Journal of Pharmaceutics & Pharmacology
Download PDF
Special Issue:Drug DeliveryResearch Article
*Address for Correspondence: Kumi Kawano, Institute of Medicinal Chemistry, Hoshi University, Ebara 2-4-41, Shinagawa, Tokyo 142-8501, Japan, Tel: +81-3-5498-5097; E-mail: kkumi@hoshi.ac.jpCitation:Miatmoko A, Kawano K, Hattori Y, Maitani Y, Yonemochi E. Evaluation of Transfersome and Protransfersome for Percutaneous Delivery of Cisplatin in Hairless Mice. J Pharmaceu Pharmacol. 2015;S(1): 7.Copyright © 2015 Kawano et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.Journal of Pharmaceutics & Pharmacology | ISSN: 2327-204X | Special Issue: 1Submission: 16 March, 2015 | Accepted: 13 April, 2015| Published: 17 April, 2015Editor:Dr. Vinayagam Kannan, Scientist Par Sterile Products, LLC, USA
Keywords: Cisplatin; Transdermal delivery; Protransfersome; Biodistribution study
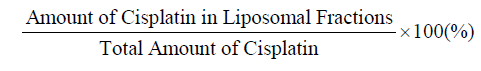 Microscopic evaluation of protransfersomeProtransfersome was observed microscopically by placing a small amount of sample on the object glass. A drop of physiological saline was added to the sample through the cavity side of the cover slip. The photomicrographs before, during, and after saline addition were taken at suitable magnifications under an optical microscope (NikonEclipse TS100-F, Nikon Inc., Tokyo, Japan).In vivo drug penetration and biodistribution studies of transfersome and protransfersomeAnimals used in this study were HR-1/Hos hairless mice (male, 8-weeks-old, and 25–32 g) and purchased from Sankyo Labo Service Corp. (Tokyo, Japan). All animals were maintained and treated inconditions complying with the guiding principles for the care and use of laboratory animals as set up by the committee on animal researchof Hoshi University.The animals were divided into three groups, which consisted of 3-4 animals per group. A short glass tube with available diffusion area of 2.54 cm2 was adhered on the abdominal skin of the mice. Then, the drug preparations were applied non-occlusively at a dose equalto 37.2 mg cisplatin per kg for 3 and 12 hours. Cisplatin solution was prepared in physiological saline with a concentration of 1 mg/mL and used for the control group. During the study, all animals were kept on a heating mat (40 °C) and anesthetized by isoflurane inhalation. At the end of experiment (3 and 12 hours application), the mice were sacrificed by decapitation, and blood were collected in heparinized tubes. To obtain the plasma fraction, blood were centrifuged at 9,100 g for 10 minutes. Skin and organs (liver, kidney, spleen, lung, and heart) were removed carefully and weighed. Tissues and plasma were stored at -20 °C until platinum analysis using graphite furnace atomic absorption spectrophotometry (Z-8100 Polarized Zeeman GF-AAS, Hitachi, Japan).Platinum levels were determined by the GF-AAS method. The samples were prepared using wet acid digestion method in concentrated HNO3 and were then diluted with 0.1 N HCl. The GFAAS analysis program involved three steps: (1) a drying stage at 80– 100 °C for 40 seconds, (2) an ashing stage at 800 °C for 30 seconds, and (3) an atomization stage by heating at 3,000 °C for 7 seconds,then cooling. The absorbance was measured at 265.9 nm. The results were expressed as µg Pt/mL plasma and µg Pt/g tissue or organ.Skin irritation by transfersome and protransfersome applicationAfter the drug penetration study (12 hours), the excess drug on the skin was removed by washing twice with a mixture of 95% ethanol: physiological saline (1:1), followed by distilled water rinsing and wiping lightly with cotton. Afterward, skin irritation was evaluated by visual observation of the application site.In addition, skin histology was observed by administering the drug preparations onto the abdominal skin surface for 12 hours under anesthesia. After the skin had been cleaned of the drug preparations, the tissue was taken, embedded in an OCT compound (Tissue Tek®, Sakura Finetek Japan Co., Ltd., Tokyo, Japan), and processed by frozen sectioning at 16 µm. Hematoxylin and eosin staining was performed to observe the skin histology under an optical microscope.Statistical analysisAll data was produced in replicates and presented as the mean ± SD. To evaluate the significance of the difference, the data was analyzed by one-way ANOVA, followed by Turkey’s post-hoc Test using SPSS Software v.17.0 with p values
