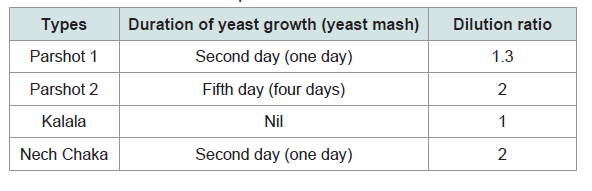Journal of Food Processing & Beverages
Download PDF
Research Article
Nutritional Value of the Alcoholic Beverage “Parshot” as a Staple and Total Nutrition Food in Dirashe Special Worenda, Sounthern Ethiopia
Yui Sunano*
- Graduate School of Bioagricultural Sciences and School of Agricultural Sciences, Nagoya University, Japan
*Address for Correspondence: Yui Sunano, Graduate School of Bioagricultural Sciences and School of Agricultural Sciences, Nagoya University, Nagoya City 457-4 Motoyama, Kamigamo, Kita-ku, Kyoto, 603-8047, Japan, Tel: 81-52-789-4057; E-mail: yui.sunano@gmail.com
Citation: Sunano Y. Nutritional Value of the Alcoholic Beverage “Parshot” as a Staple and Total Nutrition Food in Dirashe Special Worenda, Sounthern Ethiopia. J Food Processing & Beverages. 2017;5(1): 9.
Copyright: © 2017 Sunano Y. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Food Processing & Beverages | ISSN: 2332-4104 | Volume: 5, Issue: 1
Submission: 30 March, 2017| Accepted: 22 May, 2017 | Published: 9 June, 2017
Abstract
The global incidence of malnutrition has seen a decreasing trend. However, it continues to remain a serious concern in Africa, where people are dependent on rain-fed agriculture and suffer from unstable climate that governs the crop yield. To prevent malnutrition, it is a prerequisite to consume proteins and amino acids, major components of the human body, in addition to grains as an energy source. Nevertheless, the Dirashe people in the semiarid region in Africa manage to maintain health, despite the fact that they are almost entirely dependent on local liquor made of sorghum (Sorghum bicolor) and maize (Zea mays) for their nutritional intake.
I conducted a participant observation study and an investigation of the content of the meals and amount and frequency of intake; I also obtained samples of sorghum, maize, and alcoholic beverages. The analysis showed that the amount of amino acids in the alcoholic beverage was higher than that in the grains. In addition, the results of the observation study showed that the Dirashe people drink a lot of alcoholic beverages and increase the nutritional value of sorghum and maize by using them to make alcoholic beverages. Most people in Africa improve the nutrient quality of their food intake by cultivating beans; however, the Dirashe people obtain nutrients efficiently by cooking and consuming a limited number of crops.
Keywords
Food culture; Amino acid score; Fermented food; Nutritional value; Africa
Introduction
The total world population is estimated to increase from the current 6 billion to 10 billion by 2050 [1] and a rapid increase has been observed, particularly in Africa, Asia, and Latin America. In these regions, people suffer from problems with regards to “food production, access to food, water scarcity, and nutritional disorders/ malnutrition” and over 800 million people living in developing countries have been reported to be undernourished, owing to inadequate food availability [2]. Globally, malnutrition is particularly severe among children under five years old and 30% of these children are underweight. If born with low birth weight, these children are more likely to suffer from child malnutrition and premature death [2,3]. According to the estimate by Food and Agriculture Organization (FAO) in 1996, the malnourished population would decrease to 680 million by 2010, mainly in Asia, but it would continue to increase in the Sub-Sahara Africa [4,5]. The status quo in 2016 attests to this estimate, and the food supply remains to be stabilized in Africa.
Human beings are required to be consistently supplemented with nutrition to sustain life and maintain a healthy metabolism. For this, it is necessary to consume food with nutrients such as carbohydrate, protein, fat, vitamins, and dietary fiber on a constant basis. In other words, lack of these nutrients causes malnutrition. Among these nutrients, the most important are proteins that compose muscles and internal organs, and enzymes and hormones, which catalyse chemical reactions that elicit body functions and 20 types of amino acids, which compose proteins. The human body continues to synthesize and degrade these nutrients, thus requiring constant supplementation. Among these 20 types of amino acids, nine types of indispensable (termed as essential) amino acids, namely isoleucine, leucine, lysine, phenylalanine, tryptophan, threonine, methionine, valine, and histidine cannot be synthesized in the human body [6,7]. Therefore, whether people consume protein having essential amino acids in sufficient quantities is an important criterion in evaluating dietary habits.
Generally, human beings acquire calories necessary for their daily activities by consuming large quantities of cereal and root crops as a staple food [8,9]. However, the principal component of cereal and root crops is carbohydrate; thus, the protein content in such foods is not adequate and the amino acids that compose its protein are of poor quality [10,11]. They are particularly in low in lysine content, one of the essential amino acids [11]. Lysine, therefore, functions as the first limiting amino acid and restricts efficient intake of other essential amino acids as well. Therefore, along with cereal and root crops, human beings consume meat, fish, dairy products such as milk, yogurt, and butter, and pulses in order to supplement protein of good quality and essential amino acids [9]. French, Italian, Chinese, Japanese, and Indian cuisines, which are known worldwide, consist of cereals or potatoes such as bread, pasta, rice, noodles, and naan (flatbread), and animal protein such as meat, fish, milk, yogurt, and butter or plant protein such as beans. In this regard, most of the food cultures around the world efficiently combine various foodstuffs to take in nutrients required for subsistence.
