (2012) Reproductive seasonality and its control in Spanish sheep and goats. Trop Subtrop Agroecosystems 15: S47-S70.
- Roberts SJ (1986) Veterinary obstetrics and genital diseases theriogenology (3rdedn). Ann Arbor Science.
- Khodakaram-Tafti A, Davari A (2013) Congenital and acquired abnormalities of reproductive tract of non-pregnant ewes slaughtered in Fars province, Iran. Iranian J Vet Res 14: 140-144.
- Alwan AF, Amin FA (2010) Anatomical and histological lesions causing infertility in local goats (Sulaimania Region) AL-Qadsiya. J Vet Med Sci 138-146.
- Sattar A, Khan MZ (1988) Incidence and pathology ovarian diseases of goats. Pak Vet J 8: 18-21.
- Francis M (2009) Caprine ovarian and uterine lesions: an abattoir survey. Makerere University, Institutional Respository, Uganda.
- Hatipoglu F, Kiran MM, Ortatatli M, Erer H, Ciftci MK (2002) An abattoir study of genital pathology in cows: I. Ovary and oviduct. Revue Med Vet 153: 29-33.
- Rahman MH, Chowdhury EH, Saha SS, Islam A, Alam MG (2008) Abattoir study of reproductive diseases in goats. Bangladesh Vet 25: 88-91.
- Agrawal, Kumar J (2012) Genital status and reproductive disorders in small ruminants-an abattoir investigation. CSKHPKV Respository.
- Alwan AF (1998) Gross abnormalities affecting female genital tracts of Iraqi ruminants. Vet 8: 23-29.
- Regassa F, Mengesha D, Dargie M, Tolosa T (2009) Abattoir evidence on association between uterine and ovarian abnormalities in Ethiopian highland ewes. Anim Reprod Sci 111: 384-390.
- Arthur GH, Noakes DE, Parkinson TJ, England GC (1996) Veterinary reproduction and obstetrics. Saunders.
- Espey LL (1994) Current status of the hypothesis that mammalian ovulation is comparable to an inflammatory reaction. Biol Reprod 50: 233-238.
- Abeyratne AS, Atureliya DS (1982) Studies on the reproductive organs in the indigenous female goat of Sri Lanka. Ceylon Vet J 27: 17-19.
- Kennedy PC, Miller RB (1993) The female genital system. In: Jubb KV, Kennedy PC, Palmer N (Eds). Pathology of domestic animals (4thedn), Academic Press, Inc, USA, 3: 343-471.
- Karsch FJ, Battaglia DF, Breen KM, Debus N, Harris TG (2002) Mechanisms for ovarian cycle disruption by immune/inflammatory stress. Stress 5: 101-112.
- Mateus L, Lopes da Costa L, Diniz P, Ziecik AJ (2003) Relationship between endotoxin and prostaglandin (PGE2 and PGFM) concentrations and ovarian function in dairy cows with puerperal endometritis. Anim Reprod Sci 76: 143-154.
- Fathalla M, Hailat N, Lafi SQ, Basha EA, Al-Sahli AA (2000) An abattoir survey of gross reproductive abnormalities in the bovine genital tract in Northern Jordan. Isr J Vet Med 55: 83-87.
- Alrawi BT (2006) Morphological and histopathological study of goats ovary. College of Veterinary Al.Anbar University, Iraq.
- Ogunbodede MA, Oladele GM, Ode OJ, Ubah SA (2014) Survey of gross abnormalities and microbial load on the female reproductive tract of Maradi goats slaughtered at Bodija abattoir, Nigeria. Advanced J Agr Res 2: 1-7.
- Mohammad MO (2015) Pathological study of ovaries and uterine tubes in slaughtered non-pregnant cows in Sulaimani Province. Collage of Veterinary Medicine, University of Sulaimani, Iraq.
- McEntee K (1990) Reproductive Pathology of Domestic Mammals. Sandigo, California: Academic Press, Inc.
- Smith BP (2014) Large animal internal medicine (5thedn). Elsevier Health Sciences.
- Pugh DG, Baird N (2012) Sheep and goat Medicine (2ndedn). Elsevier Health Sciences.
- Solaiman SG (2010) Goat science and production. Wiley.
- Ali KN (2014) In vitro investigation of bovine tuberculosis in Sulemani province. Collage of Veterinary Medicine, University of Sulaimani, Iraq.
- Schmitt SM, Fitzgerald SD, Cooley TM, Bruning-Fann CS, Sullivan L, et al. (1997) Bovine tuberculosis in free-ranging white-tailed deer from Michigan. J Wildl Dis 33: 749-758.
- Colby MM, Smith MA, Wineland NE (2000) A survey of brucellosis and tuberculosis surveillance among sheep and goats in the United States. Proceedings of the 9th International Symposium on Veterinary Epidemiology and Economics.
- Sattar A, Khan MZ, Siddique M (1988) Incidence, pathology and bacteriology of abnormalities of fallopian tubes in goats. Pak Vet J 8: 14-17.