Microscopic evaluation of protransfersomeProtransfersome was observed microscopically by placing a small amount of sample on the object glass. A drop of physiological saline was added to the sample through the cavity side of the cover slip. The photomicrographs before, during, and after saline addition were taken at suitable magnifications under an optical microscope (NikonEclipse TS100-F, Nikon Inc., Tokyo, Japan).In vivo drug penetration and biodistribution studies of transfersome and protransfersomeAnimals used in this study were HR-1/Hos hairless mice (male, 8-weeks-old, and 25–32 g) and purchased from Sankyo Labo Service Corp. (Tokyo, Japan). All animals were maintained and treated inconditions complying with the guiding principles for the care and use of laboratory animals as set up by the committee on animal researchof Hoshi University.The animals were divided into three groups, which consisted of 3-4 animals per group. A short glass tube with available diffusion area of 2.54 cm2 was adhered on the abdominal skin of the mice. Then, the drug preparations were applied non-occlusively at a dose equalto 37.2 mg cisplatin per kg for 3 and 12 hours. Cisplatin solution was prepared in physiological saline with a concentration of 1 mg/mL and used for the control group. During the study, all animals were kept on a heating mat (40 °C) and anesthetized by isoflurane inhalation. At the end of experiment (3 and 12 hours application), the mice were sacrificed by decapitation, and blood were collected in heparinized tubes. To obtain the plasma fraction, blood were centrifuged at 9,100 g for 10 minutes. Skin and organs (liver, kidney, spleen, lung, and heart) were removed carefully and weighed. Tissues and plasma were stored at -20 °C until platinum analysis using graphite furnace atomic absorption spectrophotometry (Z-8100 Polarized Zeeman GF-AAS, Hitachi, Japan).Platinum levels were determined by the GF-AAS method. The samples were prepared using wet acid digestion method in concentrated HNO3 and were then diluted with 0.1 N HCl. The GFAAS analysis program involved three steps: (1) a drying stage at 80– 100 °C for 40 seconds, (2) an ashing stage at 800 °C for 30 seconds, and (3) an atomization stage by heating at 3,000 °C for 7 seconds,then cooling. The absorbance was measured at 265.9 nm. The results were expressed as µg Pt/mL plasma and µg Pt/g tissue or organ.Skin irritation by transfersome and protransfersome applicationAfter the drug penetration study (12 hours), the excess drug on the skin was removed by washing twice with a mixture of 95% ethanol: physiological saline (1:1), followed by distilled water rinsing and wiping lightly with cotton. Afterward, skin irritation was evaluated by visual observation of the application site.In addition, skin histology was observed by administering the drug preparations onto the abdominal skin surface for 12 hours under anesthesia. After the skin had been cleaned of the drug preparations, the tissue was taken, embedded in an OCT compound (Tissue Tek®, Sakura Finetek Japan Co., Ltd., Tokyo, Japan), and processed by frozen sectioning at 16 µm. Hematoxylin and eosin staining was performed to observe the skin histology under an optical microscope.Statistical analysisAll data was produced in replicates and presented as the mean ± SD. To evaluate the significance of the difference, the data was analyzed by one-way ANOVA, followed by Turkey’s post-hoc Test using SPSS Software v.17.0 with p values
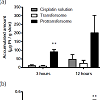
In vivo drug penetration and biodistribution studies of transfersome and protransfersome incorporating cisplatin Figure 2 shows platinum concentration in skin tissue and plasma at 3 and 12 hours after topical application of transfersome and protransfersome. The platinum concentration measured in the skin tissue of the protransfersome group was found to be significantly higher than that in the other treatment groups. In general, all drug preparations showed increased platinum levels in the skin tissue within a time that correlated with the increase of platinum concentration in the plasma. Three hours after topical drug administration of protransfersome, the drug could be rapidly detected in the plasma and increased up to 0.46 µg Pt/mL at 12 hours application; this was 30 and 40 times higher than the corresponding values in the group treated with cisplatin solution at 3 and 12 hours, respectively. On the other hand, transfersome increased the platinum levels 4-fold higher than the cisplatin solution group at 3 hours and exhibited nearly the same concentration at 12 hours (0.01 and 0.01 µg Pt/mL plasma, respectively).After topical drug administration, cisplatin entered the blood and distributed itself into the organs as shown in Figure 3. Protransfersome had the highest platinum levels in these organs compared to transfersome and cisplatin solution with increased values obtained at 12 hours after treatment. In addition, high drug levels were detected in the kidney compared to other organs.Skin irritation study of cisplatin-loaded transfersome and protransfersomeAfter the drug had been applied to the mice abdominal skin for 12 hours, skin irritation was observed visually (Figure 4). Localized erythema was observed in one of the four studied mice in the protransfersome group as visible red color skin with no papular response (Figure 4c). On the other hand, normal physiological skin appearances were observed in both cisplatin solution and transfersome groups (Figures 4a and 4b). In comparison with the normal skin tissue, no remarkable histological skin changes were observed in transfersome and protransfersome groups as shown in Figure 5. Based on these results, the change of abdominal skin color in the protransfersome group may not correlate with the cytotoxic effect of cisplatin.