This also applies to food culture in Africa, where many suffer from malnutrition. People in Kenya and Tanzania in Eastern Africa eat ugari, local porridge of degummed texture made of grain flours (sorghum (Sorghum bicolor)), pearl millet (Pennisetum glaucum), finger millet (Eleusine coracana), or maize (Zea mays) and boiled water, together with a simmered bean dish called maharage or tomato soup with vegetables [8]. In West Africa, people eat too, porridge-like dish made of peal millet and sorghum flour, which is cooked in the same manner with ugari, or couscous as a stable food [8]. To make couscous, first, pearl millet and sorghum flour are mixed with a small amount of water and rolled using hands in a halved gourd to make small pellets; next, these small pellets are shifted and steamed in a food steamer. These stable foods are always accompanied by a sauce made of ladyfinger (Abelmoschus esculentus) or a soup of vegetables or beans [8]. These traditional meals of African people are excellent in maintaining nutritional balance.
However, the unstable climate in most areas of Sub-Saharan Africa results in an unstable crop yield as well [12], thereby leading to an unbalanced nutrition intake by local inhabitants. About 70% of the African continent is located either in the arid region with an average annual precipitation rate of 100-500 mm or in the semiarid region with a rate of 500-1,000 mm [13,14]. These arid and semi-arid regions not only suffer from scarce rainfall but also spatial and temporal differences in rainfall [15]. The year-to-year differences in rainfall are large and, in some regions, the precipitation rate varies in units of several decades. African people rely on rain-fed agriculture and their food security, therefore, can be largely impacted by the regional climate and weather [16,17]. These arid and semiarid lands regularly suffer from a shortage of rainfall and its consequential and serious draught [18]. As a result, people suffer from malnutrition, despite the aforementioned excellent food culture.
On the other hand, the Dirashe people, living in the semiarid Dirashe region in southern Ethiopia, drink fermented cereal liquor called parshot as their staple food. This liquor is prepared by mixing and alcohol-fermenting sorghum and maize with small quantities of dried and powdered leaves of moringa (Moringa stenopetala)13 or Ethiopian kale (Brassica carinata). These people rarely consume any other kind of food [19]; they rarely eat meat, dairy products, and beans rich in protein and essential amino acids in their daily life, and only get to eat beef or goat meat several times a year on occasions such as wedding ceremonies or new year celebrations. They normally satisfy their appetite by drinking large quantities of parshot; however, they drink a white-colored alcoholic beverage called nech chaka produced by the alcohol-fermentation of only sorghum and maize, or kalala, which is low in alcohol concentration, in severe dry seasons when plant leaves used for parshot are scarce [19]. They also consume solid food such as cereal dumplings called hawarata made of sorghum and maize and unleavened bread called rakot, but in very small quantities. Hence, a case of malnutrition leading low immunity and poor concentration is barely observed in the region.
Globally, it is extremely rare to observe people who hardly take in protein sources and, furthermore, take in almost all of the nutrients from cereal grains, like the Dirashe people do. Moreover, their consumption of alcoholic beverages as a staple food makes their food culture more unique. It is of great importance to examine their nutritional status, with a view to improving the general nutritional status of human beings and discussing the history of food culture. This article investigates why the Dirashe people are not undernourished, despite the fact that they are almost entirely dependent on alcoholic beverages such as parshot for their nutrient intake, focusing on their nutritional value and the amount of intake. The results could be subsequently analysed to understand how such a unique culture of drinking was developed.
Methods
Research field and period
A total of 10 months (January to March and June to August 2009, January to February 2011, December 2011 to January 2012) was spent in a village called ‘W’. Participant observation and interviews regarding the brewing process of alcoholic beverages and food content were conducted during the stay.
Samples
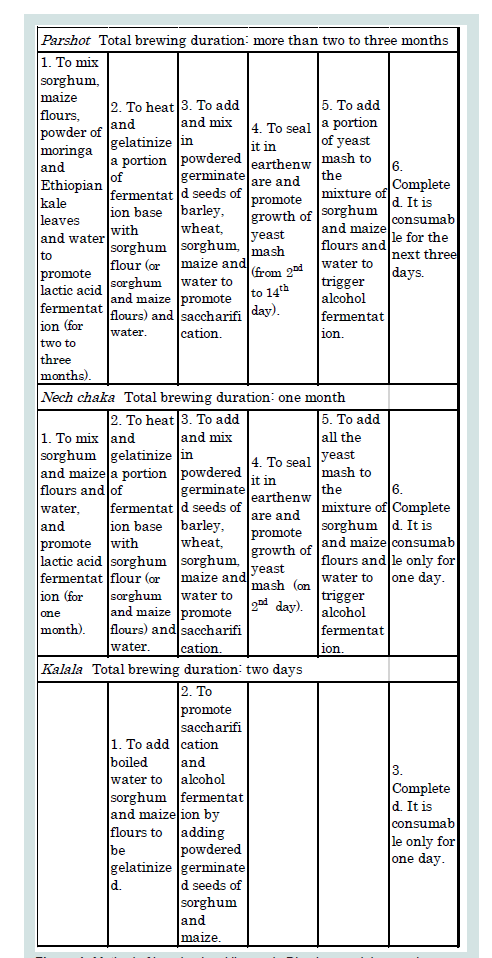
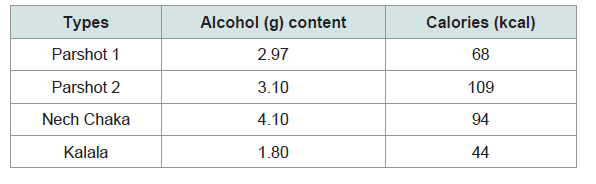
For measuring nutritional value, sorghum, maize, parshot, kalala, and nech chaka were collected as samples and brought back to Japan. Brewing parshot and nech chaka requires a certain time for the growth of yeast mash; for nech chaka, yeast mash undergoes alcohol fermentation at the latest on the second day, but for parshot, yeast mash is preserved for two days to two weeks before the process of alcohol fermentation (Figure 1). Two types of parshot sample were collected out of the types commonly available at the time in ‘W’ village. Both were one-day-old after the brewing process was completed, but with different durations of preservation of yeast mash prior to alcohol fermentation; one was preserved for two days and the other for five days. The first sample was then diluted 1.3 times with water, and the second sample diluted double with water; these samples are hereinafter referred to as parshot 1 and parshot 2, respectively. Well water was used for preparing and diluting the alcoholic beverages in this area. People drank kalala as it was without diluting, but nech chaka was doubly diluted with water. Therefore, undiluted kalala and nech chaka, which was diluted double with water, were collected as samples and analysed. The details of sample preparation are shown in Table 1. These samples were put in a heatresistant container and double-boiled at 60 degrees for 30 minutes to stop further fermentation and then frozen. In order to evaluate the nutritional value of these three samples, calories (kcal), protein (g), essential amino acid (mg), and Alcohol (g) contents per 100 g were measured. Additionally, calories (kcal), carbohydrate (g), protein (g), and essential amino acid (mg) per 100 g of sorghum and maize were also measured in order to compare the amino acid score of these three samples.