Evaluation of Transfersome and Protransfersome for Percutaneous Delivery ofCisplatin in Hairless Mice
Andang Miatmoko1,Kumi Kawano1*, Yoshiyuki Hattori1, Yoshie Maitani1,2 and EtsuoYonemochi1
- 1nstitute of Medicinal Chemistry, Hoshi University, Ebara 2-4-41, Shinagawa, Tokyo 142-8501, Japan
- 2Keio University, Shibakoen 1-5-30, Minato, Tokyo 105-8512, Japan
*Address for Correspondence: Kumi Kawano, Institute of Medicinal Chemistry, Hoshi University, Ebara 2-4-41, Shinagawa, Tokyo 142-8501, Japan, Tel: +81-3-5498-5097; E-mail: kkumi@hoshi.ac.jpCitation:Miatmoko A, Kawano K, Hattori Y, Maitani Y, Yonemochi E. Evaluation of Transfersome and Protransfersome for Percutaneous Delivery of Cisplatin in Hairless Mice. J Pharmaceu Pharmacol. 2015;S(1): 7.Copyright © 2015 Kawano et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.Journal of Pharmaceutics & Pharmacology | ISSN: 2327-204X | Special Issue: 1Submission: 16 March, 2015 | Accepted: 13 April, 2015| Published: 17 April, 2015Editor:Dr. Vinayagam Kannan, Scientist Par Sterile Products, LLC, USA
Abstract
Cisplatin is a widely used antitumor drug, but its percutaneous absorption is limited. This work aimed to evaluate percutaneous absorption of cisplatin using deformable vesicular systems such as transfersomes and protransfersomes. Cisplatin-loaded transfersomes and protransfersomes were composed of phospholipids, surfactants, ethanol, and saline. They were prepared using the thin film method and coacervation technique, respectively. The transfersomes had vesicle sizes near 100 nm with zeta potential -1.2 mV and approximately 8% entrapment efficiency. The saline hydrated-vesicles of protransfersome showed large sizes and negatively charged vesicles (1081.3 nm and -9.4 mV, respectively) with a high drug loading capacity of up to 85%. Topical application of protransfersomes onto the abdominal skin of HR-1/Hos hairless mice resulted in better drug absorption than the cisplatin solution and transfersomes. In conclusion, these results suggested that protransfersome has the potential to deliver cisplatin via transdermal routes.Keywords: Cisplatin; Transdermal delivery; Protransfersome; Biodistribution study
Introduction
Cisplatin is a platinum complex alkylating-drug that has been widely used in monochemotherapy or combination therapy with other drugs for cancers, including lung, testicular, ovarian, bladder, head, neck, and esophageal cancers [1-3]. Its numerous benefits in clinical therapy is limited by severe side effects, including nephrotoxicity, distal neuropathy, ototoxicity, nausea, vomiting, and anorexia [4-7]. Therefore, it is still necessary to improve the efficacyof cisplatin and decrease its undesired effects by minimizing the free drug concentrations in blood to avoid high drug exposure to the organs.Transdermal administration routes are useful to avoid peak drug concentration by prolonging the drug absorption through the skin. It can also maintain the drug concentration in plasma, especially for drugs with short half-lives such as cisplatin [8-9]. Unfortunately, cisplatin has limited penetration and low drug diffusion through the epidermis [10].Transfersome has been developed as a drug carrier for transdermal drug delivery systems. It is characterized by deformable liposomes that are composed of mixtures of lipid with edge activators as the additional component. This deformable property is useful for carrying drugs across the skin via the intercellular route, which has smallerpores than the vesicle size itself [11]. Moreover, some researchers reported that these vesicles also exhibit penetration-enhancing effects by modifying intercellular lipid lamellae in the stratum corneum [12]. Therefore, transfersome may become an ideal carrier for transdermal delivery of cisplatin.On the other hand, the development of a transfersomal/liposomal formulation containing cisplatin has been hampered by its poor water solubility and low lipophilicity [13]. Furthermore, the transfersomelike vesicular system possesses poor stability relating to aggregation, sedimentation, phospholipids hydrolysis, and oxidation [14].Recently, protransfersome has been investigated as a more stable vesicular system, which is characterized by gel-like liquid crystalline provesicles with minimum water and alcohol. During application onto the skin surface, it will be hydrated and converted into transfersomal vesicles by absorbing water in situ [15]. In the presence of the transepidermal osmotic gradient, the movement of these vesicles will occur from the dry surface into the water-rich region of the deeper skin stratum. This driving force provides deeper penetration of vesicles in the skin, yielding better drug transports [16]. In addition, the lipid components of protransfersome could affect the physical nature of the stratum corneum that is fluidized, making it more permeable for further drug penetration [17].In this study, to enhance transdermal absorption of cisplatin, cisplatin-loaded transfersome and protransfersome were prepared. The blood concentration and biodistribution were evaluated in hairless mice after abdominal application of the formulations.Protransfersome enhanced the absorption of cisplatin and showed potential as a drug carrier via transdermal routes.Materials and Methods
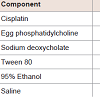
MaterialsCisplatin and sodium deoxycholate were purchased from Wako Pure Chemical Industries Co., Ltd. (Tokyo, Japan). Egg phosphatidylcholine (EPC, Coatsome NC-50) and Tween 80 were obtained from NOF Corporation (Tokyo, Japan). For graphite furnace atomic absorption spectrophotometry (GF-AAS) measurements,nitric acid (1.38; analytical grade) was used as the digestive acid solution and hydrochloric acid (AAS analytical grade) was used as the sample solvent. All other reagents and solvents used in this study were of analytical grade. Milli-Q water was used for all experiments.Preparation of transfersome and protransfersome loading cisplatinTransfersome was prepared by the dry thin film method [18] with compositions shown in Table 1. A dry thin film of EPC was hydrated with physiological saline containing cisplatin and Tween 80. The mixture was then extruded through polycarbonate membranes at 50 °C using an Avanti Polar Lipid® extruder. The final formulation consisted of EPC (4.25%) and Tween 80 (0.75%) with a total drug concentration of 0.1% w/wOn the other hand, protransfersome was prepared in a composition as shown in Table 1 using a previously reported method [15] with some modifications. The cisplatin solution in physiological saline containing sodium deoxycholate was added to the EPC solution in 95% ethanol, and then homogeneously mixed and vortexed at 65– 70 °C. The final composition of protransfersome was EPC, sodium deoxycholate, 95% ethanol, and physiological saline (1.7:0.3:1:2 weight ratio) with 0.34% w/w drug content.For further studies, transfersome and protransfersome were used without any vesicles purificationDetermination of particle size, ζ-potential, and entrapment efficiency of transfersome and protransfersomeThe average particle size and ζ-potential of the formulations were determined using a laser light scattering instrument (ELSZ-2, Otsuka Electronics, Co. Ltd., Japan) after dilution with an appropriate volume of Milli-Q water.For vesicle size measurement of protransfersome, which is a gellike preparation, approximately 50 mg of protransfersome gel were hydrated with 5 mL of physiological saline using three different methods [19], including: (a) without shaking: physiological saline was added through the tube walls carefully and kept aside for 5 min without any agitation, (b) by shaking: manually shaken 300 times (about 5 minutes), and (c) by sonication for 1 minute.Determination of the entrapment efficiency of transfersome was performed by separation of the vesicles incorporating cisplatin using a column packed with Sephadex G-50. For protransfersome, free and entrapped cisplatin were separated using an Amicon Ultra® centrifugal filter unit (MWCO 100,000, Millipore®) at 5,000 g for 30 minutes after hydration of the protransfersome gel with physiologicalsaline. Cisplatin concentration of each fraction was analyzed by highperformance liquid chromatography (HPLC) (Shimadzu, Japan) after solubilization with 67% n-propanol using an anion exchange column Inertsil AX® (250 mm × 4.6 mm, GL Sciences, Japan) as the stationary phase at room temperature. The mobile phase was composed of ethyl acetate: methanol: Milli-Q water: N,N-dimethylformamide (8:40:10:20, v/v) with a flow rate of 1 mL/min. The cisplatin peak was analyzed with a UV detector at 306 nm. The entrapment efficiency was expressed as follows: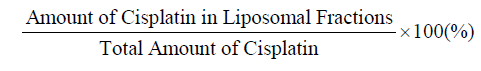 Microscopic evaluation of protransfersomeProtransfersome was observed microscopically by placing a small amount of sample on the object glass. A drop of physiological saline was added to the sample through the cavity side of the cover slip. The photomicrographs before, during, and after saline addition were taken at suitable magnifications under an optical microscope (NikonEclipse TS100-F, Nikon Inc., Tokyo, Japan).In vivo drug penetration and biodistribution studies of transfersome and protransfersomeAnimals used in this study were HR-1/Hos hairless mice (male, 8-weeks-old, and 25–32 g) and purchased from Sankyo Labo Service Corp. (Tokyo, Japan). All animals were maintained and treated inconditions complying with the guiding principles for the care and use of laboratory animals as set up by the committee on animal researchof Hoshi University.The animals were divided into three groups, which consisted of 3-4 animals per group. A short glass tube with available diffusion area of 2.54 cm2 was adhered on the abdominal skin of the mice. Then, the drug preparations were applied non-occlusively at a dose equalto 37.2 mg cisplatin per kg for 3 and 12 hours. Cisplatin solution was prepared in physiological saline with a concentration of 1 mg/mL and used for the control group. During the study, all animals were kept on a heating mat (40 °C) and anesthetized by isoflurane inhalation. At the end of experiment (3 and 12 hours application), the mice were sacrificed by decapitation, and blood were collected in heparinized tubes. To obtain the plasma fraction, blood were centrifuged at 9,100 g for 10 minutes. Skin and organs (liver, kidney, spleen, lung, and heart) were removed carefully and weighed. Tissues and plasma were stored at -20 °C until platinum analysis using graphite furnace atomic absorption spectrophotometry (Z-8100 Polarized Zeeman GF-AAS, Hitachi, Japan).Platinum levels were determined by the GF-AAS method. The samples were prepared using wet acid digestion method in concentrated HNO3 and were then diluted with 0.1 N HCl. The GFAAS analysis program involved three steps: (1) a drying stage at 80– 100 °C for 40 seconds, (2) an ashing stage at 800 °C for 30 seconds, and (3) an atomization stage by heating at 3,000 °C for 7 seconds,then cooling. The absorbance was measured at 265.9 nm. The results were expressed as µg Pt/mL plasma and µg Pt/g tissue or organ.Skin irritation by transfersome and protransfersome applicationAfter the drug penetration study (12 hours), the excess drug on the skin was removed by washing twice with a mixture of 95% ethanol: physiological saline (1:1), followed by distilled water rinsing and wiping lightly with cotton. Afterward, skin irritation was evaluated by visual observation of the application site.In addition, skin histology was observed by administering the drug preparations onto the abdominal skin surface for 12 hours under anesthesia. After the skin had been cleaned of the drug preparations, the tissue was taken, embedded in an OCT compound (Tissue Tek®, Sakura Finetek Japan Co., Ltd., Tokyo, Japan), and processed by frozen sectioning at 16 µm. Hematoxylin and eosin staining was performed to observe the skin histology under an optical microscope.Statistical analysisAll data was produced in replicates and presented as the mean ± SD. To evaluate the significance of the difference, the data was analyzed by one-way ANOVA, followed by Turkey’s post-hoc Test using SPSS Software v.17.0 with p values