Analysis
All these analyses were outsourced to Japan Food Research Laboratories (JFRL). At JFRL, calories were calculated with the quantity of carbohydrate content, protein content, fat content, water content, ash content, dietary fiber content and alcohol content. The methods used for analysing the contents are as follows: ash content by Direct ashing method, vehicle dry weight and moisture content estimation by Loss on drying under reduced pressure method, protein content by Kjeldahl method, fat content by Soxhlet extraction method, alcohol content determination by Dichromate oxidation method, dietary fiber content by Enzymatic-gravimetric method, carbohydrate content by calculation and essential amino acids by Amino acid automatic analysis. The following are details of analysis.
Ash content determination: Direct ashing method: The sample(1.0-5.5 g) was collected and subjected to preliminary ashing at 550 °C for five hours, followed by cooling the sample in a silica gel desiccator and weighing it. The ash content was calculated using the formula described below.
Ash content (g/100 g) = weight (g) / amount of collected sample (g) × 100
Vehicle dry weight and moisture content estimation: Loss on drying under reduced pressure method: The sample (1.6-4.8 g) was collected and weighed in a glass weighing dish with silica sand, dried sufficiently at 55 °C, and subjected to a final round of drying for five hours at 70 °C. Next, it was cooled in the silica gel desiccator and the subsequent weight was estimated. Moisture content in the sample was calculated from the titer as described below.
Moisture content (g/100 g) = {[glass weighing dish and silica sand (g) + sample (g)] − vehicle dry weight (g)} / sample (g) × 100
Protein content determination: Kjeldahl method: Firstly, 2.9-3.6 g of sample was taken in a Celtic tube. Next, it was subjected to pyrolysis using 15 ml of concentrated sulfuric acid and 10 g of degradation accelerator (prepared by mixing copper sulfate and potassium sulfate in a 1:9 [by volume] ratio) at 420 °C for 90 minutes. After it was cooled to room temperature (about 15-30 °C), 80 ml of water was added, following which the sample was heat-distilled with 60 ml of 40% sodium hydroxide and titrated against the standard solution of 0.05 mol/L sulfuric acid. The total protein content in the sample was calculated from the titer using the following equation.
Total protein (g/100 g) = titer (mL) × F × 0.0014 × CF × 100 / amount of collected sample (g)
F = factor of 0.05 mol/L standard solution
0.0014 = nitrogen in 1 mL of 0.05 mol/L sulfuric acid (g)
CF (conversion factor from nitrogen to protein) = 6.25 for mixed food.
Fat content determination: Soxhlet extraction method: In this procedure, 7.1 to 10.6 g of sample was collected and dissolved in 100 ml of water. To this, 10-15 mL of 7% copper sulfate solution and 10-15 mL of 10% sodium hydroxide solution were added, and the mixture was filtered, washed, and dried. Ether extraction was carried out for over 16 hours until it was completely extracted. Further, the sample was collected in a bottle and the ether was evaporated in a warm bath. Next, it was dried at 105 °C for an hour, cooled for 30 minutes inside a desiccator, and weighed to accuracy in a fine weighing balance (AX205, XP204). The total fat content present in the sample was calculated from the titer as described below.
Total fat (g/100 g) = weight (g) / amount of collected sample (g) × 100
Alcohol content determination: Dichromate oxidation method: Precipitated calcium carbonate (1 g) and water (150 ml) were added to 1.0-2.5 g of the sample. This was followed by steamdistillation, from which stationary liquid was collected and diluted to 100 ml by addition of water.
To 5-10 ml of the test liquid, 5-10 g of 1/30 mol/L potassium dichromate solution and 10 mL of concentrated sulfuric acid were added and mixed, and the mixture was incubated for an hour at room temperature (about 15-30 °C). Later, 100 ml of water and 5 ml of 10% iodine-potassium iodide solution was added, followed by addition of 1% starch indicator. This was titrated using 1/20 mol/L sodium thiosulfate solution until the color of the liquid turned light blue. As a control, a similar procedure was carried out for the blank sample: 10 ml of water (a). The total alcohol content in the sample was calculated using the following equation.