Microscopic evaluation of protransfersomeProtransfersome was observed microscopically by placing a small amount of sample on the object glass. A drop of physiological saline was added to the sample through the cavity side of the cover slip. The photomicrographs before, during, and after saline addition were taken at suitable magnifications under an optical microscope (NikonEclipse TS100-F, Nikon Inc., Tokyo, Japan).In vivo drug penetration and biodistribution studies of transfersome and protransfersomeAnimals used in this study were HR-1/Hos hairless mice (male, 8-weeks-old, and 25–32 g) and purchased from Sankyo Labo Service Corp. (Tokyo, Japan). All animals were maintained and treated inconditions complying with the guiding principles for the care and use of laboratory animals as set up by the committee on animal researchof Hoshi University.The animals were divided into three groups, which consisted of 3-4 animals per group. A short glass tube with available diffusion area of 2.54 cm2 was adhered on the abdominal skin of the mice. Then, the drug preparations were applied non-occlusively at a dose equalto 37.2 mg cisplatin per kg for 3 and 12 hours. Cisplatin solution was prepared in physiological saline with a concentration of 1 mg/mL and used for the control group. During the study, all animals were kept on a heating mat (40 °C) and anesthetized by isoflurane inhalation. At the end of experiment (3 and 12 hours application), the mice were sacrificed by decapitation, and blood were collected in heparinized tubes. To obtain the plasma fraction, blood were centrifuged at 9,100 g for 10 minutes. Skin and organs (liver, kidney, spleen, lung, and heart) were removed carefully and weighed. Tissues and plasma were stored at -20 °C until platinum analysis using graphite furnace atomic absorption spectrophotometry (Z-8100 Polarized Zeeman GF-AAS, Hitachi, Japan).Platinum levels were determined by the GF-AAS method. The samples were prepared using wet acid digestion method in concentrated HNO3 and were then diluted with 0.1 N HCl. The GFAAS analysis program involved three steps: (1) a drying stage at 80– 100 °C for 40 seconds, (2) an ashing stage at 800 °C for 30 seconds, and (3) an atomization stage by heating at 3,000 °C for 7 seconds,then cooling. The absorbance was measured at 265.9 nm. The results were expressed as µg Pt/mL plasma and µg Pt/g tissue or organ.Skin irritation by transfersome and protransfersome applicationAfter the drug penetration study (12 hours), the excess drug on the skin was removed by washing twice with a mixture of 95% ethanol: physiological saline (1:1), followed by distilled water rinsing and wiping lightly with cotton. Afterward, skin irritation was evaluated by visual observation of the application site.In addition, skin histology was observed by administering the drug preparations onto the abdominal skin surface for 12 hours under anesthesia. After the skin had been cleaned of the drug preparations, the tissue was taken, embedded in an OCT compound (Tissue Tek®, Sakura Finetek Japan Co., Ltd., Tokyo, Japan), and processed by frozen sectioning at 16 µm. Hematoxylin and eosin staining was performed to observe the skin histology under an optical microscope.Statistical analysisAll data was produced in replicates and presented as the mean ± SD. To evaluate the significance of the difference, the data was analyzed by one-way ANOVA, followed by Turkey’s post-hoc Test using SPSS Software v.17.0 with p valuesResults
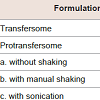
Characterizations of transfersome and protransfersomeCharacterizations of transfersome and protransfersome are shown in Table 2. The vesicle size of transfersome was nearly 100 nm, while the vesicles of protransfersome that have a gel-like consistency were determined using three methods [19]. The self-formation of vesicles was obtained during water hydration (without shaking) yielding relatively large vesicles. Furthermore, the involvement of energy input in the system decreased their sizes as observed in the shaking method, and the smallest vesicles were obtained in the sonication method. ζ-potential measurements showed a relatively neutral charge of transfersome, while protransfersome exhibited negatively charged vesicles. With regard to drug loading, transfersome had low drug entrapment efficiency, but almost 85% of the drug could be loaded into the protransfersome prepared by the sonication method. Both cisplatin-loaded transfersome and protransfersome showed no precipitation and no color changes at least two weeks storage at room temperature (data not shown).In this study, protransfersome was produced successfully using a simple method based on coacervation-phase separation approach as published [20,21]. As shown in Figure 1a, lamellar structures of protransfersome were observed by light microscopy. Further, on-site hydration with saline resulted in changes of the lamellar structures into long, oval-shaped vesicles (figure1b). Moreover, it led to their transformation into apparent transfersome-like vesicles (figure1c). Transfersome was observed as smaller particles (figure1d), which correlated well with its particle size as determined by light scattering.Figure :1 :(a) Photomicrographs of lamellar structures of protransfersome gel, (b) long, oval-shaped vesicles of protransfersome during on-site hydration with saline,(c) transfersome-like vesicles produced after dispersion of protransfersome in saline by manual shaking, and (d) transfersome on a scale bar of 40 µm. All imageswere observed under a light microscope. The black arrows indicate vesicles. (n=3).
Figure :2 :(a) : (a) Platinum concentrations found in abdominal skin tissue and (b) blood plasma of HR-1/Hos hairless mice at 3 and 12 hours after topical drugadministration. All measurements were performed in 3–4 replicates. The pvalue is lower than 0.01 (**) versus the group treated with cisplatin solution.
Discussion
After the drug had been applied to the mice abdominal skin for 12 hours, skin irritation was observed visually (Figure 4). Localized erythema was observed in one of the four studied mice in the protransfersome group as visible red color skin with no papular response (Figure 4c). On the other hand, normal physiological skin appearances were observed in both cisplatin solution and transfersome groups (Figures 4a and 4b). In comparison with the normal skin tissue, no remarkable histological skin changes were observed in transfersome and protransfersome groups as shown in Figure 5. Based on these results, the change of abdominal skin color in the protransfersome group may not correlate with the cytotoxic effect of cisplatin.Figure :4 Visual appearances of the abdominal skin of HR-1/Hos hairless mice observed 12 hours after administration of (a) cisplatin solution, (b) transfersome, and (c) protransfersome. A mild erythema appeared as a visible red color in one of the four mice treated with protransfersome (arrowhead), while no skin irritationfeatures were found in transfersome or cisplatin solution groups. Scale bar = 5 mm.
To increase systemic absorption of cisplatin through the skin, cisplatin-loaded transfersome and protransfersome were investigated. Previously, it had been reported that the protransfersome formulations provide local delivery of cisplatin [20,21]. Corresponding to these reports, protransfersome could provide high drug loading capacity, drug permeation, and drug deposition in the skin. In this study, protransfersome was prepared with ethanol, instead of isopropanol,in lesser volume and it had higher total drug loading than reported previously [20,21]. Furthermore, our study provided drug penetration data which defines that our protransfersome formulation resulted in better cisplatin penetration into the plasma than transfersome and cisplatin solutions, as shown in Figure 2. These results suggested that protransfersome has the potential to be a carrier for transdermal delivery of cisplatin.Although it has been known that cisplatin has low water solubility and poor association with lipids [3], the rapid phase separation that occurred during protransfersome preparation probably offers sudden entrapment of cisplatin between stacked phospholipids in the mesophasic region. The higher amount of entrapped drug will enable vesicles to carry more drug through the skin layers into the systemic circulation [16]. In addition, the ethanol content of protransfersome might play important roles in this improvement. It has been reported that ethanol facilitates higher drug absorption from topical applications [22]. It disturbs the organization of stratum corneum and increases the fluidity of these lipids, generating permeable barriers for further drug penetration [23].On the other hand, transfersome could not improve the transdermal absorption of cisplatin. Unfavorable properties of cisplatin, such as poor water solubility and low lipophilicity, might cause low entrapment efficiency of cisplatin in transfersome (8%) resulting in small quantities of drug to be carried into circulation. It has been reported that deformable property of transfersome could carry many kinds of drugs via transdermal route [11-12/a>]. In preliminary study for transfersome formulations, cisplatin-loaded transfersome used in this study was the most stable and the highest entrapment efficiency (data not shown). For cisplatin delivery, the ability of transfersome might be exerted by further improvement of entrapment efficiency of cisplatin in transfersome.Besides the differences in drug loading, the surfactants employed in protransfersome and transfersome also have effects on drug penetration. The vesicles of protransfersome that were prepared with sodium deoxycholate had negative charges, while the use of Tween 80 in transfersome resulted in relatively neutrally charged vesicles. Skin layer membranes have rather negative charges [24] and show interactions with some charged substances [18,25]. Negatively charged protransfersome vesicles may be repulsed and not strongly interact with the skin membranes resulting in fast movement and rapid drug release compared to transfersome, which has less repulsive interaction. Therefore, higher drug absorption could be obtained by protransfersome than transfersome as shown in this study.After 12 hours of the protransfersome penetration study, we observed localized erythema in one of the four mice suggesting the possibility of side effects