Total alcohol (g/100 g) = (K × F’ − H/4 × F) × 0.0023 × dilution ratio × [constant volume of test liquid (ml) / collection quantity of test liquid (ml)] × [100 / sample collection quantity (g)]
F = potency of 1/20 mol/L sodium thiosulfate solution
F’ = potency of 1/30 mol/L potassium dichromate solution
= (a × F × 1/20)/(5 × 1/5)
A = titer of the blank (ml)
K = collection quantity of 1/30 mol/L potassium dichromate solution (ml)
H = titer of 1/20 mol/L sodium thiosulfate solution (ml)
Dietary fiber content determination: Enzymatic-gravimetric method: In this procedure, 5 g of sample was taken in a 500 mL beaker. To this, 0.05 M phosphate buffer (pH 6.0) was added until a total volume of 50 mL was achieved. Further 0.1 mL of termamyl (120 L stock) was added, and the mixture was incubated in a boiling water bath for 30 minutes. After it was cooled to room temperature (about 15-30 °C), the pH was adjusted to 7.5±0.1 by addition of 10 mL of 0.275 mol/L sodium hydroxide solution, after which it was subjected to proteolytic digestion by adding 0.1 mL of protease (50 mg/mL phosphate buffer). The reaction was incubated in a 60 °C water bath for 30 minutes. After it was cooled to room temperature (about 15-30 °C), the pH was adjusted again to 4.3±0.3 by addition of 10 mL of 0.325 mol/L hydrochloric acid. Next, 0.1 mL of amyloglucosidase enzyme was added to the reaction mixture, which was similarly incubated in a 60 °C water bath for 30 minutes. Subsequently, four times the volume of 95% ethanol (60 °C) was added to the reaction and cooled to room temperature (about 15-30 °C). The mixture was suction filtered using a glass filter followed by three washes wherein the first wash was carried out thrice using 20 mL of 78% ethanol. Second and third washes were repeated twice, using 10 ml of 95% ethanol and acetone, respectively. The residue was dried at 525 °C overnight, cooled, and weighed (R1 and R2).
Of the two residues (R1 and R2), R1 was used for analysis of ash content (A) by the ashing method and R2 was taken up for estimation of protein content (P) by the Kjeldahl method. Total dietary fiber content in the sample was determined by applying the following equation.
Total dietary fiber (g/100 g) = R × (1 − (P + A)/100 − B)/S × 100
R = total residue (g), average of R1 and R2
P = protein in residue (%)
A = ash in residue (%)
S = amount of collected sample, average (mg)
B = blank (mg)
Carbohydrate content analysis: Total carbohydrate content was determined by subtracting the known amounts (g) of moisture, protein, fat, and ash from the total weight of the sample. It is to be noted that moisture was calculated by subtracting vehicle dry weight (g) from the wet weight of the sample.
Lysine, Histidine, Phenylalanine, Leucine, Isoleucine, Valine and Threonine estimation: Amino acid automatic analysis: To 0.5 g of the collected sample, 20 ml of 20% hydrochloric acid (including 0.04% 2-mercaptoethanol) was added. After deaeration, the sample was hydrolyzed for 24 hours at 110 °C in a sealed tube. Later, the volume was made up to 100 ml with water.
Concentration process and analysis: From a total volume of 100 ml, 10 ml was fractionated, concentrated, dried by decompressing and heating, and finally dissolved in 10 ml of sodium citrate buffer (pH 2.2). This was loaded into the automatic amino acid analyzer to estimate the total amino acid content.
Dilution process and analysis: Similarly, another 10 ml was fractionated to which sodium citrate buffer (pH 2.2) was added up to a final volume of 20 ml. The sample was analyzed in the automatic amino acid analyzer.
Tryptophan estimation: High-speed liquid chromatography: To 1 g of the collected sample, 4.5 ml of water and 0.5 ml of 60% thiodiethylene glycol were added. After the mixture was dissolved by heating, it was deaerated, collected in a sealed tube, and hydrolyzed for 12 hours at 110 °C. Next, its pH was made neutral by adding 20% hydrochloric acid, and later adjusted to a slightly alkaline pH by the addition of 3 mol/L sodium hydroxide solution until a 50 ml volume was achieved. The levels of tryptophan amino acid were estimated by high-speed liquid chromatography.
Methionine estimation: Amino acid automatic analysis: 2-Mercaptoethanol component hydrochloric acid (50 ml of 0.1%) was added to 1 g of the sample, which was then hydrolyzed for 12hours at 110 °C, and later made up to a final volume of 100 ml. From this, 10 ml was fractionated and the pH was adjusted by the addition of sodium citrate buffer (pH 2.2) to a final volume of 20 ml. The levels of methionine amino acid present in the sample were analyzed using automatic amino acid analyzer.
Calories calculation:
The calculation for calories is as follows.
Calories (kcal) = protein content (g) × 4 + fat content (g) × 9 + carbohydrate content (g) × 4 + alcohol content (g) × 7
Dietary survey
Furthermore, meal contents of five households in ‘W’ village wereobserved and recorded for one week in order to understand their intake of parshot. This sample group consisted of both males andfemales of different age groups; 11 below 10 years old, eight in their teens, five in their twenties, four in their thirties, two in their forties, two in their fifties, two in their sixties, and two in their seventies. The daily intake of parshot, nech chaka, and kalala (kg) by a male Dirasha ‘A’ was measured and recorded, as he was to consume each liquor type as main meal for one week each.
Overview of research field site
The research field is a village called ‘W’ in Dirashe special worenda, which is located approximately 550 km southwest of Addis Ababa, the capital of Ethiopia (Figure 2). The 2008 statistical data of the Health Ministry of Dirashe special worenda shows that Dirashe special worenda has an area of 1,500 square kilometers with a population of approximately 130,000, most of which is inhabited by ethnic Dirashe people. This area consists of the slope of Mount Gadolla with an elevation of 2,561 m and Segen valley plateau at about 1,000 m above sea level, which stretches below Mount Gadolla. On the slope of the mountain massif and valley plateau area, ridges and terraces are made with stones and plant residues where staple crops such as sorghum and maize are cultivated. Around the mountaintop area, the climate is relatively cool/cold and humid, owing to continuous rainfall throughout the year; the average annual precipitation rate is 1,300 mm. However, most areas of Dirashe special worenda, including ‘W’ village, are located in the semiarid land, where the climate is dry with an average annual rainfall of 800 mm in two rain seasons in a year. The highest and lowest temperatures are 31 degrees and 17 degrees, respectively.