with topical administration of protransfersome. The ethanol contained in the protransfersome possibly caused the irritation of the skin owing to its direct pharmacological action on the cutaneous microvasculature [22]. Since the skin treated with transfersome and cisplatin solution showed normal physiological appearances and no remarkable histological changes were observed, it should have no correlation to the cytotoxic effect of the drug. When cisplatin is exposed to the low chloride aqueous environment, it forms reactive species [23] that is highly reactive to nucleophilic sites and lead to cytotoxic effects. However, this seems unlikely considering that the skin was in physiological condition and contained high amounts of electrolytes [27].The protransfersome group showed the highest platinum levels in the plasma. This result indicated that protransfersome could deliver cisplatin to systemic circulation via the transdermal route. The higher platinum levels in liver, spleen, lung and heart largely reflected the plasma drug concentrations, which the drug was further distributed to organs. It has been known that cisplatin has high binding affinityto protein in tissues and additionally spleen and liver are responsible for cisplatin metabolism [28]. The high platinum concentration found in the kidney indicated that cisplatin was mainly circulated in the blood in native form, since cisplatin is rapidly excreted through glomerular filtration and tubular secretion [29,30]. The plasma concentration obtained by protransfersome administration was very low compared to the plasma concentration after intravenous injection of cisplatin [30], indicating that protransfersome might decrease undesired side effect of cisplatin. However, this result also indicated that protransfersome cannot deliver enough drug to tumor cells systemically. Recently, the chronic, frequent, and equally spaced administration of generally low doses of various chemotherapeutic drugs without extended rest periods, i.e. metronomic chemotherapy, has been applied as a novel approach to reduce the severe side effects of chemotherapy by circumventing high drug exposure to organs and inhibiting tumor growth through an anti-angiogenic action [32]. Cisplatin-loaded protransfersomes could be utilized as metronomic chemotherapy, although increasing the stability of the vesicles in the plasma by using polymers or other metal-complex systems would be needed to reduce the native form of the drug after percutaneous absorption. Further development of the cisplatin-protransfersomal system is still necessary to achieve antitumor effect in vivo.
Conclusions
In summary, we have studied the transdermal delivery of cisplatin using ultradeformable liposomes (transfersome and protransfersome). Protransfersome had better drug penetration than transfersome and cisplatin solutions. As a drug carrier, protransfersome was able to deliver cisplatin into the systemic blood circulation via the transdermal route. However, further development of the cisplatin-protransfersomal system is still necessary to obtain better transdermal delivery of cisplatin for cancer therapy.References
- Finley RS, McCune JS (2005) Lung Cancer, Pharmacotherapy: A Pathophysiologic approach, sixth ed., (DiPiro JT Ed.) McGraw-Hill, New York pp: 2365-2381.
- Brabec V, Kasparkova J (2005) 78Pt Platinum-based drugs, in metallotherapeutic drugs and metal-based diagnostic agents: the use of metals in medicine (eds M. Gielen and E. R.T. Tiekink), John Wiley & Sons, Ltd, Chichester, UK pp: 489-506.
- Sweetman SC, Blake PS, Brayfield A, McGlashan JM, Neathercoat GC, et al. (2009) Cisplatin. Martindale the complete drug reference, 36thed., (Sweetman SC Ed.) Pharmaceutical Press, London. Pp: 698-700.
- Miller RP, Tadagavadi RK, Ramesh G, Reeves WB (2010) Mechanisms of cisplatin nephrotoxicity. Toxins 2: 2490-2518.
- Quasthoff S, Hartung HP (2002) Chemotherapy-induced peripheral neuropathy. J Neurol 249: 9-17.
- Kurihara N, Kubota T, Hoshiya Y, Otani Y, Ando N, et al. (1996) Pharmacokinetics of cis-diamminedichloroplatinum (II) given as low-dose and high-dose infusions. J Surg Oncol 62: 135-138.
- Simon T, Hero B, Dupuis W, Selle B, Berthold F (2002) The incidence of hearing impairment after successful treatment of neuroblastoma. Klin Padiatr 214: 149-152.
- Cevc G, Vierl U (2010) Nanotechnology and the transdermal route: A state of the art review and critical appraisal. J Control Release 141: 277-299.
- Roberts MS, Cross SE, Pellett MA (2002) Skin Transport. Dermatological and transdermal formulations. (Walters KA Ed.) Marcel Dekker, New York. pp. 102-209.
- Simonetti LD, Gelfuso GM, Barbosa JC, Lopez RF (2009) Assessment of the percutaneous penetration of cisplatin: the effect of monoolein and the drug skin penetration pathway. Eur J Pharm Biopharm 73: 90-94.
- Cevc G (2004) Lipid vesicles and other colloids as drug carriers on the skin. Adv Drug Deliv Rev 56: 675-711.
- Elsayed MM, Abdallah OY, Naggar VF, Khalafallah NM (2007) Lipid vesicles for skin delivery of drugs: reviewing three decades of research. Int J Pharm 332: 1-16.
- Liu WP, Ye QS, Yu Y, Chen XZ, Hou SQ, et al. (2008) Novel lipophilic platinum (II) compounds of salicylate derivatives. Platinum Metals Rev 52: 163-171.
- Grit M, Zuidam NJ, Underberg WJM, Crommelin DJA (1993) Hydrolysis of partially saturated egg phosphatidylcholine in aqueous liposome dispersions and the effect of cholesterol incorporation on hydrolysis kinetics. J Pharm Pharmacol 45: 490-495.
- Jain S, Sapre R, Tiwary AK, Jain NK (2005) Proultraflexible lipid vesicles for effective transdermal delivery of levonorgestrel: development, characterization, and performance evaluation, AAPS PharmSciTech 6: E513-522.
- Honeywell-Nguyen PL, WouterGroenink HW, de Graaff AM, Bouwstra JA (2003) The in vivo transport of elastic vesicles into human skin: effects of occlusion, volume and duration of application. J Control Release 90: 243-255.
- Jukanti R, Sheela S, Bandari S, Veerareddy PR (2011) Enhanced bioavailability of exemestane via proliposomes based transdermal delivery. J Pharm Sci 100: 3208-3222.
- Dragicevic-Curic N, Grafe S, Gitter B, Winter S, Fahr A (2010) Surface charged temoporfin-loaded flexible vesicles: in vitro skin penetration studies and stability. Int J Pharm 384: 100-108.
- Vora B, Khopade AJ, Jain NK (1998) Proniosome based transdermal delivery of levonorgestrel for effective contraception. J Control Release 54: 149-165.
- Gupta V, Agrawal RC, Trivedi P (2011) Reduction in cisplatin genotoxicity (micronucleus formation) in non target cells of mice by protransfersome gel formulation used for management of cutaneous squamous cell carcinoma. Acta Pharm 61: 63-71.
- Gupta V, Karthikeyan C, Trivedi P (2012) Localized delivery of cisplatin for the effective management of squamous cell carcinoma from protransfersome formulation. Arch Pharm Res 35: 851-859.
- Lachenmeier DW (2008) Safety evaluation of topical applications of ethanol on the skin and inside the oral cavity. J Occup Med Toxicol 3: 26.
- Touitou E, Dayan N, Bergelson L, Godin B, Eliaz M (2000) Ethosomes - novel vesicular carriers for enhanced delivery: characterization and skin penetration properties. J Control Release 65: 403-418.
- Brunette RR, Ongpittanakul B (1987) Characterization of the permselective properties of excised human skin through iontophoresis. J Pharm Sci 76: 765-773.
- Chen Y, Zhou L, Zhang Z, Liu X, Wu Q (2012) Formulation, characterization and evaluation of in vitro skin permeation and in vivo pharmacodynamics of surface-charged tripterine-loaded nanostructured lipid carriers. Int J Nanomedicine 7: 3023-3032.
- De Waal WA, Maessen FJ, Kraak JC (1990) Analytical methodologies for the quantitation of platinum anti-cancer drugs and related compounds in biological media. J Pharm Biomed Anal 8: 1-30.
- Volden G, Thorsrud AK, Bjornson I, Jellum E (1980) Biochemical composition of suction blister fluid determined by high resolution multicomponent analysis (capillary gas chromatography-mass spectrophotometry and two-dimensional electrophoresis). J Invest Dermatol 75: 421-424.
- Junior AD, Mota LG, Nunan EA, Wainstein AJ, Wainstein AP, et al. (2007) Tissue distribution evaluation of stealth pH-sensitive liposomal cisplatin versus free cisplatin in Ehrlich tumor-bearing mice. Life Sci 80: 659-664.
- Yao X, Panichsipal K, Kurtzman N, Nugent K (2007) Cisplatin nephrotoxicity: A Review. Am J Med Sci 334: 115-124.
- DeConti R, Toftness BR, Lange RC, Creasey WA (1973) Clinical and pharmacological studies with cis-diamminedichloroplatinum (II). Cancer Res 33: 1310-1315.
- 31. Nishiyama N, Okazaki S, Cabral H, Miyamoto M, Kato Y, et al. (2003) Novel cisplatin-incorporated polymeric micelles can eradicate solid tumors in mice. Cancer Res 63: 8977-8983.
- Shen FZ, Wang J, Liang J, Mu K, Hou JY et al. (2010) Low-dose metronomic chemotherapy with cisplatin: can it suppress angiogenesis in H22 hepatocarcinoma cells? Int J Exp Pathol 91: 10-16.