The Dirashe people consume the fermented alcoholic beverage parshot, most of the year. The brewing process of parshot is complicated (Figure 1). First, sorghum and maize flours are mixed with a small amount of dried and powdered moringa or Ethiopian kale leaves and kept for two to three months, during which lactic acid fermentation occurs. Next, it is heated to be gelatinized with sorghum flour or a mixture of sorghum and maize flour and water. Finally, powdered germinated seeds of grains such as sorghum, maize, barley (Hordeum vulgare), and wheat (Triticum aestivum) are added to promote saccharification and alcohol fermentation (Figure 1) [19]. During the short dry season in January and February, in which moringa and Ethiopian kale do not bear leaves, people brew cereal liquors called nech chaka and kalala and drink these, instead of parshot, as a staple food. The brewing process of nech chaka is similar to that of parshot (Figure 1). Sorghum and maize flours, first, go through lactic acid fermentation for one month, then are heated and gelatinized, followed by saccharification and alcohol fermentation by adding a powder of germinated seeds [19]. Kalala is made by heating and gelatinizing sorghum and maize flours, followed by saccharification and alcohol fermentation by adding powered germinated seeds (Figure 1) [19]. Two types of solid diet are also consumed by the Dirashe people. Sorghum flours are rolled into ball-shapes and boiled with moringa leaves; this is called hawarata. The other is called rakot, a disk-shaped bread, made by lactic acid fermentation of sorghum flour. These solid foods are an important source of nutrients for children below 10 years old, but the amount of intake in adults is very small; adults only snack on them in the morning and evening, together with parshot.
Results
Intake of fermented cereal liquors
Observation of the participating adults revealed that they do not consume much of solid food such as hawarata and rakot and only snack on them along with parshot, whereas children under 10 years of age are dependent on them as a principle source of energy. Specifically, adults eat hawarata and rakot in the mornings and evenings of the agricultural off-season and in the mornings of the busy season. Apart from this, they almost solely drink parshot and hardly eat or drink other kind of foods. The size of hawarata is about that of the first joint of the thumb, and local people in their late teens eat 6 to 21 pieces of hawarata per a meal. Adults consume parshot not only as a staple food, but also as a beverage; therefore, they drink parshot when they feel thirsty as well. Thus, the amount of intake of parshot is very large.
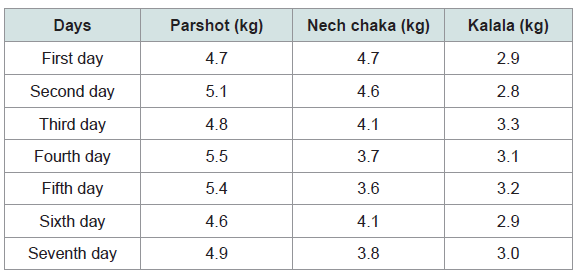
As stated above, the intake of parshot, nech chaka, and kalala per day by a Dirashe male ‘A’ in ‘W’ village was measured (Table 2). He is 31 years old, his weight is 70 kg, and height is 178 cm. His intake of each alcoholic beverage was recorded for 1 week. On average, in a day, he consumed 5 kg of parshot, regardless of whether it was parshot 1 or parshot 2, 3 kg of kalala without dilution, and 4 kg of nech chaka that was double-diluted with water.
Although the alcohol concentration of undiluted kalala and diluted parshot and nech chaka is not high, the intake of alcoholic content by Dirashe people is rather large because they drink large quantities of the liquors every day. They prefer parshot to other types of liquor and its amount of intake is the largest. In ‘W’ village, one female drinks, on average, 7 kg of parshot every day.
Daily intake of calories from fermented cereal liquors: Calorie
Calories contained per 100 g (wet weight) of each liquor are givenin Table 3. Based on these values, it was calculated that the Dirashe male ‘A’ partakes 3,400 kcal from parshot 1, 5,450 kcal from parshot 2, 1,230 kcal from kalala, and 4,760 kcal from nech chaka, when consumed as staple food.
The male ‘A’ works in the agricultural field 6 days a week during the busy farming season and 2 to 3 days a week during the off-season. The recommended daily calorie intake for him can be calculated based on the Harris Benedict Equation (1919) [20].
Firstly, given his weight, height, and age, his basal metabolism rate (BMR) is calculated as below:
BMR = 66 + (13.7 × Weight [kg]) + (5 × Height [cm]) - (6.8 × Age [yr])
Based on these numbers, a daily basal metabolism of male ‘A’ who is engaged in heavy physical labor is 1,704.20 kcal.
Secondly, his exercise level factor is calculated based on this BMR. The exercise level factor is equal to BMR × 1.375 when a test body is engaged in physical labor for 1 to 3 days a week, and BMR × 1.725 when engaged in physical labor for 6 days a week. Therefore, the exercise level factor of male ‘A’ can be calculated as BMR × 1.375 during the off-season and BMR × 1.725 during the busy farming season. Then, the recommended daily calorie intake is 2,965.62 kcal during the busy farming season and 2,343 kcal during the off-season, according to the following formula:
Daily calorie intake = Exercise level factor × 66 + {(13.7 × Weight [kg]) + (5 × Height [cm]) - (6.8 × Age [yr])}
However, calories per 100 g of kalala are as low as 44 kcal and people can only eat up to 3 kg of kalala in a day. Therefore, regardless of the season, it is difficult to take all the necessary daily calories from only kalala. On the other hand, 4.36 kg of parshot 1, 2.72 kg of parshot 2, and 2.5 kg of nech chaka are able to provide the recommended daily calories during the busy farming season. Since less calories are required during the off farming season, 3.45 kg of parshot 1, 2.15 kg of parshot 2, and 1.97 kg of nech chaka are adequate.
Daily intake of calories from fermented cereal liquors: Calorie
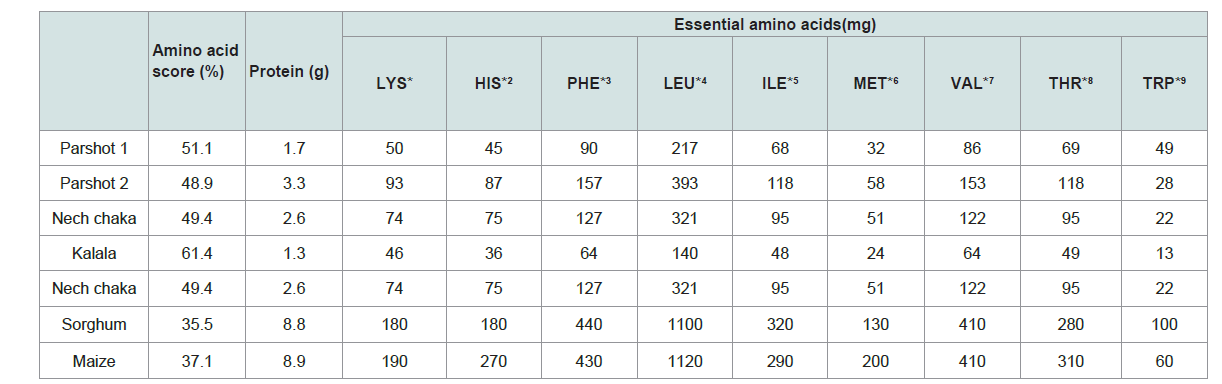
Protein contained per 100 g (wet weight) of each liquor is given in Table 4. This means that male ‘A’ consumes 85 g of protein from parshot 1, 165 g from parshot 2, 39 g from kalala, and 104 g from nech chaka in a day.
Table 4: The volumes of protein (g) and essential amino acids (mg) per 100 g (wet weight) and amino acid score (%) calculated based on those values.
Ideally, one should partake 1 gram of protein per pound13 of body weight [21], but this is difficult to achieve. People engaged in intense exercise or heavy physical labor typically require more protein compared to those with less physical activity. The latter need 0.5 to 0.65 g of protein per pound of body weight or 1.2 to 1.4 g per kilogram body weight [22,23]. Elderly people also require a substantial amount of protein-more than 50% of RDA (recommended dietary allowance) or 0.45 to 0.6 g per kilogram of body weight [24,25]. The recommended daily protein intake for a muscular male ‘A’ (weight 70 kg) who is engaged in heavy physical labor, therefore, could be calculated according to the following formula: 70 (kg) × 1.2 to 1.4 = 84 g to 98 g.
If kalala is consumed as a staple food, the total intake of protein is only 39 g and not sufficient to meet the recommended intake per day. On one hand, when nech chaka is taken as staple food, it is possible to take 104 g of protein in a day; this fulfils the minimum recommended intake shown above. On the other hand, parshot 1, when taken as a staple food, supplements 85 g of protein; this meets the minimum recommended daily intake but not the maximum. However, parshot 2 can provide 165 g of protein because the proliferation of yeast mashes progresses more compared to that in parshot 1, which, as a result, increases protein content slightly. Furthermore, ingestion of protein in parshot 2 is the most efficient among all these alcoholic beverages, and thus, the intake of 2.50 to 2.96 kg a day can supply more protein than the recommended daily intake.
Daily intake of nutrients from fermented cereal liquors: Essential amino acids
In general, the lysine content of cereal grains is the least amongst all the essential amino acids [11]. Lysin contained per 100 g (wet weight) of each liquor is given in Table 4. Based on these quantities, it can be calculated that male ‘A’ partakes 2,500 mg of lysine from parshot 1, 4,650 mg from parshot 2, 1,380 g from kalala, and 2,960 g from nech chaka daily.
FAO and WHO have set the recommended daily lysine intake as 2,100 mg for a male weighing 70 kg in his thirties (like male ‘A’) [26]. While the lysine content in kalala is low and insufficient to meet the recommended intake per day, parshot and nech chaka can supply more than the recommended intake when consumed as staple food.
Improving nutritional value by alcohol fermentation: Amino acid score
Amino acid score is a parameter for protein quantity, calculated by comparing the essential amino acid profile (mg) contained in a gram of nitrogen13 from food protein to the standard essential amino acid profile (mg) in a gram of nitrogen from body protein, and then using an essential amino acid with the lowest content ratio as a standard evaluation value. If the amino acid score is less than 100, the tested food is said to be deficit in the essential amino acid. Among the essential amino acids, one with the least content ratio is called the first-limiting amino acid [26,27].
In this study, lysine functioned as the first-limiting amino acid in every sample (Table 4). The essential amino acids contained per 100 g of each of sorghum and maize, main ingredients for parshot 1, parshot 2, kalala, and nech chaka, were quantified. The amino acid scores obtained were 35.5% for sorghum, 37.1% for maize, 51.1% for parshot 1, 48.9% for parshot 2, 61.4% for kalala, and 49.9% for nech chaka.
The amino acid scores were lower than that of high-quality protein contained in meat, fish, dairy products, and egg. It is notable, however, that the alcohol fermentation process increases lysine content in these liquors in comparison to that in their ingredients, sorghum and maize, and thus improves their amino acid scores as well (Table 3).
Conclusion
Diverse food cultures have developed around the world. In most cultures, people consume carbohydrates contained abundantly in grains such as barley, wheat, rice, sorghum, maize, finger millet, and pearl millet, potatoes such as Solanum tuberosum, taro and yam, and bananas, used as staple food. Moreover, other nutrients are supplemented by side dishes made of meat, vegetables, and beans. It is particularly important to take in meat, dairy products, and beans that contain an abundance of protein and amino acids, which constitute the human body.
However, the Dirashe people rarely consume such protein sources. They drink parshot as a staple food, which is made of sorghum and maize flours and a small amount of powdered moringa and Ethiopian kale leaves, and eat only a small quantity of solid food such as hawarata and rakot in mornings and evenings. It should be noted that the ingredients of these solid foods are the same as that of parshot-sorghum, maize, and a small quality of moringa. This indicates that the Dirashe people partake most nutrients from fermented sorghum and maize. However, similar to that in other cereal grains, protein contained in sorghum is very low in lysine content [10,28,29]. The lysine content in maize is far less than that sorghum [10,1130] which causes the quality of protein to be poor. Nevertheless, the Dirashe people do not have poor physical health due to malnutrition and many live longer than 70 years. The reasons behind this may be that the Dirashe people consume parshot in large quantities and the nutritional value of cereal grains is improved by alcohol fermentation.
Only a small number of tribes consume liquor as a staple food. Some examples are the Konso people in southern Ethiopia who drink cereal liquor called chaka [31-33] and the Manon tribe in southern Guinea who seasonally consume palm liquor as their staple food [34]. The Konso people live in the Konso region adjacent to the Dirashe region, and they descended from the Oromo ethnic group, similar to the Dirashe people. These two tribes share common aspects such as subsistence, culture, society, and language [35]. However, the Konso people also cultivate a variety of crops, including grains such as barley and wheat, potatoes, beans, and vegetables, on stone terraces cut out of the steep hillsides, in addition to sorghum, maize, and moringa [31-33]. They drink chaka as the staple food twice out of four meals a day, but also consume tuber and grain dumplings as main dish and beans and vegetables as side dish for the other two meals [32,33]. The Manon people living in the tropical rainforest area in southern Guinea drink approximately 0.8 liters of palm liquor per day when it is in season, which enables them to obtain 336 kcal. However, this is not their year-round dietary habit and they additionally consume vegetables and beans even during the palm-liquor season [34]. In this way, although both the Konso and Manon people take the fermented liquor as staple food, they also take nutrients from other food sources throughout the year.
The Dirashe people, on the other hand, drink as much as approximately 5 kg of parshot, on average, slowly over time in a day regardless of age and sex (excluding children in early teens or younger). Consuming a large quantity of fermented liquor enables them to take in about 3,400 kcal to 5,450 kcal of calories, about 85 g to 165 g of protein and about 2,500 mg to 4,650 mg of lysine, which almost fulfill the recommended daily intakes. The Dirashe people prefer drinking parshot to other types of liquor such as kalala and nech chaka because parshot has a moderate alcohol concentration, and thus has higher food safety, and is superior in flavour [19].
Besides, the fermentation process of food induces yeast mashes and bacteria to break down and resynthesize fatty acid and amino acid, which improves nutritional value [36,37] dissolves unnecessary components [36,38] eliminates labor of food preparation [36], and also facilitates portability owing to the reduced volume. This is also the case with parshot mainly made of sorghum and maize. Fermentation increases lysine content (mg), which functions as the first-limiting amino acid, and consequently improves nutritional value. Hence, the amino acid score directly related to the quality of protein is higher in parshot, kalala, and nech chaka than in sorghum, the main ingredient of these liquors (Table 4).
As stated above, the Dirashe people maintain their health by drinking the fermented cereal liquor parshot, which can function as a total nutrition food, thus taking in various nutrients at more than the recommended amount. Why has such a food culture developed, which is almost entirely dependent on fermented cereal liquor for nutritional intake? The annual rainfall in the Dirashe area is only up to 800 mm on average, with two rainy seasons. While the rainfall in the first rainy season from February to July is about 500 mm, it is only about 280 mm in the second rainy season from August to November. Furthermore, the rainfall is not stable in each season and often falls below the average except for a year of rich precipitation that comes once every several years. In addition to a shortage of rainfall, insect-mediated damage also severely affects agriculture in the area. Therefore, according to the local people, it is difficult to grow vegetables and beans that require a stable water supply. In this semiarid land, it is also difficult to provide sufficient amount of grass or plants to raise livestock such as goats and cattle, which can be a source of protein. According to an ancient local tradition that an elderly told the author, they started to cultivate sorghum, with high adaptability to the environment and disease resistance, as a staple crop since the 16th century when their ancestral Omoro people migrated and settled in the area. They have been cultivating maize only for the past few decades.
In the neighbouring areas, the aforementioned Konso people consume sorghum liquor as staple food and the Borana people drink sorghum liquor as a luxury item, both has descended from the Omoro tribe. Similarities are observed in brewing methods employed by the Dirashe people and these ethnic groups. It is very likely that the Dirashe people have developed a food culture to consume sorghum liquor as a staple food, which they had consumed as an alcoholic beverage in the past, because it was difficult to grow crops other than sorghum in the area in which they had lived for several hundred years. They manage to improve the nutritional value of sorghum by alcoholic fermentation and keep alcoholic concentration relatively low by lactic acid fermentation [19]. Owing to the low alcoholic concentration; they can consume parshot in large quantities even though spending a long time and as a result, partake the necessary amount of various nutrients.
When it comes to the issues related to health and food, we often think improving a production capacity or having a well-balanced meal as a solution. Nevertheless, the food culture of the Dirashe people, which is to consume fermented alcoholic beverage made only of sorghum and maize as a staple food and total nutrition food, not only provides us with an opportunity to investigate how human beings spread to areas where it is apparently impossible to survive, but also proposes a potential for a new food culture.
References
- Byrnes BH, Bumb BL (1998) Population growth, food production and nutrient requirements. J of Crop Produc 1: 1-27.
- Pinstrup-Andersen P (1999) Selected aspects of the future global food situation. 25th International Fertilizer Industry Association Enlarged Council Meeting. Rome, Italy.
- UNDP (1999) Human Development Report 1999. Oxford University Press, Newyork.
- FAO (1996) Food, agriculture and food security: Developments since the World Food Conference and prospects. World Food Summit; Technical Background Document No. 1. Rome, Italy.
- FAO (1999) The state of food insecurity in the world. FAO, Rome, Italy.
- Young VR (1994) Adult amino acid requirements: the case for a major revision in current recommendations. J Nutr 124(8 Suppl): 1517S-1523S.
- Reeds PJ (2000) Dispensable and indispensable amino acids for humans. J Nutr 130: 1835S-1840S.
- Ogawa R (2004) Sekai no shokubunka 11: Afurika (Food culture of world 11: Africa). Rural Culture Association Japan, Tokyo, Japan.
- Fieldhouse P (1991) Shoku to eiyou no bunkajinruigaku: Hito wa naze sore wo taberunoka (Food and nutrition: customs and culture). Trans Wani K Chuohoki Publishing Co., Ltd. Tokyo, Japan.
- Chavan JK, Kadam SS (1989) Nutritional improvement of cereals byfermentation. Crit Rev Food Sci Nutr 28: 349-400.
- Lukmanji Z, Hertzmark E, Mlingi N, Assey V, Ndossi G, et al. (2008) Tanzania Food Composition Tables. Harvard School of Public Health, Boston, USA.
- IPCC (2001) Climate change 2001: Impacts, adaptation, and vulnerability. Contribution of Working Group II to the Third Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, USA.
- UNDP (2007) Human Development Report 2007/2008: Fighting climate change: Human solidarity in a divided world. UNDP Press, Newyork.
- Jahnke HE (1982) Livestock production systems and livestock development in tropical Africa. Kieler Wissenschaftverlag Vauk, Kiel, Germany.
- Hulme M, Doherty R, Ngara T, New M, Lister D (2001) African climate change: 1900-2100. Clim Res 17: 145-168.
- Gregory PJ, Ingram JS, Brklacich M (2005) Climate change and food security. Philos Trans R Soc Lond B Biol Sci 360: 2139-2148.
- Verdin J, Funk C, Senay G, Choularton R (2005) Climate science and famine early warning. Philos Trans R Soc Lond B Biol Sci 360: 2155-2168.
- Bruins HJ, Evenari M, Nessler U (1986) Rainwater-harvesting agriculture for food production in arid zones: the challenge of the African famine. App Geo 6: 13-32.
- Sunano Y (2015) Procedure of brewing alcohol as a staple food: Case study of the fermented cereal liquor “Parshot” as a staple food in Dirashe special woreda, southern Ethiopia. Food Sci Nutr 4: 544-554.
- Harries JA, Benedict FG (1919) A biometric study of basal metabolism in man. Carnegie Institution of Washington, Washington.
- Mettler S, N Mitchell, Tipton KD (2010) Increased protein intake reduces lean body mass loss during weight loss in athletes. Med Sci Sports Exerc 42: 326-337.
- Lemon PW (1995) Do athletes need more dietary protein and amino acids? Int J Sport Nutr 5 Suppl: S39-S61.
- Phillips SM (2006) Dietary protein for athletes: from requirements to metabolic advantage. Appl Physiol Nutr Metab 31: 647-654.
- Morais JA, Chevalier, Gougeon R (2006) Protein turnover and requirements in the healthy and frail elderly. J Nutr Health Aging 10: 272-283.
- Gaffney-Stomberg E, Insogna KL, Rodriguez NR, Kerstetter JE (2009) Increasing dietary protein requirements in elderly people for optimal muscle and bone health. J Am Geriatr Soc 57: 1073-1079.
- FAO, WHO, UNU (2007) Protein and amino acid requirements in human nutrition Report of a joint FAO/WHO/UNU expert consultation (Technical Report Series 935). WHO, Geneva, Switzerland.
- Schaafsma G (2000) The protein digestibility-corrected amino acid score. J Nutr 130: 1865S-1867S.
- Taylor JR, Schussler L (1986) The protein compositions of the different anatomical parts of sorghum grain. J of Cer Sci 4: 361-369.
- Murty DS, Renard C (2001) Sorghum. In: Raemaekers, Brussels RH (Eds), Crop production in tropical Africa. Belgium, pp. 68-96.
- FAO, WHO (1973) Energy and protein requirements; Report of a Joint FAO/ WHO Ad Hoc Expert Committee. FAO, WHO, Geneva, Switzerland.
- Shinohara T (1998) Are they really greedy? Chikumashobo Ltd. Tokyo, Japan.
- Shinohara T (2000) Echiopia Konso shakai no noukou to kachiku (Agriculture and livestock in the Konso society in Ethiopia). In: Matsui T (Ed), Shizenkan no Jinruigaku [Anthropology of a view on nature]. Gajumaru Book, Okinawa,Japan, pp. 69-94.
- Shinohara T (2002) Echiopia Konso shakai niokeru noukou no shuyakusei (Specialization of agriculture in the Konso society in Ethiopia). In: Kakeai M (Ed), Ahurika noukoumin no sekai: Sono zairaisei to henyou (World of African agriculturalists: Conventionalism and transition). Kyoto University Press, Kyoto, Japan, pp. 125-162.
- Ito M (2010) Yashizake to tomoni ikiru - Ginia kyowakoku tounanbu nettairin chiiki ni okeru raffia yashi riyou- (Living with palm liquor - Utilization of raffia palm trees in the tropical forest area in southeastern Republic of Guinea). In: Kimura D, Kitanishi K (Eds), Morizumi no seitaishi - Afurika nettairin no hito, shizen, rekishi I - (Ecological record of those living in forest – People, nature and history of African tropical forest I). Kyoto University Press. Kyoto, Japan, pp. 243-261.
- Hallpike CR (2008) The Konso of Ethiopia: A study of the values of an East Cushitic people. AuthorHouse, London.
- Simango C (1997) Potential use of traditional fermented foods for weaning in Zimbabwe. Soc Sci Med 44: 1065-1068.
- Mohammed NA, Ahmed AM, Babiker EE (2011) Nutritional evaluation of sorghum flour (Sorghum bicolor L. Moench) during processing of Injera. Int J of Biol, Biom, Agri, Food and Biot Eng 5: 99-103.
- Sharma A, Kapoor AC (1996) Levels of antinutritional factors in pearl millet as affected by processing treatments and various types of fermentation. Plant Foods Hum Nutr 49: 241-252.